Advanced Rhodium Catalysis for High-Purity Axial Chiral Biaryl P-Stereocenter Manufacturing
Introduction to Patent CN113292598A Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to complex chiral scaffolds, particularly those featuring axial chirality and phosphorus stereocenters which are critical for advanced ligand design. Patent CN113292598A introduces a groundbreaking methodology for the synthesis of axial chiral biaryl compounds possessing a P-stereocenter. This technology leverages a trivalent rhodium catalyst, specifically [CpXRh(III)], to facilitate an enantioselective coupling reaction between diaryl phosphonamides and diaryl acetylenes. The significance of this invention lies in its ability to construct these complex molecular architectures through double C-H bond activation, bypassing the need for pre-functionalized substrates that typically plague traditional synthetic routes. By utilizing simple and stable diaryl acetylenes as starting materials, this process not only streamlines the synthetic pathway but also enhances the overall atom economy, presenting a compelling solution for the reliable supply of high-purity pharmaceutical intermediates.
Furthermore, the patent details a robust catalytic system assisted by silver triflate or silver hexafluoroantimonate alongside silver acetate, ensuring high levels of stereocontrol. The resulting compounds exhibit stable properties and are synthesized under relatively mild reaction conditions, typically ranging from 50°C to 70°C. This technological advancement addresses the growing demand for cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and simplifying the operational workflow. For R&D directors and procurement managers alike, understanding the nuances of this Rh(III)-catalyzed transformation is essential for evaluating its potential integration into existing supply chains for chiral ligands and active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biaryl axial chiral compounds has relied heavily on the use of pre-functionalized aromatic reagents such as aryl bromides, iodides, or boronic acids. These conventional approaches often necessitate stoichiometric amounts of chiral auxiliary agents or require harsh reaction conditions to achieve the desired stereochemical outcome. The reliance on halogenated starting materials introduces significant challenges, including the generation of stoichiometric salt waste and the need for rigorous purification steps to remove metal residues. Moreover, the synthesis of the requisite arylating agents themselves can be multi-step and costly, thereby inflating the overall production expenses and extending the lead time for high-purity chiral compounds. These inefficiencies create bottlenecks in the commercial scale-up of complex polymer additives and pharmaceutical precursors, limiting the accessibility of these valuable chiral building blocks for broader industrial applications.
The Novel Approach
In stark contrast to legacy methodologies, the novel approach disclosed in CN113292598A utilizes simple diaryl acetylenes as the arylation reagent, effectively expanding the source of available starting materials. This strategy employs a dual C-H activation mechanism where both the aryl phosphonamide and the diaryl acetylene undergo activation, allowing for the direct construction of the biaryl axis with exceptional stereospecificity. 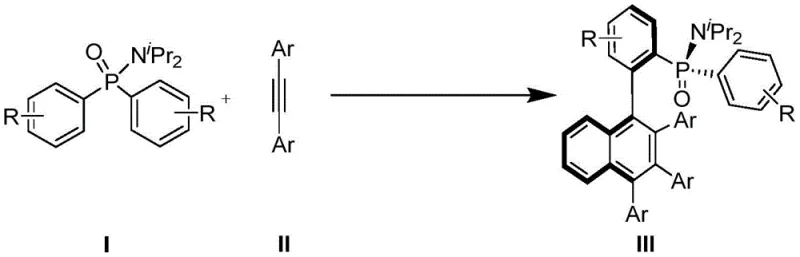 As illustrated in the reaction scheme, this method avoids the use of halogenated precursors entirely, thereby eliminating the associated waste streams and purification burdens. The process operates under mild thermal conditions and utilizes a highly active rhodium catalyst system that ensures excellent enantioselectivity and diastereoselectivity. This paradigm shift not only simplifies the synthetic route but also offers substantial cost savings by reducing the number of synthetic steps and reagent costs, making it an attractive option for the commercial production of specialized chiral ligands.
As illustrated in the reaction scheme, this method avoids the use of halogenated precursors entirely, thereby eliminating the associated waste streams and purification burdens. The process operates under mild thermal conditions and utilizes a highly active rhodium catalyst system that ensures excellent enantioselectivity and diastereoselectivity. This paradigm shift not only simplifies the synthetic route but also offers substantial cost savings by reducing the number of synthetic steps and reagent costs, making it an attractive option for the commercial production of specialized chiral ligands.
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation
The core of this innovative synthesis lies in the unique capability of the pentamethylcyclopentadienyl rhodium(III) complex, denoted as [CpXRh(III)], to mediate asymmetric C-H bond functionalization. 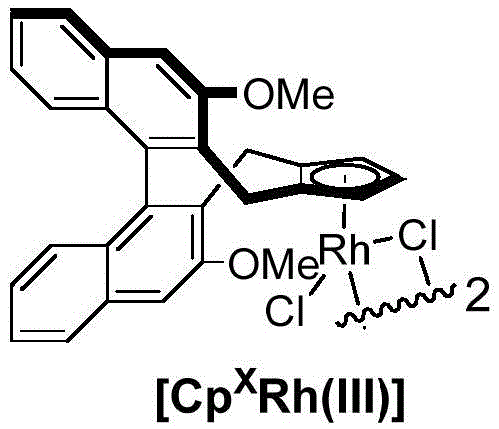 This catalyst facilitates the coordination and subsequent activation of the C-H bonds on both the phosphonamide substrate and the alkyne coupling partner. The mechanism likely proceeds through a rhodacycle intermediate formed via directed C-H metallation, followed by the insertion of the alkyne moiety. The chiral environment provided by the catalyst ligand framework dictates the facial selectivity of the alkyne insertion and the subsequent reductive elimination or protonolysis steps, ultimately forging the axially chiral biaryl bond with high fidelity. The presence of silver additives plays a crucial role in regenerating the active cationic rhodium species and facilitating the departure of leaving groups, ensuring the catalytic cycle continues efficiently.
This catalyst facilitates the coordination and subsequent activation of the C-H bonds on both the phosphonamide substrate and the alkyne coupling partner. The mechanism likely proceeds through a rhodacycle intermediate formed via directed C-H metallation, followed by the insertion of the alkyne moiety. The chiral environment provided by the catalyst ligand framework dictates the facial selectivity of the alkyne insertion and the subsequent reductive elimination or protonolysis steps, ultimately forging the axially chiral biaryl bond with high fidelity. The presence of silver additives plays a crucial role in regenerating the active cationic rhodium species and facilitating the departure of leaving groups, ensuring the catalytic cycle continues efficiently.
From an impurity control perspective, the high selectivity of this catalytic system is paramount. The reaction conditions are tuned to favor the formation of the desired atropisomer while suppressing the formation of regioisomers or racemic byproducts. The patent reports enantiomeric ratios (er) reaching up to 98:2 and diastereoselectivity ratios exceeding 19:1 in optimized examples. This level of precision minimizes the burden on downstream purification processes, such as chiral HPLC or recrystallization, which are often the most expensive and time-consuming stages in chiral synthesis. By achieving such high purity directly from the reaction pot, manufacturers can significantly reduce solvent consumption and processing time, aligning with green chemistry principles and enhancing the overall sustainability of the manufacturing process for these high-value intermediates.
How to Synthesize Axial Chiral Biaryl Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these complex molecules. The procedure involves charging a reaction vessel with the diaryl phosphonamide substrate and the diaryl acetylene coupling partner in a molar ratio preferably ranging from 1:2.5 to 1:3. To this mixture, the [CpXRh(III)] catalyst is added at a loading of 3 to 5 mol%, along with silver trifluoromethanesulfonate or silver hexafluoroantimonate (16-30 mol%), silver acetate (2.5-3 equivalents), and cyclohexanecarboxylic acid (2-3 equivalents). The reaction is conducted in an organic solvent such as dichloromethane or chlorobenzene under a nitrogen atmosphere at temperatures between 50°C and 70°C. Following the reaction period, typically around 24 hours, the solvent is removed, and the crude product is purified via silica gel chromatography.
- Combine diaryl phosphonamide, diaryl acetylene, [CpXRh(III)] catalyst, silver salt additive, silver acetate, and cyclohexanecarboxylic acid in an organic solvent under nitrogen.
- Heat the reaction mixture to 50-70°C in a closed vessel and stir for approximately 24 hours to ensure complete conversion.
- Remove the solvent via rotary evaporation and purify the crude residue using silica gel column chromatography to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rh(III)-catalyzed technology offers distinct strategic advantages over traditional synthetic routes. The primary benefit stems from the utilization of readily available and stable diaryl acetylenes as starting materials, which are generally less expensive and easier to source in bulk quantities compared to specialized aryl halides or boronic acids. This shift in raw material sourcing mitigates supply chain risks associated with volatile pricing or limited availability of halogenated precursors. Furthermore, the elimination of pre-functionalization steps reduces the overall number of unit operations required, leading to a drastically simplified manufacturing process that enhances throughput and reduces facility occupancy time.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the significant reduction in raw material costs and waste disposal expenses. By avoiding the use of stoichiometric chiral auxiliaries and halogenated reagents, the process eliminates the need for expensive waste treatment protocols associated with halide salts. Additionally, the high catalytic activity allows for lower catalyst loadings relative to the substrate, and the mild reaction conditions reduce energy consumption for heating and cooling. These factors collectively contribute to a leaner cost structure, enabling competitive pricing for the final chiral intermediates without compromising on quality or purity standards required by regulatory bodies.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining a steady supply of critical intermediates. The use of common organic solvents like dichloromethane and standard purification techniques like silica gel chromatography means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment. This flexibility enhances supply chain resilience, allowing for rapid scale-up in response to market demand fluctuations. The stability of the starting materials also permits longer storage times, reducing the risk of raw material degradation and ensuring continuous production capabilities even during logistical disruptions.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this atom-economical approach aligns perfectly with modern green chemistry mandates. The reduction in waste generation lowers the environmental footprint of the manufacturing process, simplifying compliance with increasingly stringent environmental regulations. The absence of heavy metal halide byproducts facilitates easier waste stream management and reduces the burden on effluent treatment plants. Moreover, the straightforward workup procedure involving simple solvent evaporation and filtration makes the process highly amenable to kilogram and ton-scale production, ensuring that the technology can meet the volumetric requirements of large-scale pharmaceutical and agrochemical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial production.
Q: What are the key advantages of this Rh(III)-catalyzed method over traditional arylation?
A: Unlike conventional methods requiring pre-functionalized aryl halides or boronic acids, this patent utilizes simple diaryl acetylenes via direct C-H activation, significantly improving atom economy and reducing raw material costs.
Q: What level of enantioselectivity can be achieved with this process?
A: The process demonstrates excellent stereocontrol, achieving enantiomeric ratios (er) up to 98:2 and diastereoselectivity greater than 19:1 under optimized conditions.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the reaction operates under mild temperatures (50-70°C) with readily available starting materials and standard purification techniques, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Biaryl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Rh(III)-catalyzed C-H activation technology disclosed in CN113292598A for the production of high-value chiral intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of axial chiral biaryl compounds delivered meets the highest international standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your specific project requirements. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and cost-effectiveness.
