Advanced Manufacturing of m-Phenoxy Benzaldehyde: A Cost-Effective Route for Pyrethroid Intermediates
The global demand for high-efficiency, broad-spectrum pyrethroid pesticides continues to drive the need for robust supply chains of key intermediates, specifically m-phenoxy benzaldehyde. A significant technological breakthrough in this sector is detailed in patent CN112707801A, which outlines a novel preparation method that fundamentally shifts the synthetic paradigm from expensive, complex precursors to cost-effective commodity chemicals. This patent describes a streamlined four-step synthesis starting from m-cresol and halogenated benzene, achieving a remarkable total yield of 83.5% and a gas chromatography purity of 99.3%. For R&D directors and procurement strategists, this represents a critical opportunity to optimize the manufacturing of agrochemical intermediates by replacing scarce brominated aldehydes with abundant cresols, thereby securing supply continuity and drastically reducing raw material expenditure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of m-phenoxy benzaldehyde has been plagued by economic and technical inefficiencies inherent in traditional routes. One common method involves the bromination of benzaldehyde followed by condensation with sodium phenolate. However, this approach suffers from severe selectivity issues; the initial bromination generates unwanted p-bromobenzaldehyde isomers, and the subsequent alkaline condensation often triggers disproportionation reactions. As illustrated in the reaction scheme below, these side reactions produce difficult-to-remove impurities such as m-phenoxybenzoic acid and m-phenoxybenzyl alcohol, necessitating complex purification protocols that erode overall yield.
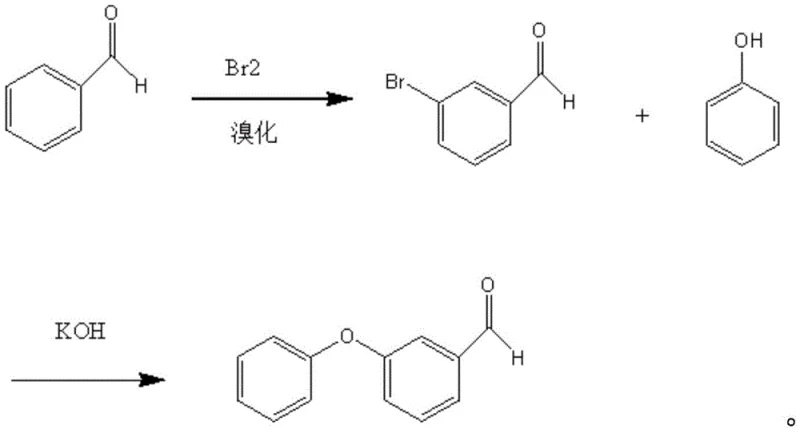
Alternative strategies, such as those disclosed in CN101337868A, attempt to mitigate these issues by protecting the aldehyde group with ethylene glycol prior to condensation. While this improves selectivity, it introduces two additional unit operations—protection and deprotection—which increase solvent consumption, extend cycle times, and lower the cumulative yield. Furthermore, other industrial methods rely on gas-phase catalytic hydrogenation of m-phenoxy benzoic acid esters. These processes require extreme operating conditions, specifically temperatures ranging from 300-450°C, posing significant safety hazards and demanding specialized, high-cost reactor infrastructure that limits scalability for many fine chemical manufacturers.
The Novel Approach
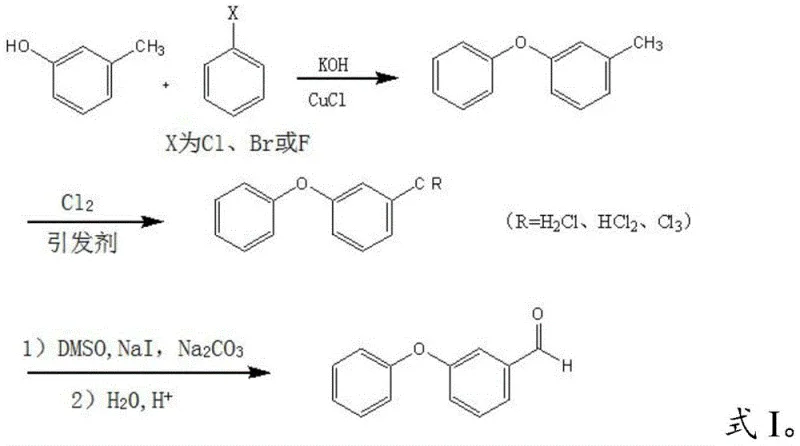
In stark contrast to these legacy methods, the technology defined in CN112707801A introduces a highly efficient pathway that bypasses aldehyde protection entirely. The core innovation lies in the construction of the ether linkage prior to the formation of the aldehyde functionality. By condensing m-cresol with halogenated benzene to form m-phenoxy toluene, the process utilizes stable, low-cost starting materials. The methyl group is then selectively chlorinated and subsequently oxidized to the aldehyde using a modified Kornblum oxidation. This strategic sequencing eliminates the risk of aldehyde disproportionation during the etherification step and avoids the harsh thermal conditions of gas-phase hydrogenation. The complete reaction sequence, demonstrating the conversion from m-cresol to the final high-purity product, is depicted in the following schematic.
Mechanistic Insights into Kornblum Oxidation and Impurity Control
The heart of this novel synthesis is the application of Kornblum oxidation to convert the chloromethyl intermediate into the target aldehyde. In this specific embodiment, the reaction utilizes dimethyl sulfoxide (DMSO) not merely as a solvent but as the primary oxidant, facilitated by a dual-catalyst system of sodium iodide (40-60% mass ratio) and sodium carbonate (15-25% mass ratio). The mechanism proceeds via a nucleophilic substitution where the iodide ion displaces the chloride to form an intermediate iodide, which is then attacked by DMSO to form a sulfonium salt. Subsequent elimination yields the aldehyde. Crucially, the reaction is conducted at a controlled temperature range of 100-150°C under nitrogen protection. This thermal window is narrow enough to prevent the over-oxidation of the aldehyde to the corresponding carboxylic acid, a common failure mode in less controlled oxidation processes, yet high enough to ensure rapid kinetics and complete conversion of the chlorinated precursor.
Impurity control is further enhanced by the specific hydrolysis protocol employed in the final stage. Following the oxidation, the reaction mixture is treated with urotropine (hexamethylenetetramine) and water to form a salt, which is then hydrolyzed using acetic acid. This "one-pot" transition from oxidation to hydrolysis minimizes the exposure of the sensitive aldehyde intermediate to harsh isolation conditions. The patent specifies monitoring the reaction via gas chromatography, terminating the hydrolysis only when the residual chlorinated precursor drops below 0.1%. This rigorous endpoint control ensures that the final crude product contains minimal halogenated impurities, directly contributing to the reported gas phase content of 99.3% without the need for extensive recrystallization or column chromatography, making it ideally suited for large-scale agrochemical manufacturing.
How to Synthesize m-Phenoxy Benzaldehyde Efficiently
The implementation of this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize the 83.5% total yield. The process begins with the Ullmann-type condensation of m-cresol and halogenated benzene, followed by radical chlorination and the critical oxidation-hydrolysis sequence. Operators must pay close attention to the removal of excess DMSO prior to the hydrolysis step to prevent side reactions. For a detailed, step-by-step breakdown of the reagent quantities, reaction times, and workup procedures required to replicate this high-yield process in a pilot or production setting, please refer to the standardized synthesis guide below.
- Condense m-cresol with halogenated benzene using cuprous chloride catalyst and potassium hydroxide at 170-190°C to form m-phenoxy toluene.
- Perform radical substitution on m-phenoxy toluene with chlorine gas and an initiator (e.g., AIBN) to generate the chlorinated methyl intermediate.
- Execute Kornblum oxidation using dimethyl sulfoxide (DMSO), sodium iodide, and sodium carbonate at 100-150°C, followed by urotropine salt formation and acidic hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN112707801A methodology offers transformative economic benefits driven by raw material arbitrage and process intensification. The most significant cost driver in traditional synthesis is the reliance on m-bromobenzaldehyde, a specialized and expensive reagent subject to volatile pricing and limited supplier availability. By switching to m-cresol and commodity halobenzenes, manufacturers can decouple their production costs from the niche bromine market, leading to substantial reductions in the Bill of Materials (BOM). Furthermore, the elimination of the aldehyde protection and deprotection steps removes the need for ethylene glycol and acidic deprotection reagents, simplifying the material inventory and reducing waste disposal costs associated with these auxiliary chemicals.
- Cost Reduction in Manufacturing: The economic advantage of this route is primarily derived from the substitution of high-value precursors with low-cost bulk chemicals. Since m-cresol is a widely produced commodity compared to functionalized benzaldehydes, the baseline material cost is significantly lower. Additionally, the process achieves a high total yield of over 83%, which means less raw material is wasted per kilogram of finished product. The removal of protection group chemistry also reduces the number of reactor batches required, lowering labor and utility consumption per unit of output, thereby enhancing the overall gross margin for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is improved by diversifying the raw material base. m-Cresol and chlorobenzene are produced at massive scales for various industries, ensuring consistent availability and reducing the risk of supply interruptions that often plague specialized fine chemical intermediates. The process also avoids the use of rare or expensive transition metal catalysts often required in cross-coupling reactions, relying instead on inexpensive cuprous chloride and sodium salts. This reliance on ubiquitous chemicals ensures that production schedules can be maintained even during periods of global chemical supply constraints.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this liquid-phase process is far superior to the high-temperature gas-phase alternatives. Operating at temperatures below 200°C eliminates the need for specialized high-pressure, high-temperature reactors, reducing capital expenditure (CAPEX) for scale-up. Moreover, the high selectivity of the Kornblum oxidation minimizes the generation of heavy organic waste and by-products like phenoxybenzoic acid. This cleaner reaction profile simplifies wastewater treatment and reduces the environmental footprint, facilitating easier regulatory compliance in jurisdictions with strict discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in CN112707801A, offering clarity on yield expectations, impurity profiles, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process optimization.
Q: What are the primary advantages of the m-cresol route over traditional bromobenzaldehyde methods?
A: The m-cresol route utilizes significantly cheaper and more readily available raw materials compared to m-bromobenzaldehyde. It eliminates the need for aldehyde protection and deprotection steps, thereby reducing operational complexity and improving total yield to over 83%.
Q: How does the new process control impurities like m-phenoxybenzoic acid?
A: By employing Kornblum oxidation under controlled temperatures (100-150°C) with specific catalysts (NaI/Na2CO3), the process selectively oxidizes the chloromethyl group to an aldehyde without over-oxidation to the carboxylic acid, which is a common issue in alkaline condensation methods.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates at moderate temperatures and pressures compared to gas-phase catalytic hydrogenation methods requiring 300-450°C. The use of standard liquid-phase reactors and common solvents like carbon tetrachloride and DMSO facilitates safe commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Phenoxy Benzaldehyde Supplier
The technological advancements detailed in patent CN112707801A underscore the potential for significant efficiency gains in the production of pyrethroid intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex laboratory methodologies into robust commercial realities. Our engineering team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications (99.3% GC) achieved in the lab are consistently met in multi-ton batches. Our rigorous QC labs utilize advanced gas chromatography and mass spectrometry to monitor every critical control point, guaranteeing that our m-phenoxy benzaldehyde meets the stringent quality requirements of global agrochemical formulators.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cost-effective synthesis route for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations that reflect the economic advantages of this next-generation manufacturing process.
