Advanced Manufacturing of Axial Chiral Biaryls and Fluorenols for Global Pharmaceutical Supply Chains
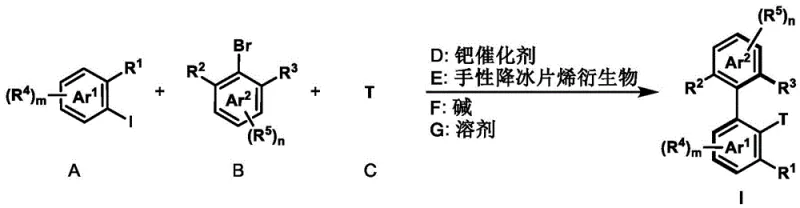
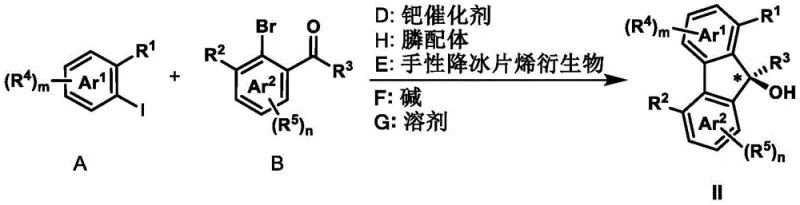
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral scaffolds essential in modern drug discovery. Patent CN111662158A introduces a groundbreaking preparation method for axial chiral biaryl compounds and chiral fluorenol compounds, addressing critical bottlenecks in the manufacturing of complex pharmaceutical intermediates. This technology leverages a cooperative palladium and chiral norbornene catalytic system to construct sterically hindered biaryl axes with exceptional stereocontrol. By utilizing simple aryl iodides and aryl bromides as starting materials, the process bypasses the need for expensive, pre-functionalized substrates often required in conventional routes. The reaction operates under mild thermal conditions ranging from 50°C to 150°C in common organic solvents, demonstrating remarkable robustness and functional group tolerance. For R&D directors and process chemists, this represents a significant leap forward in accessing privileged chiral structures that serve as backbones for novel ligands and bioactive molecules. The ability to generate these motifs efficiently positions this methodology as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to streamline their synthetic pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for constructing axial chiral biaryl skeletons have long been plagued by inherent inefficiencies and substrate limitations. Classical approaches often rely on asymmetric cross-coupling or oxidative self-coupling, which frequently necessitate the pre-installation of specific directing groups or the use of stoichiometric chiral auxiliaries. These requirements not only increase the step count and overall cost but also generate substantial chemical waste, complicating downstream purification and environmental compliance. Furthermore, methods involving dynamic kinetic resolution or desymmetrization often suffer from a theoretical maximum yield of 50% unless coupled with intricate racemization processes, severely impacting material throughput. The reliance on specialized catalysts that are difficult to synthesize or recover further exacerbates the economic burden, making large-scale production of high-purity pharmaceutical intermediates challenging. Additionally, many existing protocols exhibit poor functional group compatibility, limiting their applicability to complex molecular architectures found in late-stage drug candidates. These cumulative drawbacks create significant supply chain vulnerabilities, leading to extended lead times and inconsistent quality for critical chiral building blocks.
The Novel Approach
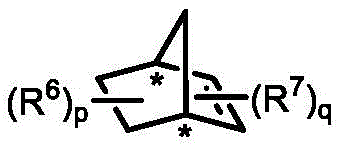
In stark contrast, the methodology disclosed in CN111662158A offers a streamlined, catalytic solution that fundamentally reshapes the synthesis of these valuable motifs. By employing a palladium-catalyzed Catellani-type reaction mechanism, this approach enables the direct coupling of readily available aryl iodides and aryl bromides with various termination reagents such as alkenes, alkynes, or boronic acids. The integration of a chiral norbornene derivative as a co-catalyst ensures precise stereochemical control, achieving enantiomeric excess (ee) values as high as 99% without the need for resolution steps. This divergent strategy allows for the simultaneous formation of the biaryl axis and the installation of diverse functional groups in a single operational sequence. The reaction conditions are notably mild, typically proceeding at temperatures between 105°C and 120°C in solvents like acetonitrile or ethylene glycol dimethyl ether, which facilitates easier handling and safer scale-up. Moreover, the system demonstrates exceptional universality, tolerating a wide array of substituents including esters, nitriles, halogens, and ethers, thereby expanding the chemical space accessible to medicinal chemists. This efficiency translates directly into cost reduction in fine chemical manufacturing by minimizing raw material costs and processing time.
Mechanistic Insights into Pd-Catalyzed Catellani-Type Reaction
The core innovation of this technology lies in the intricate interplay between the palladium catalyst and the chiral norbornene mediator, which orchestrates a series of selective C-H activation and bond-forming events. The catalytic cycle initiates with the oxidative addition of the aryl iodide to the palladium(0) species, generating an aryl-palladium(II) intermediate. This species then undergoes a crucial norbornene insertion, forming a palladium-norbornene complex that directs the subsequent ortho-C-H activation of the aryl bromide component. This step is pivotal as it establishes the steric environment necessary for axial chirality induction. The chiral norbornene derivative, depicted in the patent structures, acts as a transient mediator that temporarily bridges the two aryl rings, enforcing a specific spatial arrangement that dictates the stereochemical outcome of the final product. Following the C-H activation, the termination reagent—whether it be an alkene for Heck-type coupling or a boronic acid for Suzuki-type coupling—intercepts the palladium center. This interception leads to reductive elimination, releasing the axially chiral biaryl product and regenerating the active palladium(0) catalyst for the next turnover. In the absence of an external termination reagent, if the substrate contains an intramolecular carbonyl group, the cycle diverts to form chiral fluorenol compounds via an intramolecular nucleophilic attack, showcasing the versatility of the mechanistic pathway.
Understanding the impurity profile is critical for ensuring the quality of high-purity pharmaceutical intermediates produced via this route. The high selectivity of the norbornene-mediated C-H activation minimizes the formation of regioisomers, which are common byproducts in direct cross-coupling reactions. The use of specific phosphine ligands, such as tris(2-furyl)phosphine, further stabilizes the palladium center and suppresses side reactions like homocoupling of the aryl halides. The reaction's tolerance to various functional groups means that protecting group strategies can often be eliminated, reducing the potential for deprotection-related impurities. However, careful control of the base and solvent is essential; for instance, potassium carbonate in acetonitrile has been optimized to balance reactivity and selectivity. The purification process typically involves standard filtration through celite followed by column chromatography, which effectively removes palladium residues and ligand byproducts. This robust control over the reaction pathway ensures that the final products meet stringent purity specifications required for downstream applications in asymmetric catalysis and drug synthesis.
How to Synthesize Axial Chiral Biaryl Compounds Efficiently
The practical implementation of this synthesis protocol is designed for reproducibility and ease of operation in both laboratory and pilot plant settings. The process begins by establishing an inert atmosphere, typically using argon, to protect the sensitive palladium catalyst from oxidation. A dry reaction vessel is charged with the palladium source, such as palladium acetate, along with the base and the chiral norbornene derivative. The choice of solvent is flexible, with acetonitrile being preferred for its ability to dissolve the reactants while maintaining optimal reaction kinetics. Once the catalyst system is prepared, the aryl iodide, aryl bromide, and the chosen termination reagent are introduced. The mixture is then heated to the specified temperature range, usually around 105°C to 120°C, and stirred for a period extending from 1 to 48 hours depending on the substrate reactivity. Monitoring the reaction progress via TLC or HPLC ensures that the conversion is complete before workup. Upon completion, the mixture is cooled to room temperature and filtered to remove insoluble salts and palladium black. The filtrate is concentrated under reduced pressure, and the crude residue is purified using silica gel column chromatography to isolate the target chiral compound in high yield and enantiopurity. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture under argon protection using aryl iodide, aryl bromide, and a termination reagent such as an alkene or boronic acid.
- Add palladium catalyst (e.g., Pd(OAc)2), chiral norbornene derivative, phosphine ligand, and base (e.g., K2CO3) in an organic solvent like acetonitrile.
- Stir the mixture at 50°C to 150°C until completion, then filter, concentrate, and purify via column chromatography to obtain the target chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant simplification of the raw material supply chain. By utilizing commodity chemicals like aryl iodides and aryl bromides, which are widely available from multiple global vendors, manufacturers can mitigate the risks associated with sourcing specialized, single-source reagents. This diversification of supply sources enhances supply chain reliability and reduces the vulnerability to market fluctuations or geopolitical disruptions. Furthermore, the elimination of complex multi-step sequences required by traditional methods leads to a drastically simplified production workflow. Fewer synthetic steps mean reduced consumption of solvents, reagents, and energy, which directly correlates to substantial cost savings in manufacturing overheads. The high atom economy of the Catellani-type reaction ensures that a larger proportion of the starting mass is incorporated into the final product, minimizing waste disposal costs and aligning with green chemistry principles. Additionally, the scalability of the process, demonstrated by gram-scale examples in the patent, indicates a clear path to commercial scale-up of complex pharmaceutical intermediates without the need for extensive process re-engineering.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the use of inexpensive palladium salts and the avoidance of costly chiral resolving agents. Traditional resolution methods often discard half of the material, whereas this asymmetric synthesis produces the desired enantiomer directly with high efficiency. This intrinsic efficiency eliminates the need for recycling streams or additional separation units, thereby lowering capital expenditure and operating costs. The mild reaction conditions also reduce energy consumption compared to high-temperature or high-pressure alternatives. Moreover, the high yields reported, often exceeding 70-90%, maximize the output per batch, further driving down the unit cost of the active pharmaceutical ingredient precursors. By streamlining the synthesis, companies can achieve a more competitive pricing structure while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for continuous drug manufacturing. This method's reliance on stable, shelf-stable reagents ensures that production schedules are not disrupted by the degradation of sensitive materials. The robustness of the catalytic system against moisture and air (when properly managed) allows for more flexible manufacturing windows. The broad substrate scope means that a single platform technology can be adapted to produce a library of different chiral intermediates, reducing the need for multiple dedicated production lines. This flexibility allows supply chain planners to respond more agilely to changes in demand for different drug candidates. The consistent quality and high enantiomeric purity reduce the risk of batch failures and regulatory delays, ensuring a steady flow of materials to downstream formulation sites.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the homogeneous nature of this catalytic system facilitates smooth translation from bench to plant. The use of common solvents like acetonitrile and ethanol simplifies solvent recovery and recycling systems, which are critical for meeting environmental regulations. The reduction in waste generation, due to higher selectivity and fewer steps, lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations aiming to meet their ESG (Environmental, Social, and Governance) targets. The ability to produce large quantities of high-value chiral compounds with minimal environmental impact positions this technology as a sustainable choice for long-term production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The answers reflect the current state of the art as described in the intellectual property, ensuring accuracy and relevance for technical evaluation.
Q: What are the key advantages of this Pd-catalyzed method over traditional biaryl synthesis?
A: This method utilizes readily available aryl iodides and bromides without requiring pre-functionalized substrates. It achieves high enantioselectivity (up to 99% ee) under mild conditions (50-150°C), significantly simplifying the process compared to traditional kinetic resolution or oxidative coupling methods.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates gram-scale synthesis with high yields and excellent functional group tolerance. The use of cheap metal palladium salts and common solvents like acetonitrile supports scalability and cost-effective manufacturing for complex intermediates.
Q: What types of termination reagents are compatible with this catalytic system?
A: The system is highly versatile, accepting alkenes, alkynes, boronic acids, cyanides, and ketones as termination reagents. This flexibility allows for the diverse functionalization of the biaryl skeleton, enabling the synthesis of various chiral ligands and drug precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Biaryl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111662158A for the future of pharmaceutical manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet the rigorous quality standards demanded by the global healthcare industry. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing detailed chiral analysis and impurity profiling, guaranteeing that every batch of axial chiral biaryl or fluorenol compound adheres to stringent purity specifications. We understand that consistency and reliability are the cornerstones of a successful partnership in the fine chemical sector.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel chiral ligands or large-scale production of drug intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your molecule of interest. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can optimize your supply chain and accelerate your development timelines. Let us be your partner in turning complex chemical challenges into commercial successes.
