Advanced Sitagliptin Intermediate Synthesis: Scalable Routes for Global Supply Chains
The pharmaceutical landscape for Type II diabetes treatment has been significantly shaped by the introduction of Sitagliptin, a dipeptidyl peptidase-IV (DPP-4) inhibitor. As demand for this high-value API continues to grow globally, the efficiency of its supply chain relies heavily on the availability of cost-effective and scalable intermediates. Patent CN102838511A introduces a groundbreaking synthetic methodology for Sitagliptin intermediates, specifically focusing on the preparation of (S)-3-hydroxy-4-(2,4,5-trifluorophenyl) butyronitrile. This innovation addresses critical bottlenecks in traditional manufacturing by utilizing epichlorohydrin, a commercially abundant and inexpensive raw material, to establish the chiral center. Unlike conventional methods that depend on complex chiral reagents or precious metal catalysis, this approach streamlines the synthetic route, offering substantial advantages in terms of environmental impact and production economics. For procurement and supply chain leaders, understanding this technological shift is vital for securing a resilient and competitive source of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sitagliptin has been dominated by routes developed by major pharmaceutical innovators, which often involve intricate asymmetric catalytic hydrogenation steps. As illustrated in the prior art reaction schemes, these conventional methods typically require the use of expensive chiral transition metal catalysts, such as rhodium or ruthenium complexes, to induce chirality.  . These processes present significant challenges for generic manufacturers and supply chain managers. The reliance on precious metals not only inflates raw material costs but also introduces rigorous purification requirements to ensure residual metal levels meet strict regulatory standards. Furthermore, asymmetric hydrogenation reactions can be sensitive to scale-up effects, where heat transfer and mixing efficiency become critical variables that impact yield and optical purity. The need for specialized equipment and the high cost of chiral ligands create a high barrier to entry, limiting the number of qualified suppliers and potentially jeopardizing supply continuity in a volatile market.
. These processes present significant challenges for generic manufacturers and supply chain managers. The reliance on precious metals not only inflates raw material costs but also introduces rigorous purification requirements to ensure residual metal levels meet strict regulatory standards. Furthermore, asymmetric hydrogenation reactions can be sensitive to scale-up effects, where heat transfer and mixing efficiency become critical variables that impact yield and optical purity. The need for specialized equipment and the high cost of chiral ligands create a high barrier to entry, limiting the number of qualified suppliers and potentially jeopardizing supply continuity in a volatile market.
The Novel Approach
In stark contrast to the metal-dependent pathways, the methodology disclosed in CN102838511A leverages a chiral pool strategy starting from (R)-epichlorohydrin. This novel approach bypasses the need for asymmetric catalysis entirely by transferring the existing chirality from the inexpensive epoxide starting material to the target intermediate through a series of robust chemical transformations. The process begins with a Grignard reaction between a trifluorophenyl magnesium bromide and the chiral epoxide, establishing the carbon framework with high stereochemical fidelity. This shift from catalytic chirality to stoichiometric chiral sources drastically simplifies the process architecture. By eliminating the expensive hydrogenation step and the associated catalyst recovery systems, the overall operational complexity is reduced. This simplification translates directly into enhanced process reliability and lower capital expenditure for manufacturing facilities. For procurement teams, this represents a strategic opportunity to diversify the supplier base with vendors capable of executing simpler, more cost-efficient chemistry without compromising on the critical quality attributes of the intermediate.
Mechanistic Insights into Epichlorohydrin-Mediated Chiral Induction
The core of this technological advancement lies in the precise manipulation of the chiral epoxide ring to construct the Sitagliptin side chain. The reaction mechanism initiates with the formation of a Grignard reagent from 2,4,5-trifluorobromobenzene and magnesium metal in an anhydrous solvent system. 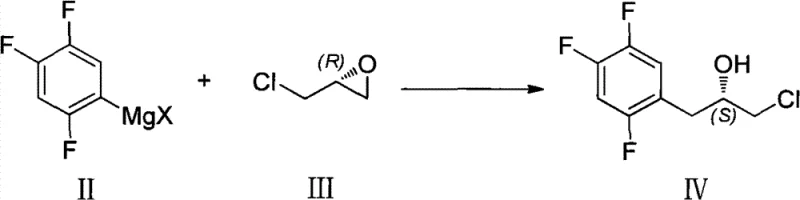 . This organometallic species then undergoes a nucleophilic attack on the less hindered carbon of the (R)-epichlorohydrin ring. This ring-opening event is highly regioselective and stereospecific, resulting in the inversion of configuration at the reaction center to yield the (S)-configured chlorohydrin intermediate. The subsequent intramolecular cyclization under alkaline conditions regenerates the epoxide ring, preserving the chiral information while displacing the chloride atom. Finally, a ring-opening reaction with a cyanating agent, such as sodium cyanide or potassium cyanide, introduces the nitrile functionality required for the final API structure. This sequence ensures that the chiral integrity established at the beginning of the synthesis is maintained throughout the pathway, delivering the target intermediate with high optical purity without the need for chiral resolution steps.
. This organometallic species then undergoes a nucleophilic attack on the less hindered carbon of the (R)-epichlorohydrin ring. This ring-opening event is highly regioselective and stereospecific, resulting in the inversion of configuration at the reaction center to yield the (S)-configured chlorohydrin intermediate. The subsequent intramolecular cyclization under alkaline conditions regenerates the epoxide ring, preserving the chiral information while displacing the chloride atom. Finally, a ring-opening reaction with a cyanating agent, such as sodium cyanide or potassium cyanide, introduces the nitrile functionality required for the final API structure. This sequence ensures that the chiral integrity established at the beginning of the synthesis is maintained throughout the pathway, delivering the target intermediate with high optical purity without the need for chiral resolution steps.
From an impurity control perspective, this route offers distinct advantages over catalytic methods. The absence of transition metal catalysts eliminates the risk of metal contamination, which is a common failure mode in regulatory audits for API manufacturing. The by-products generated in this process are primarily inorganic salts and simple organic solvents, which are easier to separate and treat compared to the complex organometallic residues found in hydrogenation routes. The use of standard unit operations such as extraction, crystallization, and filtration allows for tight control over impurity profiles. Furthermore, the reaction conditions are relatively mild, typically operating at temperatures ranging from -20°C to 60°C, which minimizes the formation of thermal degradation products. This robust impurity profile simplifies the downstream purification process, leading to higher overall yields and reduced waste generation. For R&D directors, this mechanistic clarity provides confidence in the scalability and reproducibility of the process, ensuring that the quality of the intermediate remains consistent from pilot scale to commercial production.
How to Synthesize (S)-3-hydroxy-4-(2,4,5-trifluorophenyl) butyronitrile Efficiently
The synthesis of this critical Sitagliptin intermediate involves a sequence of well-defined chemical transformations that prioritize yield and purity. The process begins with the preparation of the Grignard reagent under inert atmosphere, followed by its addition to the chiral epoxide. The resulting chlorohydrin is then cyclized to the epoxy intermediate, which serves as the substrate for the cyanation step. Each stage requires careful control of reaction parameters such as temperature, stoichiometry, and addition rates to maximize efficiency. The detailed standardized synthesis steps, including specific solvent choices, reaction times, and workup procedures, are outlined in the structured guide below to ensure reproducibility for technical teams evaluating this route for implementation.
- Preparation of Grignard Reagent: React 2,4,5-trifluorobromobenzene with magnesium powder in anhydrous THF under nitrogen protection to form the corresponding Grignard reagent.
- Chiral Induction via Epichlorohydrin: React the Grignard reagent with (R)-epichlorohydrin to introduce the chiral center, forming (S)-1-chloro-3-(2,4,5-trifluorophenyl)-2-propanol.
- Cyclization and Cyanation: Cyclize the chlorohydrin to the epoxy compound under alkaline conditions, followed by ring-opening with a cyanating agent to yield the key nitrile intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound commercial benefits that extend beyond simple chemistry improvements. For procurement managers, the primary advantage lies in the drastic reduction of raw material costs. By substituting expensive chiral catalysts and ligands with commodity chemicals like epichlorohydrin and cyanide salts, the direct material cost of the intermediate is significantly lowered. This cost structure provides a buffer against market volatility and allows for more competitive pricing in long-term supply agreements. Additionally, the simplified process flow reduces the number of processing steps, which in turn lowers labor and utility costs associated with manufacturing. The elimination of precious metal recovery systems further reduces capital and operational expenditures, making the production facility more efficient and profitable.
- Cost Reduction in Manufacturing: The economic impact of removing precious metal catalysts from the synthesis cannot be overstated. Traditional routes require significant investment in rhodium or ruthenium catalysts, which are subject to fluctuating market prices and supply constraints. By avoiding these materials, manufacturers eliminate a major cost driver and the associated logistical burden of catalyst procurement and recycling. Furthermore, the simplified purification process reduces solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective manufacturing operation. This structural cost advantage allows suppliers to offer more competitive pricing while maintaining healthy margins, creating a win-win scenario for both producers and buyers in the pharmaceutical supply chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The reagents used in this novel route, such as epichlorohydrin and trifluorobromobenzene, are widely produced commodity chemicals with robust global supply networks. This contrasts sharply with specialized chiral catalysts, which often have limited suppliers and long lead times. By relying on readily available starting materials, manufacturers can mitigate the risk of supply disruptions and ensure consistent production schedules. The robustness of the chemistry also means that production is less susceptible to batch failures caused by catalyst deactivation or sensitivity, further enhancing the reliability of supply. For supply chain heads, this translates to reduced risk of stockouts and greater confidence in meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges, particularly with sensitive catalytic reactions. The non-catalytic nature of this epichlorohydrin-based route makes it inherently more scalable, as it relies on standard chemical engineering principles rather than complex kinetic control. The reaction conditions are mild and manageable in large-scale reactors, reducing the risk of thermal runaways or mixing issues. From an environmental perspective, the absence of heavy metals simplifies waste treatment and aligns with increasingly stringent environmental regulations. The process generates less hazardous waste, reducing the environmental footprint of the manufacturing site. This compliance advantage is crucial for maintaining operational licenses and meeting the sustainability goals of global pharmaceutical customers who prioritize green chemistry in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the patent specifications and are intended to provide clarity for technical evaluators and procurement professionals. Understanding these details is essential for assessing the feasibility of integrating this intermediate into your existing supply chain and for ensuring that quality standards are met throughout the production lifecycle.
Q: How does this route reduce manufacturing costs compared to Merck's original process?
A: This route eliminates the need for expensive chiral transition metal catalysts (such as Rhodium or Ruthenium) and complex chiral auxiliaries required in prior art. By utilizing low-cost epichlorohydrin as the chiral source, raw material costs are significantly reduced, and the purification burden associated with removing heavy metal residues is completely avoided.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids sensitive asymmetric hydrogenation steps that often face scale-up challenges due to heat management and catalyst deactivation. The use of standard Grignard chemistry and simple hydrolysis steps ensures robust process control and consistent quality at commercial volumes.
Q: What is the optical purity of the intermediate produced by this method?
A: The method utilizes (R)-epichlorohydrin as a chiral pool starting material, which transfers chirality directly to the product without the need for resolution. This ensures high optical purity of the (S)-configured intermediate, meeting the stringent requirements for downstream API synthesis without additional chiral separation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
The technological potential of the epichlorohydrin-mediated synthesis route represents a significant opportunity for optimizing the Sitagliptin supply chain. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards required for API synthesis. Our capability to handle complex chiral chemistry without relying on scarce precious metals positions us as a strategic partner for long-term supply security.
We invite global pharmaceutical companies and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this efficient synthetic route, we can help you achieve substantial reductions in COGS while enhancing supply chain resilience. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to build a more sustainable and cost-effective supply chain for Sitagliptin and other critical diabetes medications, ensuring that life-saving treatments remain accessible and affordable.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
