Advanced Synthetic Route for High-Purity Piperidine Intermediates in JAK Inhibitor Manufacturing
The pharmaceutical landscape for treating Rheumatoid Arthritis (RA) has been significantly transformed by the advent of JAK inhibitors, with Tofacitinib standing out as a pivotal therapeutic agent. The efficient and scalable production of its key intermediates remains a critical challenge for global supply chains. Patent CN103755624A discloses a groundbreaking synthetic method for a specific piperidine derivative, which serves as a crucial building block in the manufacture of Tofacitinib. This innovation addresses long-standing inefficiencies in chiral resolution, offering a pathway to higher yields and superior purity. By leveraging a novel strategy involving the introduction of an S-configured substituent prior to resolution, this method fundamentally alters the thermodynamics of salt formation, enabling the use of inexpensive L-tartaric acid to achieve results previously only possible with costly reagents.
For R&D directors and process chemists, the structural integrity and stereochemical purity of the intermediate are paramount. The target molecule, a tartrate salt of a compound with structural formula (I), features a specific (3R,4R) configuration that is essential for the biological activity of the final drug. 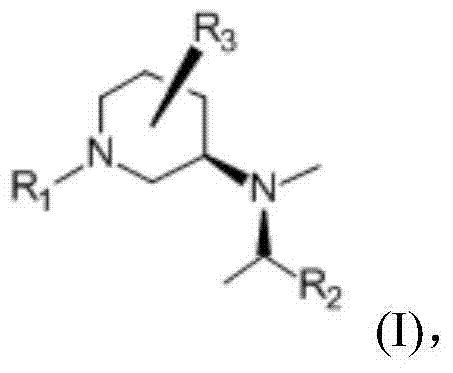 The patent details how this derivative is obtained through the reaction of a precursor compound (Formula II) with L-tartaric acid, followed by precise separation techniques. This approach not only streamlines the synthesis but also ensures that the final product meets the rigorous quality standards required for active pharmaceutical ingredient (API) production, making it a highly attractive candidate for commercial scale-up.
The patent details how this derivative is obtained through the reaction of a precursor compound (Formula II) with L-tartaric acid, followed by precise separation techniques. This approach not only streamlines the synthesis but also ensures that the final product meets the rigorous quality standards required for active pharmaceutical ingredient (API) production, making it a highly attractive candidate for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral piperidine intermediates for Tofacitinib has been plagued by low efficiency and high costs. Traditional methods often rely on the use of expensive resolving agents such as L-di-p-toluoyl-L-tartaric acid (L-DPTT). While effective to a degree, these conventional routes suffer from significant drawbacks, primarily a low separation yield that hovers around 42%. This inefficiency means that more than half of the valuable material is lost or requires extensive recycling, driving up the cost of goods sold (COGS). Furthermore, the reliance on complex, bulky resolving agents complicates the downstream processing and waste management, creating bottlenecks in large-scale manufacturing. Another prior art method involves reductive amination followed by manual separation, which yields less than 18%, rendering it commercially unviable for high-volume production.
The Novel Approach
The methodology described in CN103755624A represents a paradigm shift in chiral resolution technology. By strategically introducing an S-configured substituent, specifically an (S)-N-methyl-1-phenylethylamino group, to the amino position at the 3-position of the piperidine ring, the inventors have created a diastereomeric environment that is far more conducive to resolution. 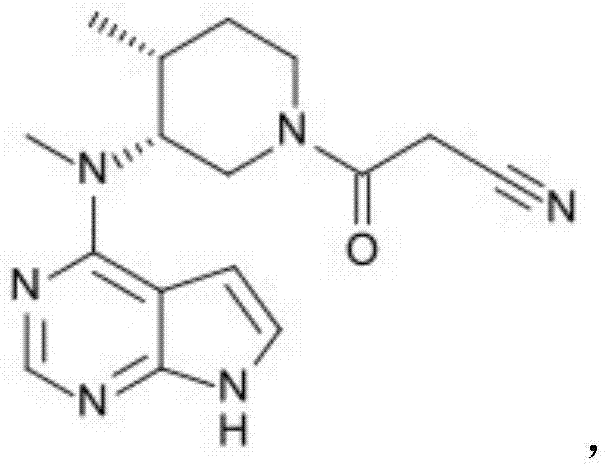 This modification dramatically enhances the binding affinity and selectivity when reacting with L-tartaric acid. Consequently, the yield of the desired tartrate salt skyrockets to over 70%, nearly doubling the efficiency of previous methods. This novel approach not only utilizes a cheap, abundant, and environmentally benign resolving agent but also simplifies the purification process, allowing for the direct crystallization of high-purity intermediates without the need for complex chromatographic separations.
This modification dramatically enhances the binding affinity and selectivity when reacting with L-tartaric acid. Consequently, the yield of the desired tartrate salt skyrockets to over 70%, nearly doubling the efficiency of previous methods. This novel approach not only utilizes a cheap, abundant, and environmentally benign resolving agent but also simplifies the purification process, allowing for the direct crystallization of high-purity intermediates without the need for complex chromatographic separations.
Mechanistic Insights into Chiral Resolution via L-Tartaric Acid
The core of this technological advancement lies in the stereoselective interaction between the modified piperidine amine and L-tartaric acid. The process begins with the reductive amination of a 4-methyl-3-piperidone derivative with a chiral amine, such as (S)-N-methyl-1-phenylethylamine. This step installs a chiral auxiliary that dictates the spatial arrangement of the molecule. When this mixture is subsequently treated with L-tartaric acid in a solvent like ethanol, a diastereomeric salt is formed. The specific spatial configuration of the (3R,4R) isomer allows for a more stable crystal lattice formation with the tartrate anion compared to its enantiomeric counterparts. 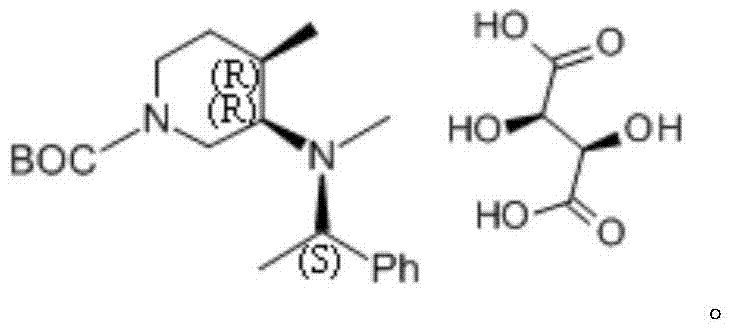 This difference in solubility and crystallization kinetics is the driving force behind the high separation efficiency. The patent specifies that heating the mixture to dissolve the solids followed by cooling to below 10°C induces the selective precipitation of the desired isomer, leaving impurities and the unwanted enantiomer in the mother liquor.
This difference in solubility and crystallization kinetics is the driving force behind the high separation efficiency. The patent specifies that heating the mixture to dissolve the solids followed by cooling to below 10°C induces the selective precipitation of the desired isomer, leaving impurities and the unwanted enantiomer in the mother liquor.
Following the resolution, the chiral auxiliary must be removed to reveal the free amine necessary for the final coupling reaction. This is achieved through a catalytic hydrogenation step using palladium-carbon under nitrogen protection. The cleavage of the benzyl-type bond in the chiral auxiliary restores the secondary amine functionality while retaining the established (3R,4R) stereochemistry at the piperidine ring. The result is a highly pure intermediate, typically exhibiting a chemical purity of greater than 99% and an enantiomeric excess (ee) of greater than 99%. This level of purity is critical for preventing the formation of genotoxic impurities or inactive isomers in the final API, ensuring patient safety and regulatory compliance.
How to Synthesize (3R,4R)-Piperidine Derivative Efficiently
The synthesis of this high-value intermediate involves a sequence of well-defined chemical transformations that are robust and scalable. The process starts with the protection of the piperidine nitrogen, followed by the key reductive amination with the chiral amine source. The subsequent resolution step with L-tartaric acid is the heart of the process, where careful control of temperature and stoichiometry ensures maximum recovery of the desired isomer. Finally, deprotection and coupling with the pyrrolopyrimidine core complete the synthesis of the Tofacitinib precursor. For a detailed, step-by-step breakdown of the reaction conditions, molar ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Perform reductive amination of 4-methyl-3-piperidone derivative with (S)-N-methyl-1-phenylethylamine using a reducing agent like sodium borohydride acetate.
- React the resulting amine mixture with L-tartaric acid in an organic solvent such as ethanol, heating to dissolve and then cooling to crystallize the tartrate salt.
- Separate the precipitated (3R,4R)-tartrate salt, followed by hydrogenation to remove the chiral auxiliary and obtain the pure piperidine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and operational benefits. The most significant advantage is the drastic reduction in raw material costs associated with the resolving agent. Replacing expensive, specialized resolving agents like L-DPTT with commodity-grade L-tartaric acid results in substantial cost savings per kilogram of produced intermediate. Additionally, the near-doubling of the resolution yield from roughly 42% to over 70% means that less starting material is required to produce the same amount of final product, further driving down the overall manufacturing cost. This efficiency gain translates directly into a more competitive pricing structure for the final API.
- Cost Reduction in Manufacturing: The elimination of costly chiral resolving agents and the significant improvement in yield create a leaner manufacturing process. By avoiding the need for expensive reagents and reducing the volume of waste generated from low-yield steps, the overall production cost is significantly lowered. This makes the supply of Tofacitinib intermediates more economically sustainable and resilient against fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: L-tartaric acid is a widely available, bulk commodity chemical, unlike specialized resolving agents which may have limited suppliers and longer lead times. Switching to a ubiquitous reagent mitigates the risk of supply disruptions. Furthermore, the simplified process flow reduces the number of unit operations, decreasing the potential for batch failures and ensuring a more consistent and reliable supply of high-quality intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of common solvents like ethanol and the avoidance of heavy metal catalysts in the resolution step align with green chemistry principles. The process is inherently safer and easier to scale from pilot plant to commercial production volumes. The reduced waste stream and the use of biodegradable resolving agents simplify wastewater treatment and environmental compliance, reducing the regulatory burden and associated disposal costs for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of implementing this route in a commercial setting.
Q: What is the primary advantage of using L-tartaric acid over L-DPTT for resolution?
A: The use of L-tartaric acid significantly improves the resolution yield to over 70%, compared to approximately 42% with L-DPTT, while utilizing a much more cost-effective and readily available resolving agent.
Q: How does the introduction of an S-configured substituent affect the synthesis?
A: Introducing an S-configured group, such as (S)-N-methyl-1-phenylethylamine, at the 3-position amino group enhances the binding capacity with L-tartaric acid, thereby drastically improving the separation efficiency of the desired (3R,4R) enantiomer.
Q: What represents the final purity and ee value achievable with this method?
A: This synthetic route consistently achieves a chemical purity greater than 99% and an enantiomeric excess (ee) greater than 99% for the final piperidine intermediate, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tofacitinib Intermediate Supplier
The synthetic methodology outlined in CN103755624A represents a significant leap forward in the production of JAK inhibitor intermediates, offering a blend of high efficiency, superior purity, and cost-effectiveness. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of piperidine derivative meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can enhance your project's success and profitability.
