Advanced Iron-Catalyzed Synthesis of Pyrroles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable methodologies for constructing heterocyclic scaffolds, which serve as the backbone of countless active pharmaceutical ingredients (APIs). Patent CN101798279B introduces a transformative approach to synthesizing pyrrole and pyrrolocyclic compounds by leveraging inexpensive ferric salts (FeX3) as catalysts, marking a significant departure from traditional noble metal-dependent processes. This innovation addresses critical pain points in modern organic synthesis, specifically the high cost and environmental toxicity associated with palladium, gold, and silver catalysts. By utilizing iron, a ubiquitous and non-toxic transition metal, this technology offers a greener alternative that maintains high efficiency and yield while drastically reducing the ecological footprint of chemical manufacturing. For R&D directors and procurement specialists, this represents a strategic opportunity to optimize supply chains and reduce the cost of goods sold (COGS) for complex heterocyclic intermediates without compromising on purity or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrroles via the [4C+1N] cycloaddition of 4-pentynones with primary amines has relied heavily on Lewis acids based on precious metals such as NaAuCl4, Na2PdCl4, AgOTf, and CuI. While these catalysts are effective, they present substantial drawbacks for large-scale commercial applications, primarily due to their exorbitant cost and the stringent regulatory limits on heavy metal residues in final drug products. The removal of trace amounts of gold, palladium, or silver often necessitates complex and expensive purification steps, such as scavenging resins or repeated recrystallizations, which lower overall process yield and increase waste generation. Furthermore, the supply chain for these noble metals is volatile and subject to geopolitical fluctuations, introducing significant risk to long-term production planning. The narrow substrate scope and prolonged reaction times often observed with these traditional catalytic systems further exacerbate the inefficiency, making them less attractive for the rapid development cycles required in modern drug discovery.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by substituting expensive noble metals with trivalent iron salts (FeX3), which act as potent Lewis acid catalysts for the cyclization reaction. This novel approach not only democratizes access to high-quality pyrrole derivatives by lowering raw material costs but also aligns perfectly with the principles of green chemistry by eliminating toxic heavy metals from the process stream. The iron-catalyzed system demonstrates remarkable versatility, accommodating a wide range of substituents on both the ynone and the amine components, thereby enabling the synthesis of diverse libraries of bioactive molecules. Operational simplicity is another key advantage, as the reaction proceeds under mild conditions in common organic solvents like toluene or benzene, facilitating easier heat management and safety protocols in a plant setting. This shift from precious to base metal catalysis represents a paradigm shift in process chemistry, offering a sustainable pathway for the manufacture of high-value heterocyclic building blocks.
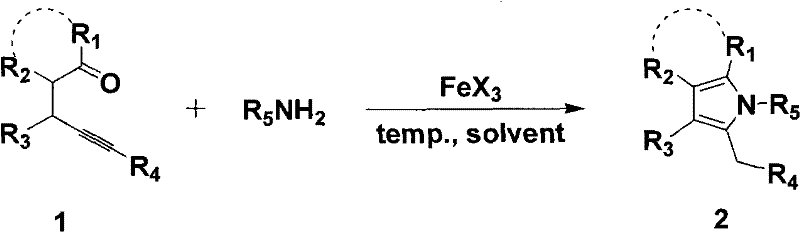
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the ability of trivalent iron species to activate the alkyne moiety of the omega,gamma-ynone substrate towards nucleophilic attack by the primary amine. Mechanistically, the iron center coordinates with the triple bond, increasing its electrophilicity and facilitating the initial addition of the amine nitrogen to form an enamine intermediate. This activation lowers the energy barrier for the subsequent intramolecular cyclization, where the carbonyl oxygen or the enol form participates in closing the five-membered pyrrole ring. Unlike softer noble metals, iron offers a unique balance of Lewis acidity and redox stability that promotes this transformation efficiently without inducing unwanted side reactions such as polymerization or over-oxidation. The catalytic cycle is robust, allowing for turnover numbers that make the process economically viable even at low catalyst loadings, typically around 10mol%. Understanding this mechanism is crucial for process chemists aiming to fine-tune reaction parameters for specific substrates, ensuring maximum conversion and minimal impurity formation during scale-up.
From an impurity control perspective, the use of iron salts simplifies the downstream processing significantly compared to noble metal catalysts. Iron residues are generally easier to remove via aqueous workup or simple filtration, reducing the burden on chromatographic purification steps which are often the bottleneck in manufacturing. The reaction profile suggests a clean conversion with high atom economy, as the primary byproducts are minimal and easily separable from the desired pyrrole product. This cleanliness is vital for pharmaceutical applications where the impurity profile must be strictly controlled to meet regulatory standards. The ability to achieve high purity directly from the reaction mixture, or with minimal purification, enhances the overall throughput of the manufacturing line. Furthermore, the compatibility of the iron catalyst with various functional groups ensures that sensitive moieties on the substrate remain intact, preserving the structural complexity required for advanced drug candidates.
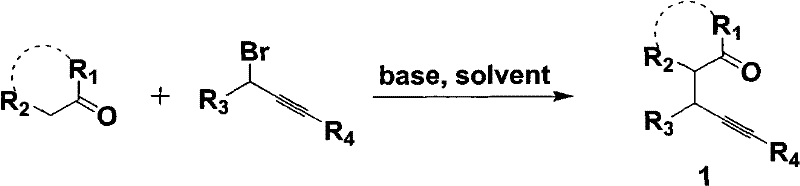
How to Synthesize Pyrrole Derivatives Efficiently
The synthetic route outlined in the patent involves a streamlined two-step sequence that begins with the preparation of the key 4-pentynone intermediate followed by the iron-catalyzed ring closure. The initial step involves the alkylation of a ketone with propargyl bromide in the presence of a mild base such as potassium carbonate, a reaction that is well-established and highly reliable in industrial settings. Once the ynone precursor is secured, it is subjected to the cyclization conditions using a primary amine and the iron catalyst in a suitable solvent. Detailed standard operating procedures for this synthesis, including specific stoichiometric ratios, temperature profiles, and workup techniques, are essential for reproducibility and quality control.
- Synthesize the 4-pentynone precursor by reacting a ketone with propargyl bromide in the presence of a base like K2CO3 in DMF.
- Mix the 4-pentynone compound with a primary amine and a catalytic amount of ferric salt (FeX3) in a non-aqueous solvent such as toluene.
- Heat the reaction mixture to 50-130°C to facilitate the ring-closing reaction, followed by standard extraction and purification to isolate the pyrrole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers tangible benefits that extend beyond mere technical feasibility, impacting the bottom line through significant cost optimization and risk mitigation. The substitution of gold or palladium with iron eliminates the exposure to volatile precious metal markets, stabilizing the cost structure of the intermediate and allowing for more accurate long-term budgeting. Additionally, the simplified purification process reduces the consumption of expensive chromatography media and solvents, further driving down the variable costs associated with production. The environmental compliance aspect cannot be overstated, as reducing heavy metal waste simplifies disposal logistics and lowers the regulatory burden on the manufacturing facility. This technology enables a more resilient supply chain by relying on commodity chemicals that are readily available globally, ensuring continuity of supply even during market disruptions.
- Cost Reduction in Manufacturing: The primary economic driver for adopting this technology is the drastic reduction in catalyst costs, as iron salts are orders of magnitude cheaper than their noble metal counterparts. By eliminating the need for expensive metal scavengers and complex purification protocols required to meet strict residual metal limits, the overall processing cost is significantly lowered. This cost efficiency allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins. The operational savings are compounded by the ability to run reactions at moderate temperatures, reducing energy consumption compared to processes requiring extreme heating or cooling.
- Enhanced Supply Chain Reliability: Relying on iron-based catalysis decouples the production process from the supply constraints of rare earth and precious metals, which are often subject to geopolitical tensions and mining limitations. Iron is one of the most abundant elements on earth, ensuring a stable and secure supply of the catalyst for decades to come. This reliability translates into consistent lead times for customers, as production schedules are less likely to be disrupted by raw material shortages. Furthermore, the robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without significant re-validation, enhancing logistical flexibility.
- Scalability and Environmental Compliance: The green chemistry credentials of this method make it highly attractive for large-scale industrial production, where waste management and environmental impact are critical concerns. The absence of toxic heavy metals simplifies the treatment of effluent streams, reducing the cost and complexity of wastewater treatment facilities. The reaction scalability is proven by the successful synthesis of various substituted pyrroles, demonstrating that the methodology holds up well when moving from gram-scale laboratory experiments to kilogram or ton-scale production. This ease of scale-up minimizes the technical risk associated with process transfer, allowing for faster time-to-market for new drug candidates.
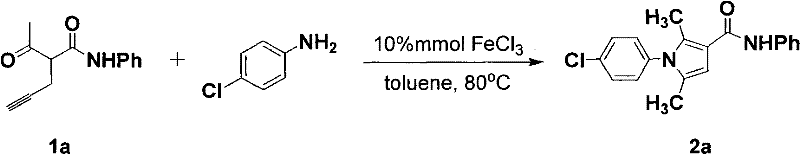
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis method, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the fit of this technology within existing manufacturing frameworks and for anticipating potential challenges during process integration.
Q: Why is iron catalysis preferred over palladium or gold for pyrrole synthesis?
A: Iron catalysts (FeX3) are significantly more cost-effective and environmentally benign compared to noble metals like Pd, Au, or Ag. They eliminate the risk of toxic heavy metal residues in pharmaceutical intermediates and simplify the purification process.
Q: What are the typical reaction conditions for this cyclization?
A: The reaction typically proceeds in non-polar solvents like toluene or benzene at temperatures ranging from 50°C to 130°C, using a catalyst loading of 0.1mol% to 30mol%, with 10mol% being optimal for most substrates.
Q: Can this method be scaled for industrial production?
A: Yes, the method is specifically designed for large-scale industrial production due to the use of inexpensive reagents, mild reaction conditions, and straightforward workup procedures that do not require specialized equipment for handling toxic metals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology and have integrated it into our extensive portfolio of custom synthesis capabilities. As a leading CDMO partner, we possess the technical expertise and infrastructure to scale diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch. We understand that in the pharmaceutical industry, consistency is key, and our robust quality management systems are designed to deliver high-purity pyrrole derivatives that meet the most demanding regulatory standards.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how switching to iron catalysis can optimize your budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline while reducing your environmental footprint. Together, we can build a more sustainable and efficient supply chain for the next generation of therapeutic agents.
