Advanced Asymmetric Hydrogenation Technology for Commercial L-Menthol Production
The global demand for optically pure L-menthol continues to surge across the flavors, fragrances, and pharmaceutical sectors, driven by its unique cooling properties and high market value compared to its enantiomers. A pivotal advancement in this domain is documented in patent CN108083980B, which discloses a highly efficient method for preparing optically pure L-menthol through the selective hydrogenation of L-isopulegol. This technology addresses the critical limitations of natural extraction, such as climate dependency and price volatility, by offering a robust synthetic route that ensures consistent quality and supply continuity. By leveraging a specialized transition metal catalyst system combined with a novel chiral phosphine ligand, this process achieves exceptional stereoselectivity, transforming the landscape for a reliable flavors & fragrances supplier seeking to secure long-term procurement contracts. The technical breakthrough lies not only in the high yield but also in the ability to process isopulegol mixtures with varying optical purities, thereby broadening the scope of feasible raw material sources for industrial manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of L-menthol has been heavily reliant on the extraction of natural mint oils, a process inherently susceptible to agricultural uncertainties such as climatic conditions, pest infestations, and seasonal yield fluctuations. These factors lead to significant instability in both product quality and market pricing, creating substantial risks for downstream users in the food and personal care industries. Furthermore, existing synthetic routes, such as those utilizing the Rh-BINAP catalyst system starting from myrcene, often face constraints related to the availability of natural product-derived raw materials like myrcene, which are also subject to supply chain disruptions. Other chemical synthesis methods involving citral require complex purification steps, including melt crystallization, to achieve the necessary optical purity, resulting in high equipment investment, excessive energy consumption, and low space-time efficiency. These conventional approaches struggle to meet the modern industrial demand for large-scale, low-cost, and continuous stable supply, often necessitating costly post-reaction purification to remove unwanted enantiomers and impurities.
The Novel Approach
The innovative methodology outlined in the patent data introduces a paradigm shift by enabling the direct selective hydrogenation of L-isopulegol to optically pure L-menthol using a cost-effective transition metal catalyst. This approach eliminates the need for expensive and energy-intensive melt crystallization steps, as the high stereoselectivity of the catalyst ensures that the product is formed with high optical purity directly from the reaction mixture. The process is versatile enough to handle isopulegol substrates with varying enantiomeric excess, ranging from 0% to 98% ee, allowing manufacturers to utilize cheaper racemic mixtures without compromising the final product quality. By achieving conversion rates of up to 99.9% and yields of 99.8% based on L-isopulegol, this novel route drastically simplifies the production workflow and enhances overall process economics. The ability to operate under relatively mild conditions while maintaining high activity makes this method particularly suitable for the commercial scale-up of complex flavors & fragrances, offering a sustainable alternative to resource-intensive natural extraction.

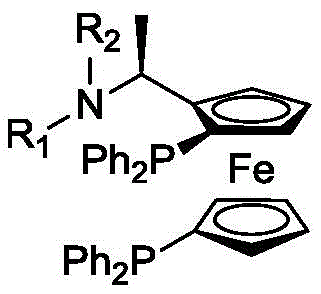
Mechanistic Insights into Ferrocene-Based Chiral Phosphine Ligand Catalysis
At the heart of this high-performance synthesis lies a sophisticated catalyst system comprising a transition metal compound, preferably rhodium, and a specifically designed chiral phosphine ligand. The ligand features a ferrocene backbone with two phosphorus atoms and variable amine substituents, creating a unique steric environment around the metal center. This structural configuration is critical for inducing the desired asymmetry during the hydrogenation process, as the steric hindrance provided by the ligand ensures that the transition metal atom specifically coordinates with the L-isopulegol isomer rather than the D-isomer. The precise spatial arrangement of the phenyl groups and the alkyl substituents on the nitrogen atom modulates the electronic properties of the phosphine, enhancing the catalyst's stability and turnover number (TON), which can reach impressive levels of 50000 to 100000. This mechanistic precision allows for the effective control of the optical purity of the catalytic hydrogenation product, ensuring that the resulting L-menthol meets the stringent 99% ee specification required for premium applications.
Furthermore, the impurity control mechanism inherent in this catalytic system is driven by the high chemoselectivity of the transition metal complex, which minimizes the formation of side products such as menthone or neomenthol. The catalyst's ability to distinguish between the subtle stereochemical differences of the isopulegol isomers means that downstream purification burdens are significantly reduced, leading to a cleaner reaction profile and higher overall process efficiency. The use of rhodium complexes, such as Rh(cod)Cl dimer, in conjunction with these ligands facilitates a smooth hydrogenation cycle where hydrogen activation and substrate insertion occur with minimal energy barriers. This deep understanding of the catalytic cycle allows process chemists to fine-tune reaction parameters, such as pressure and temperature, to maximize the yield of the desired L-enantiomer while suppressing competing reaction pathways. The result is a robust manufacturing process that delivers high-purity L-menthol with consistent batch-to-batch reproducibility, a key requirement for regulatory compliance in pharmaceutical and food-grade production.
How to Synthesize Optically Pure L-Menthol Efficiently
The implementation of this synthesis route requires careful attention to catalyst preparation and reaction conditions to fully realize the benefits of the patented technology. The process begins with the in-situ or pre-formation of the active catalyst species by combining the transition metal precursor with the chiral ligand in a suitable solvent such as toluene. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst system by combining a transition metal compound, preferably rhodium-based, with a specific chiral phosphine ligand containing a ferrocene backbone.
- Charge the reactor with isopulegol substrate, which may contain varying ratios of L- and D-isomers, and introduce the prepared catalyst system.
- Conduct selective hydrogenation under controlled pressure of 10-50 bar and temperature of 20-80°C to achieve high stereoselectivity and conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative advantages in terms of cost stability and supply security. By shifting away from agriculture-dependent natural extraction, manufacturers can decouple their production costs from the volatile fluctuations of mint crop yields and weather patterns, ensuring a predictable pricing structure for long-term contracts. The elimination of complex purification steps like melt crystallization translates to significantly reduced capital expenditure on specialized equipment and lower operational energy costs, directly impacting the bottom line. Additionally, the high turnover number of the catalyst system implies that less precious metal is required per unit of product, contributing to substantial cost savings in raw material procurement without sacrificing performance. This efficiency gain allows companies to offer more competitive pricing in the global market while maintaining healthy margins, a critical factor in the highly competitive flavors and intermediates sector.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the use of inexpensive transition metal catalysts and the ability to recycle the homogeneous catalyst system after product separation. By avoiding the need for high-energy melt crystallization and reducing the number of unit operations, the overall manufacturing footprint is minimized, leading to lower utility consumption and waste disposal costs. The high selectivity of the reaction means that raw material utilization is maximized, with minimal loss to by-products, further enhancing the economic viability of the process. These factors combine to create a lean manufacturing model that is resilient to market pressures and capable of sustaining profitability even during periods of raw material price inflation.
- Enhanced Supply Chain Reliability: Synthetic production of L-menthol via this method provides a consistent and reliable supply source that is immune to the seasonal and climatic disruptions affecting natural mint farming. The ability to use racemic or partially enriched isopulegol as a starting material broadens the supplier base for raw materials, reducing the risk of supply bottlenecks. Furthermore, the robustness of the catalyst system allows for continuous or semi-continuous operation, enabling manufacturers to maintain steady inventory levels and meet sudden spikes in demand from downstream customers. This reliability is crucial for multinational corporations that require just-in-time delivery and consistent quality to maintain their own production schedules and brand reputation.
- Scalability and Environmental Compliance: The technology is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large-scale reactors and a catalyst system that can be effectively separated and reused. The reduction in solvent usage and the elimination of energy-intensive purification steps contribute to a lower environmental footprint, aligning with increasingly strict global regulations on industrial emissions and waste. The process generates less three-waste (wastewater, waste gas, solid waste) compared to traditional methods, simplifying compliance with environmental standards and reducing the costs associated with waste treatment. This sustainability profile enhances the corporate social responsibility standing of the manufacturer, making it a preferred partner for eco-conscious global brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-menthol synthesis technology, based on the specific advantages and operational parameters detailed in the patent literature.
Q: How does this process handle racemic isopulegol mixtures?
A: The patented catalyst system exhibits high chemoselectivity, preferentially hydrogenating L-isopulegol while leaving D-isopulegol largely unreacted or allowing for easy separation, thus enabling the use of less expensive racemic starting materials.
Q: What is the optical purity achievable with this method?
A: The process consistently delivers optically pure L-menthol with an enantiomeric excess (ee) of up to 99%, meeting the stringent quality requirements for high-end flavor and pharmaceutical applications.
Q: Is the catalyst system reusable for industrial scale-up?
A: Yes, the homogeneous catalyst system can be separated from the product via distillation and reused after activation, significantly reducing catalyst consumption costs and waste generation in large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthol Supplier
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating advanced patent technologies like CN108083980B into commercial reality for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high stereoselectivity and yield demonstrated in the lab are faithfully reproduced at an industrial scale. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of L-menthol meets the exacting standards required for food, pharmaceutical, and personal care applications. We understand that the transition from pilot to production involves complex engineering challenges, and our team is dedicated to optimizing every parameter, from catalyst loading to downstream processing, to maximize efficiency and minimize cost.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this technology can enhance your product portfolio. Whether you are looking to secure a stable supply of high-purity L-menthol or explore new opportunities in chiral intermediates, NINGBO INNO PHARMCHEM is committed to delivering value through innovation, reliability, and technical excellence.
