Advanced Anhydrous Synthesis of m-Phenoxy Benzaldehyde for Scalable Agrochemical Production
Introduction to Patent CN112661624B
The global demand for high-efficiency, low-toxicity pyrethroid pesticides continues to drive the need for robust supply chains of key intermediates, specifically m-phenoxy benzaldehyde. A significant technological breakthrough in this sector is detailed in Chinese Patent CN112661624B, which introduces a novel preparation method that fundamentally alters the synthetic landscape for this critical agrochemical building block. Unlike traditional routes that suffer from inherent chemical instability of the aldehyde group under alkaline conditions, this invention leverages a strictly anhydrous environment to facilitate a clean Williamson ether synthesis. By forming a phenolic salt of m-hydroxybenzaldehyde in a polar aprotic solvent and subsequently reacting it with halobenzene, the process effectively circumvents the notorious Cannizzaro disproportionation reaction. This technical advancement not only secures a yield range of 79.9-81% but also guarantees a gas phase purity of 98.9-99.3%, addressing the long-standing pain points of purification difficulty and low throughput that have plagued manufacturers for decades.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of m-phenoxy benzaldehyde has been fraught with inefficiencies and chemical hurdles that compromise both economic viability and product quality. One prevalent legacy method involves the bromination of benzaldehyde followed by condensation with phenol, a pathway riddled with selectivity issues. As illustrated in the reaction scheme below, the initial bromination step inevitably generates para-isomers alongside the desired meta-isomer, complicating downstream purification. Furthermore, the subsequent condensation typically occurs in aqueous alkaline media, where the aldehyde functionality is highly susceptible to disproportionation. This side reaction converts valuable aldehyde starting materials into unwanted carboxylic acids and alcohols, drastically reducing atom economy and creating a complex impurity profile that is difficult to separate.
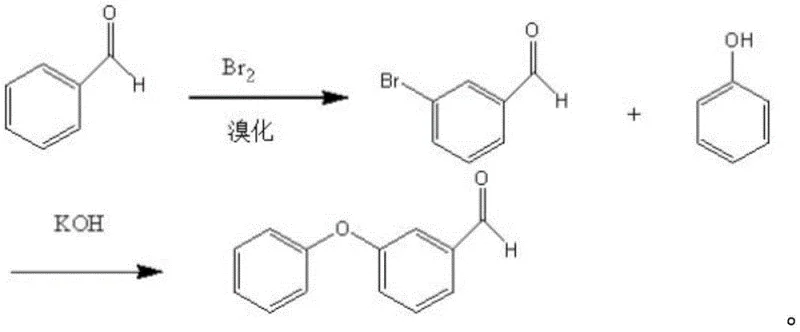
Another existing approach attempts to mitigate aldehyde instability by employing protection group chemistry, converting m-bromobenzaldehyde into an ethylene acetal prior to coupling. While this strategy protects the aldehyde, it introduces significant operational burdens, including additional reaction steps for protection and deprotection, increased solvent consumption, and the use of expensive m-bromobenzaldehyde as a feedstock. The cumulative effect of these extra unit operations is a substantial increase in production time and cost, alongside a reduction in overall yield due to losses incurred at each transformation stage. These conventional methodologies fail to meet the rigorous efficiency standards required for modern large-scale agrochemical manufacturing.
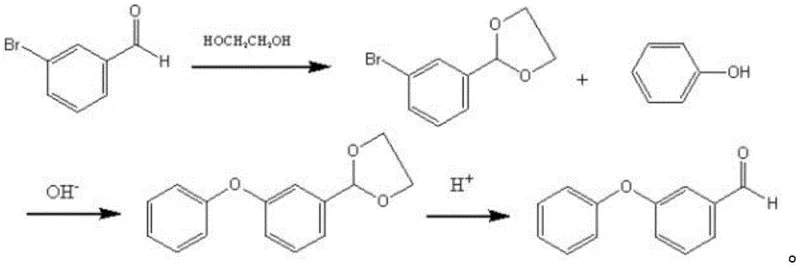
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in Patent CN112661624B offers a streamlined, direct substitution pathway that maximizes resource utilization. By initiating the reaction with m-hydroxybenzaldehyde, a readily available and cost-effective raw material, the process bypasses the need for expensive halogenated aldehyde precursors. The core innovation lies in the execution of the reaction under strictly anhydrous conditions using polar aprotic solvents such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). In this environment, the phenolic hydroxyl group is converted into a reactive phenoxide salt without triggering the hydrolysis or disproportionation of the aldehyde moiety. This strategic shift allows for a direct nucleophilic attack on halobenzene, resulting in the formation of the ether linkage with exceptional selectivity. The elimination of aqueous workups and protection groups not only simplifies the operational workflow but also significantly enhances the final product's purity profile, making it ideally suited for sensitive downstream pesticide synthesis.
Mechanistic Insights into Anhydrous Williamson Ether Synthesis
The success of this novel preparation method hinges on the precise control of the reaction microenvironment to favor nucleophilic substitution over competing degradation pathways. The mechanism begins with the deprotonation of the phenolic hydroxyl group of m-hydroxybenzaldehyde by a strong base, such as anhydrous potassium carbonate, within the polar aprotic solvent matrix. This step generates a highly nucleophilic phenoxide anion, stabilized by the solvent's ability to solvate cations while leaving the anion relatively "naked" and reactive. Crucially, the absence of water prevents the hydration of the aldehyde carbonyl, which is the precursor to the Cannizzaro reaction in basic media. As the reaction temperature is maintained between 90-100°C, the phenoxide ion attacks the electron-deficient carbon of the halobenzene (preferably bromobenzene), displacing the halide leaving group in a classic SNAr or benzyne-mediated mechanism depending on the specific substituents and conditions. This direct coupling ensures that the aldehyde group remains intact throughout the transformation, preserving the structural integrity required for subsequent pyrethroid assembly.
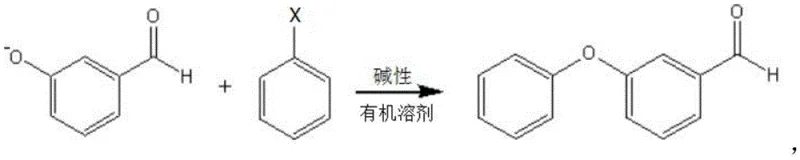
Furthermore, the impurity control mechanism is inherently built into the anhydrous design. In traditional aqueous systems, hydroxide ions act as both bases and nucleophiles, attacking the aldehyde carbonyl to initiate redox disproportionation. By replacing water with solvents like N-methylpyrrolidone (NMP) or sulfolane and utilizing carbonate bases, the concentration of free hydroxide ions capable of attacking the carbonyl carbon is minimized. The reaction pH is carefully controlled between 10 and 14, sufficient to generate the phenoxide but managed within a non-aqueous context to avoid aldehyde degradation. This results in a reaction mixture where the primary by-products are simply inorganic salts, which can be easily removed via filtration, leaving a liquid phase rich in the desired m-phenoxy benzaldehyde. The subsequent fractional distillation at 170-190°C further refines the product, removing any unreacted halobenzene or trace high-boiling impurities, delivering a specification-grade intermediate ready for immediate use.
How to Synthesize m-Phenoxy Benzaldehyde Efficiently
Implementing this advanced synthesis route requires careful attention to solvent dryness and temperature control to replicate the high yields reported in the patent literature. The process is designed to be operationally simple, relying on standard chemical engineering unit operations such as stirred tank reactions, filtration, and vacuum distillation. The following guide outlines the critical phases of the synthesis, emphasizing the transition from salt formation to the final isolation of the high-purity ether product. For laboratory or pilot-scale replication, adherence to the specified molar ratios and thermal profiles is essential to prevent the onset of side reactions.
- Perform a salt-forming reaction by mixing m-hydroxybenzaldehyde with an alkaline reagent (e.g., anhydrous potassium carbonate) in a polar aprotic solvent like DMF at 90-100°C.
- Add halobenzene (such as bromobenzene) dropwise to the salt solution and maintain the temperature at 90-100°C for 2 hours to facilitate the substitution reaction.
- Cool the reaction mixture to 30°C, perform solid-liquid separation to remove salts, and fractionate the liquid phase under reduced pressure to collect the product at 170-190°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anhydrous synthesis technology represents a strategic opportunity to optimize the cost structure and reliability of the agrochemical intermediate supply chain. The shift from multi-step protection strategies to a direct one-pot substitution significantly reduces the number of processing units required, thereby lowering capital expenditure and operational overheads. By eliminating the need for expensive protecting reagents like ethylene glycol and the associated acidic deprotection steps, the variable cost per kilogram of the final product is substantially decreased. Moreover, the use of m-hydroxybenzaldehyde as a starting material leverages a more abundant and stable feedstock compared to the volatile and costly m-bromobenzaldehyde, insulating the supply chain from raw material price fluctuations and availability risks.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly translates to lower manufacturing costs through the elimination of redundant chemical transformations. By avoiding the protection and deprotection cycles inherent in older methods, manufacturers save significantly on reagent consumption, solvent usage, and energy inputs required for heating and cooling multiple reaction stages. The high yield of nearly 80% means that less raw material is wasted, improving the overall mass balance and reducing the cost of goods sold. Additionally, the simplified workup procedure, which relies on straightforward filtration rather than complex extractions or chromatographic separations, reduces labor costs and processing time, allowing for faster batch turnover and higher facility utilization rates.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step syntheses often introduces multiple points of failure in the supply chain, where a bottleneck in one step can halt the entire production line. This novel method consolidates the synthesis into fewer, more robust steps, enhancing the overall reliability of production scheduling. The raw materials, specifically m-hydroxybenzaldehyde and common halobenzenes, are commodity chemicals with well-established global supply networks, reducing the risk of shortages. The robustness of the reaction conditions, which tolerate a range of polar aprotic solvents, provides flexibility in sourcing, ensuring that production can continue even if specific solvent supplies are constrained. This resilience is critical for maintaining continuous supply to downstream pesticide formulators who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process offers distinct advantages that align with modern green chemistry principles and regulatory requirements. The reduction in reaction steps inherently lowers the volume of wastewater and hazardous waste generated, simplifying effluent treatment and reducing disposal costs. The absence of heavy metal catalysts or toxic protecting group reagents minimizes the environmental footprint of the manufacturing site. Furthermore, the process is highly scalable; the exothermic nature of the salt formation and substitution can be managed effectively in large-scale reactors using standard cooling jackets. The ability to recover and recycle the polar aprotic solvents through distillation further enhances the sustainability profile, making this method attractive for facilities aiming to reduce their carbon intensity and meet stringent environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: Why is the anhydrous condition critical for synthesizing m-phenoxy benzaldehyde?
A: Traditional aqueous alkaline conditions often trigger the Cannizzaro disproportionation reaction of the aldehyde group, leading to significant by-products like m-phenoxybenzoic acid and m-phenoxybenzyl alcohol. The anhydrous method described in Patent CN112661624B prevents this side reaction, ensuring higher purity and yield.
Q: What are the primary advantages of using m-hydroxybenzaldehyde over m-bromobenzaldehyde as a starting material?
A: Using m-hydroxybenzaldehyde avoids the need for expensive m-bromobenzaldehyde and eliminates complex protection-deprotection steps required for the aldehyde group. This simplifies the workflow, reduces raw material costs, and minimizes waste generation.
Q: What purity levels can be achieved with this new synthesis method?
A: The patented method consistently achieves gas chromatography (GC) purity levels between 98.9% and 99.3%, with isolated yields ranging from 79.9% to 81%, significantly outperforming conventional methods which often struggle to exceed 97% purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Phenoxy Benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex organic intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the anhydrous conditions and specialized solvents required for this advanced synthesis, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process safety, we guarantee a consistent supply of high-purity m-phenoxy benzaldehyde that adheres to the highest industry standards, enabling our clients to produce next-generation pyrethroids with confidence.
We invite forward-thinking agrochemical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
