Advanced Synthesis of High-Purity Esketamine Hydrochloride Ketone Body for Commercial API Production
Advanced Synthesis of High-Purity Esketamine Hydrochloride Ketone Body for Commercial API Production
The pharmaceutical landscape for anesthetics and rapid-acting antidepressants has been significantly shaped by the development of Esketamine hydrochloride. Central to the efficient manufacturing of this critical active pharmaceutical ingredient (API) is the availability of high-purity intermediates, specifically the Esketamine hydrochloride ketone body, chemically known as o-chlorophenyl cyclopentyl ketone. A groundbreaking preparation method disclosed in patent CN112299972A offers a transformative approach to synthesizing this vital intermediate. This novel protocol leverages a strategic combination of acyl chlorination, metallization, and Grignard reactions to overcome the longstanding limitations of prior art. By shifting away from harsh Lewis acid catalysis and prolonged reaction times, this technology delivers a product with exceptional purity profiles and robust yields, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to support the growing global demand for Esketamine-based therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-chlorophenyl cyclopentyl ketone has been plagued by significant technical and operational hurdles that impact both cost and quality. The first conventional pathway involves a Friedel-Crafts acylation using o-chlorobenzoyl chloride and cyclopentene, catalyzed by anhydrous aluminum trichloride. While this method can achieve yields around 81.62%, it suffers from severe selectivity issues, generating substantial quantities of the elimination byproduct (2-chlorophenyl)-1-cyclopenten-1-ylmethanone. This impurity profile necessitates rigorous and costly downstream purification to meet pharmaceutical standards. Furthermore, the second traditional method, involving the addition of cyclopentyl magnesium bromide to o-chlorobenzonitrile, is operationally inefficient, requiring reaction times extending up to three days to achieve a mediocre yield of merely 68 percent. Both legacy routes rely heavily on toxic organic solvents and harsh conditions, creating environmental burdens and safety risks that are increasingly untenable in modern green chemistry frameworks.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in CN112299972A introduces a streamlined, three-step sequence that prioritizes kinetic control and selectivity. By utilizing cyclopentanecarboxylic acid and o-chlorobromobenzene as starting materials, the process constructs the carbon skeleton through a highly efficient Grignard coupling with an acid chloride intermediate. This approach not only drastically reduces reaction times but also operates under significantly milder thermal conditions, typically ranging from -10°C to 50°C depending on the specific step. The result is a dramatic enhancement in product quality, with experimental data demonstrating purities reaching 99.57% and yields climbing to 88.3%. This shift represents a paradigm change in cost reduction in API manufacturing, as it eliminates the need for expensive catalyst removal and complex impurity scrubbing, thereby simplifying the overall production workflow for commercial scale-up of complex pharmaceutical intermediates.
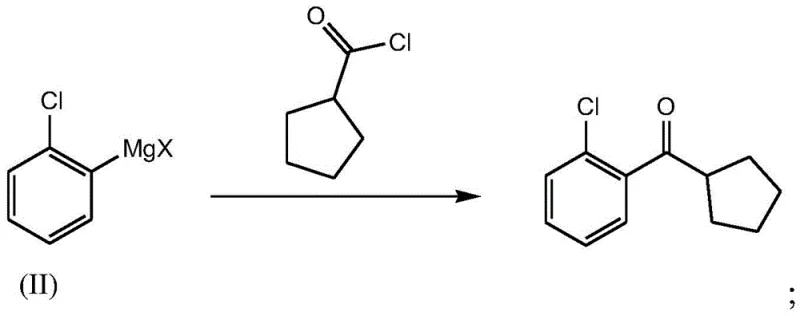
Mechanistic Insights into Grignard Coupling and Acyl Chlorination
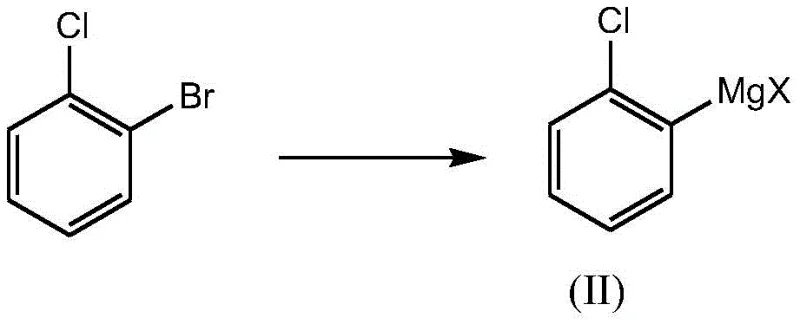
The core of this synthetic breakthrough lies in the precise orchestration of organometallic chemistry. The process initiates with the conversion of cyclopentanecarboxylic acid into cyclopentanecarbonyl chloride via acyl chlorination, typically employing thionyl chloride as the chlorinating agent. This activation step is crucial as it transforms a relatively unreactive carboxylic acid into a highly electrophilic acid chloride, primed for nucleophilic attack. Following this, the generation of the nucleophile involves the metalation of o-chlorobromobenzene. Unlike traditional Grignard formation which can be sluggish, the use of advanced reagents such as isopropyl magnesium chloride lithium chloride complexes facilitates rapid and complete metal-halogen exchange at low temperatures (0-10°C). This ensures the formation of a stable and reactive aryl magnesium species without inducing unwanted side reactions on the sensitive aromatic ring.

The final coupling stage is where the true mechanistic elegance is displayed. The nucleophilic aryl magnesium species attacks the carbonyl carbon of the cyclopentanecarbonyl chloride. This addition-elimination mechanism proceeds rapidly at low temperatures (-10 to 0°C), effectively suppressing competitive side reactions such as enolization or further addition that could lead to tertiary alcohols. The subsequent acidic quenching with dilute hydrochloric acid cleanly hydrolyzes the magnesium alkoxide intermediate to release the target ketone. Crucially, this pathway avoids the carbocation rearrangements and elimination reactions characteristic of the Friedel-Crafts route, thereby inherently minimizing the formation of the troublesome cyclopentenyl impurity. This mechanistic fidelity is the key driver behind the observed high purity, ensuring that the reducing lead time for high-purity pharmaceutical intermediates is achieved without compromising on quality specifications required for regulatory filing.
How to Synthesize o-Chlorophenyl Cyclopentyl Ketone Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits observed in the patent examples. The process is designed to be modular, allowing for the independent optimization of the acid chloride preparation and the Grignard reagent generation before the final convergence. Operators must maintain strict inert atmosphere conditions, utilizing nitrogen or argon shielding throughout the metalation and coupling steps to prevent moisture ingress which could deactivate the sensitive organometallic species. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and temperature ramps necessary to replicate the high yields reported in the intellectual property.
- Perform acyl chlorination on cyclopentanecarboxylic acid using thionyl chloride to obtain cyclopentanecarbonyl chloride.
- Conduct a metalation reaction on o-chlorobromobenzene with a Grignard reagent (e.g., isopropyl magnesium chloride lithium chloride) to generate the organometallic intermediate.
- Execute the final Grignard reaction by coupling the acid chloride with the organometallic intermediate, followed by acidic quenching and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages beyond mere technical performance. The shift to a Grignard-based coupling strategy fundamentally alters the cost structure of producing this key anesthetic intermediate. By eliminating the reliance on stoichiometric amounts of aluminum trichloride and the associated aqueous workup required to remove aluminum salts, the process significantly reduces the volume of hazardous waste generated. This translates directly into lower disposal costs and a reduced environmental footprint, aligning with increasingly stringent global sustainability mandates. Furthermore, the higher isolated yield means that less raw material is required per kilogram of finished product, driving down the variable cost of goods sold and improving margin potential for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and harsh Lewis acids removes the need for expensive chelating agents and complex purification columns often required to meet heavy metal limits in pharmaceuticals. Additionally, the shorter reaction cycles increase reactor turnover rates, allowing existing infrastructure to produce more volume without capital expenditure, leading to substantial cost savings in operational overhead.
- Enhanced Supply Chain Reliability: The starting materials, cyclopentanecarboxylic acid and o-chlorobromobenzene, are commodity chemicals with robust global supply chains, reducing the risk of raw material shortages that can plague specialized reagent-dependent processes. The mild reaction conditions also reduce the risk of thermal runaways, enhancing plant safety and ensuring consistent production schedules without unplanned downtime due to safety incidents.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, moving seamlessly from laboratory bench scales to multi-ton production campaigns. The use of common solvents like tetrahydrofuran and dichloromethane, which are easily recovered and recycled via distillation, supports a circular economy approach within the manufacturing site. This ease of solvent recovery further contributes to cost efficiency and minimizes the discharge of volatile organic compounds into the environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and process descriptions found in the source patent documentation, providing a clear view of the operational realities and benefits for potential partners.
Q: What are the primary impurities in conventional Esketamine ketone body synthesis?
A: Conventional Friedel-Crafts acylation methods often produce significant amounts of (2-chlorophenyl)-1-cyclopenten-1-ylmethanone as a byproduct, requiring complex purification steps to achieve pharmaceutical grade purity.
Q: How does the novel Grignard route improve yield compared to nitrile addition methods?
A: The novel Grignard coupling with acid chloride achieves yields up to 88.3% with purity exceeding 99.5%, significantly outperforming the traditional nitrile addition method which typically yields around 68% over extended reaction times.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method utilizes mild reaction conditions (temperatures between -10°C and 50°C) and readily available starting materials, making it highly scalable and safer for industrial manufacturing compared to harsh Lewis acid catalyzed processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chlorophenyl Cyclopentyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation anesthetics depends on the reliability and quality of the supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Esketamine ketone body meets the highest international pharmacopeial standards. We are committed to being a reliable pharmaceutical intermediate supplier that not only delivers product but also provides technical partnership to optimize your entire value chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply strategy. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your path to market is supported by the most efficient and robust chemistry available in the industry.
