Scaling Novel Indole-Disulfanyl Hybrids for Next-Generation Agrochemical Applications
Scaling Novel Indole-Disulfanyl Hybrids for Next-Generation Agrochemical Applications
The agricultural sector faces escalating challenges from resistant plant bacterial diseases, necessitating the development of innovative active ingredients with novel modes of action. Patent CN111285860B introduces a groundbreaking class of indole derivatives containing disulfanyl heterocyclic structures, specifically designed to combat pathogens such as Xanthomonas oryzae and Xanthomonas axonopodis. This technology represents a significant leap forward in agrochemical intermediate design by fusing the bioactive indole core with 1,2,4-triazole or 1,3,4-oxadiazole moieties linked via a dynamic disulfide bond. For R&D directors and procurement specialists, this patent offers a robust blueprint for synthesizing high-purity intermediates that address the critical need for effective, environmentally friendly crop protection agents. The structural versatility allows for extensive SAR (Structure-Activity Relationship) optimization, ensuring that supply chains can adapt to evolving regulatory and efficacy standards.
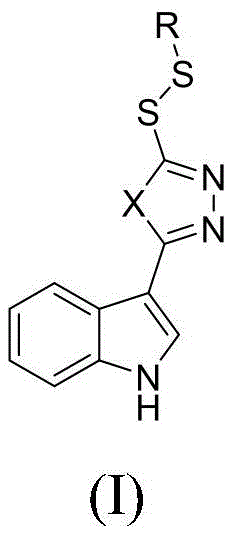
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of antibacterial agents often relies on single-heterocycle scaffolds which, while effective initially, frequently lead to rapid resistance development in target pathogens. Conventional routes to functionalized indoles typically involve harsh electrophilic substitutions that lack regioselectivity, resulting in complex impurity profiles that are costly to remove. Furthermore, older generation bactericides like bismerthiazol have raised concerns regarding toxicity and environmental persistence, forcing manufacturers to seek greener alternatives. The reliance on heavy metal catalysts or extreme reaction conditions in legacy processes also poses significant safety hazards and increases the total cost of ownership for chemical producers. These limitations create a bottleneck for reliable agrochemical intermediate suppliers who must balance yield, purity, and sustainability.
The Novel Approach
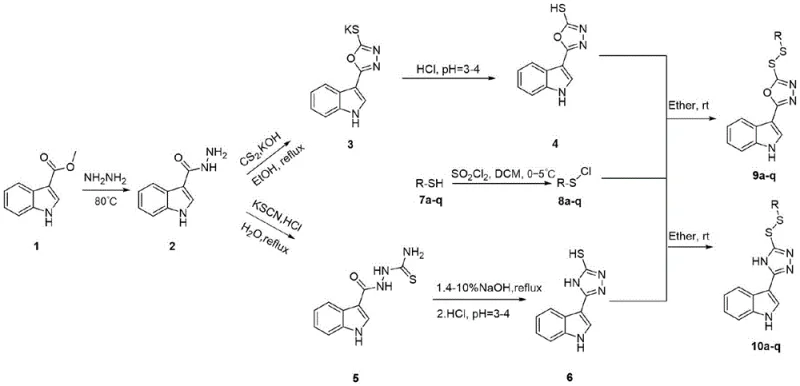
The methodology outlined in CN111285860B overcomes these hurdles through a convergent synthetic strategy that modularly assembles the indole, heterocycle, and disulfide components. By utilizing a hydrazinolysis-cyclization sequence followed by a mild oxidative coupling, the process achieves high atom economy and minimizes waste generation. The key innovation lies in the formation of the disulfide bridge under mild conditions using sulfenyl chlorides, which preserves the integrity of the sensitive indole nucleus. This approach not only simplifies the purification workflow but also enables the rapid generation of diverse libraries for biological screening. For procurement managers, this translates to a more resilient supply chain capable of delivering cost reduction in agrochemical manufacturing through streamlined operations and reduced solvent consumption.
Mechanistic Insights into Disulfide-Mediated Heterocycle Coupling
The core of this technology involves a nucleophilic substitution mechanism where the thiol group of the triazole or oxadiazole intermediate attacks the electrophilic sulfur of the sulfenyl chloride. This reaction, typically conducted in anhydrous ether at room temperature, proceeds via an SN2-like pathway to form the stable S-S bond. The choice of solvent is critical; anhydrous conditions prevent the hydrolysis of the reactive sulfenyl chloride species, thereby maximizing yield and minimizing side reactions. The preceding cyclization step, which converts the acyl hydrazide into the thiol-containing heterocycle, is driven by base-mediated intramolecular condensation. Understanding these mechanistic nuances is vital for process chemists aiming to optimize reaction kinetics and control exotherms during scale-up.
Impurity control is inherently built into this design through the crystallization properties of the intermediates. The acyl hydrazide precursor, formed via reflux with hydrazine hydrate, precipitates readily upon cooling, allowing for a simple filtration that removes unreacted esters and hydrazine salts. Similarly, the final disulfide products often exhibit distinct solubility profiles that facilitate purification via recrystallization or column chromatography. This inherent purity reduces the burden on downstream processing units and ensures that the final active ingredient meets stringent quality specifications. For supply chain heads, this predictability in impurity profiles means fewer batch failures and more consistent delivery schedules for high-purity agrochemical intermediates.
How to Synthesize 3-(5-Disulfanyl-1,2,4-triazole)-1H-indole Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing these valuable compounds, starting from commercially available methyl indole-3-carboxylate. The process is divided into three logical stages: hydrazide formation, heterocycle cyclization, and final disulfide coupling. Each stage has been optimized for yield and operational simplicity, making it accessible for both laboratory research and pilot plant production. The detailed standardized synthesis steps below outline the precise conditions required to achieve reproducible results.
- Preparation of Indole-3-carbohydrazide via refluxing methyl indole-3-carboxylate with hydrazine hydrate.
- Cyclization to form 5-(indol-3-yl)-4H-1,2,4-triazole-3-thiol or oxadiazole analogues using potassium hydroxide and carbon disulfide.
- Final coupling with sulfenyl chlorides in anhydrous ether at room temperature to establish the critical disulfide linkage.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers substantial strategic benefits for chemical manufacturers looking to optimize their portfolio. The reliance on commodity chemicals like indole-3-carboxylic acid derivatives and carbon disulfide ensures that raw material costs remain stable and predictable. Unlike processes requiring exotic catalysts or cryogenic conditions, this method operates under standard thermal conditions, significantly lowering energy consumption and capital expenditure requirements. The elimination of transition metal catalysts further simplifies the regulatory approval process for the final product, as there is no need for expensive and time-consuming heavy metal removal steps. These factors collectively contribute to a more competitive cost structure.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and complex purification trains associated with heavy metal removal. By utilizing straightforward acid-base workups and crystallization techniques, manufacturers can drastically simplify the production workflow. This reduction in unit operations directly lowers labor and utility costs, enhancing overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: The starting materials, including indole esters and various thiols, are widely available from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or pressure, ensuring consistent batch-to-batch quality. This stability allows for better inventory planning and reduces the likelihood of supply disruptions.
- Scalability and Environmental Compliance: The synthetic route avoids the use of highly toxic reagents or generates hazardous waste streams that require specialized disposal. The solvents used, such as ethanol and ether, are easily recoverable and recyclable, aligning with modern green chemistry principles. This environmental compatibility facilitates easier permitting for new production facilities and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indole derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders.
Q: What is the primary biological advantage of introducing a disulfide bond to the indole scaffold?
A: The introduction of a biologically active disulfide bond significantly enhances the compound's antibacterial and antifungal potency. According to patent data, these hybrids exhibit superior inhibition against pathogens like Xanthomonas oryzae compared to traditional single-heterocycle agents, addressing issues of drug resistance.
Q: Are the synthesis conditions suitable for large-scale industrial production?
A: Yes, the process utilizes standard reagents like hydrazine hydrate and carbon disulfide under reflux or mild room temperature conditions. The final coupling step occurs in ether at room temperature, avoiding extreme pressures or temperatures that complicate scale-up, making it highly viable for commercial manufacturing.
Q: How does this technology improve upon existing bactericides like Bismerthiazol?
A: Existing commercial agents often suffer from high toxicity and environmental persistence. These novel indole derivatives offer a greener alternative with demonstrated high inhibitory rates against rice bacterial blight and citrus canker, providing a sustainable solution for crop protection with potentially lower environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
As the demand for advanced crop protection solutions grows, partnering with an experienced CDMO is essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indole derivatives meets the highest international standards. We understand the complexities of heterocyclic chemistry and are dedicated to supporting your R&D initiatives with high-quality intermediates.
We invite you to collaborate with our technical team to explore how this technology can enhance your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us be your partner in driving innovation and efficiency in the agrochemical sector.
