Advanced Manufacturing of High-Purity Esketamine Hydrochloride Ketone Body for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for critical anesthetic intermediates, and patent CN112299972B represents a significant advancement in the manufacturing of the esketamine hydrochloride ketone body. This specific intermediate, chemically known as o-chlorophenyl cyclopentyl ketone, serves as the foundational scaffold for producing esketamine, a vital medication used for anesthesia induction and increasingly recognized for its rapid-acting antidepressant properties. The disclosed methodology shifts away from legacy processes that have long plagued manufacturers with impurity profiles and environmental concerns, offering instead a streamlined pathway characterized by exceptional chemical fidelity. By leveraging a sophisticated Grignard coupling strategy between cyclopentylcarbonyl chloride and an ortho-chlorophenyl magnesium species, the inventors have achieved a breakthrough in both yield optimization and impurity control. This technical evolution is not merely an academic exercise but a commercially viable solution that addresses the stringent quality demands of modern Good Manufacturing Practice (GMP) facilities. For global supply chain stakeholders, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent, high-quality material.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-chlorophenyl cyclopentyl ketone has relied heavily on two primary methodologies, both of which present substantial drawbacks for modern commercial production. The first conventional approach utilizes o-chlorobenzoyl chloride as a starting material in a Friedel-Crafts acylation reaction catalyzed by anhydrous aluminum trichloride. While this method can achieve yields around 81.62%, it suffers from severe selectivity issues, generating significant quantities of unwanted byproducts such as (2-chlorophenyl)-1-cyclopenten-1-ylmethanone. Furthermore, the reliance on stoichiometric amounts of Lewis acids creates massive volumes of hazardous waste that require complex and costly disposal protocols. The second traditional method involves the addition of cyclopentyl magnesium bromide to o-chlorobenzonitrile, a process that is notoriously sluggish, requiring reaction times extending up to three days to reach completion. This extended duration not only ties up reactor capacity but also results in a comparatively low yield of approximately 68%, rendering it economically inefficient for large-scale operations. Both legacy routes also depend heavily on toxic organic solvents, compounding the environmental burden and increasing the operational risks associated with solvent recovery and worker safety.
The Novel Approach
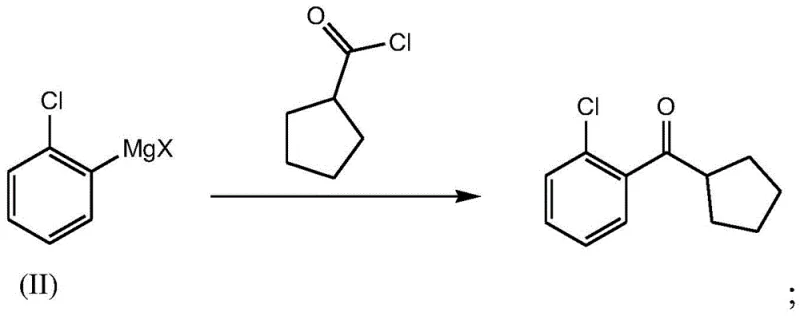
In stark contrast to these outdated techniques, the novel approach detailed in the patent introduces a highly efficient Grignard reaction between cyclopentylcarbonyl chloride and a specifically prepared organometallic intermediate. This strategy fundamentally alters the reaction landscape by utilizing a more reactive acylating agent and a highly active metalated aromatic species, allowing the coupling to proceed rapidly under mild thermal conditions. The process eliminates the need for harsh Lewis acid catalysts, thereby removing the source of the problematic alkene impurities found in Friedel-Crafts reactions. Experimental data from the patent demonstrates that this new route can achieve yields as high as 88.3% with purities reaching 99.57%, a significant improvement over the 81% and 68% benchmarks of the prior art. The operational simplicity is further enhanced by the ability to perform the reaction in common ether solvents like tetrahydrofuran, which are easier to recover and recycle than the chlorinated solvents often required in older methods. This shift represents a paradigm change in cost reduction in API manufacturing, as it directly translates to higher throughput and reduced waste treatment costs.

Mechanistic Insights into Grignard Coupling and Acyl Chlorination
The success of this synthetic route hinges on the precise execution of two critical activation steps: the conversion of cyclopentanecarboxylic acid to its corresponding acid chloride and the generation of the ortho-chlorophenyl magnesium species. The acyl chlorination step typically employs thionyl chloride as the activating reagent, reacting with the carboxylic acid at moderate temperatures of 40-50°C. This transformation is crucial because the resulting acid chloride is a much more electrophilic species than the parent acid or ester, facilitating a rapid nucleophilic attack by the Grignard reagent. The use of thionyl chloride is particularly advantageous as the byproducts, sulfur dioxide and hydrogen chloride, are gaseous and can be easily removed from the reaction mixture, driving the equilibrium forward and simplifying the workup procedure. Following this, the acid chloride is isolated or used in situ, ready for the subsequent coupling event which forms the carbon-carbon bond central to the molecule's architecture.
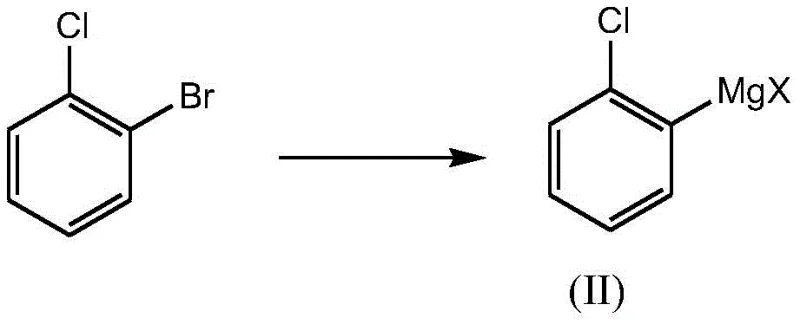
Simultaneously or sequentially, the aromatic component is activated through a metalation reaction involving o-chlorobromobenzene and a specialized Grignard reagent, preferably isopropyl magnesium chloride lithium chloride. This specific reagent, often referred to as a Turbo Grignard, exhibits superior reactivity compared to traditional magnesium turnings, allowing for rapid halogen-magnesium exchange even at low temperatures ranging from 0 to 10°C. The presence of lithium chloride enhances the solubility and stability of the organomagnesium species, preventing the formation of inactive aggregates that often plague Grignard preparations. Once formed, this nucleophilic aryl species attacks the carbonyl carbon of the cyclopentanecarbonyl chloride, forming a tetrahedral intermediate that collapses to release the magnesium chloride salt and yield the desired ketone. The entire sequence is designed to minimize side reactions, such as over-addition of the Grignard reagent to form tertiary alcohols, by carefully controlling stoichiometry and temperature, ensuring the final product meets rigorous purity specifications.
How to Synthesize o-Chlorophenyl Cyclopentyl Ketone Efficiently
The synthesis of this critical intermediate requires strict adherence to inert atmosphere techniques and precise temperature control to maximize yield and safety. The process begins with the activation of the cyclopentane ring via acyl chlorination, followed by the generation of the nucleophilic aromatic partner, and concludes with their convergent coupling. Operators must ensure that all glassware is thoroughly dried and that the reaction environment is maintained under nitrogen or argon to prevent the degradation of moisture-sensitive reagents. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution. Detailed standardized synthesis steps are provided in the guide below.
- Perform acyl chlorination on cyclopentanecarboxylic acid using thionyl chloride to obtain cyclopentanecarbonyl chloride.
- Conduct a metalation reaction on o-chlorobromobenzene using a Grignard reagent like isopropyl magnesium chloride lithium chloride to form the organometallic intermediate.
- Execute the final Grignard coupling by reacting the cyclopentanecarbonyl chloride with the organometallic intermediate, followed by acidic quenching and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic benefits that extend far beyond simple chemical yield improvements. The transition from aluminum trichloride-catalyzed processes to this Grignard-based methodology fundamentally alters the cost structure of production by eliminating the need for expensive and hazardous Lewis acid disposal. Traditional methods generate substantial amounts of aluminum sludge, the treatment of which incurs significant environmental compliance costs and logistical burdens. By replacing this with a cleaner reaction profile that produces soluble magnesium salts, the new process drastically simplifies waste management and reduces the overall environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only lowers operational expenditures but also future-proofs the supply chain against increasingly stringent global environmental regulations.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substantial increase in reaction efficiency and the simplification of downstream processing. By achieving yields exceeding 88% compared to the historical 68-81% range, manufacturers can significantly reduce the amount of raw materials required per kilogram of finished product. Furthermore, the elimination of complex purification steps needed to remove aluminum residues and alkene impurities shortens the production cycle time, allowing for greater asset utilization. The use of readily available starting materials like cyclopentanecarboxylic acid and o-chlorobromobenzene ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic reagents.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for pharmaceutical intermediates, and this method enhances reliability by utilizing robust, well-understood chemical transformations. The reaction conditions are mild, operating at temperatures between -10°C and 50°C, which reduces the risk of thermal incidents that could halt production. Additionally, the shorter reaction times—reducing a three-day process to a matter of hours—mean that manufacturing slots can be turned over more quickly, increasing the overall capacity of the production facility to meet surging demand. This agility is crucial for maintaining consistent inventory levels and preventing stockouts that could disrupt the downstream synthesis of the final active pharmaceutical ingredient.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, but this route is inherently scalable due to its reliance on homogeneous reaction conditions and standard distillation purification. The absence of viscous sludge or solid catalysts ensures that heat transfer and mixing remain efficient even in large reactors, mitigating the risks of hot spots or runaway reactions. From an environmental perspective, the process generates less hazardous waste and utilizes solvents that are easier to recover, supporting corporate sustainability goals. This combination of scalability and compliance makes the technology an ideal candidate for long-term commercial partnerships and multi-ton annual production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on purity profiles, safety considerations, and scalability potential. Understanding these details is critical for technical teams evaluating the feasibility of integrating this intermediate into their existing supply networks.
Q: How does this new method improve purity compared to traditional Friedel-Crafts acylation?
A: Traditional Friedel-Crafts methods often generate significant impurities like (2-chlorophenyl)-1-cyclopenten-1-ylmethanone due to harsh Lewis acid conditions. The novel Grignard route described in patent CN112299972B operates under milder conditions, achieving purities up to 99.57% by avoiding these specific elimination side reactions.
Q: What are the safety advantages of using Turbo Grignard reagents in this synthesis?
A: The use of isopropyl magnesium chloride lithium chloride complexes allows for rapid metalation at lower temperatures (0-10°C), reducing the risk of thermal runaway associated with traditional magnesium turnings activation. This enhances process safety and reproducibility on a commercial scale.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard unit operations such as distillation and liquid-liquid extraction. The elimination of difficult-to-remove solid catalysts like aluminum trichloride simplifies downstream processing, making it highly scalable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chlorophenyl Cyclopentyl Ketone Supplier
As the global demand for esketamine continues to rise, securing a supply partner with deep technical expertise and proven manufacturing capabilities is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN112299972B to deliver intermediates of unparalleled quality. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on integrity. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this superior manufacturing process. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
