Scalable Production of High-Purity Tofacitinib Intermediates via Optimized Chiral Control
Scalable Production of High-Purity Tofacitinib Intermediates via Optimized Chiral Control
The pharmaceutical landscape for Janus Kinase (JAK) inhibitors continues to evolve, with Tofacitinib citrate remaining a cornerstone therapy for rheumatoid arthritis and ulcerative colitis. As global demand surges, the efficiency and purity of its synthetic intermediates have become critical bottlenecks for supply chain stability. Patent CN112679508A introduces a transformative preparation method for key Tofacitinib intermediates, specifically addressing the persistent challenge of enantiomeric impurity control. This technology shifts the paradigm from relying solely on ultra-high-purity chiral starting materials to an active process control strategy that purifies impurities during the reaction sequence itself. By integrating optimized alkaline hydrolysis with a novel crystallization protocol and precision hydrogenation, this method ensures the production of high-purity intermediates suitable for rigorous regulatory standards.
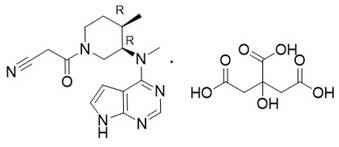
The significance of this innovation lies in its ability to mitigate the enrichment of difficult-to-remove impurities early in the synthesis. Conventional routes often struggle with the carryover of enantiomers that possess physicochemical properties nearly identical to the target molecule, necessitating costly and yield-reducing purification steps at the final API stage. The disclosed method effectively breaks this cycle by targeting impurity reduction at the Compound I to Compound II transformation stage. This proactive approach not only enhances the overall yield but also simplifies the downstream processing requirements, offering a robust solution for manufacturers aiming to optimize their production costs while maintaining stringent quality specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Tofacitinib intermediates frequently encounter significant hurdles regarding stereochemical purity. In standard processes, if the starting chiral materials contain even trace amounts of enantiomeric impurities, these contaminants tend to persist through subsequent reaction steps due to their similar reactivity profiles. The industry has historically relied on sourcing extremely high-optical-purity raw materials to circumvent this issue, which drastically inflates raw material costs and limits supplier options. Furthermore, attempting to remove these enantiomers at the final product stage is often technically formidable; because the impurities share such close physicochemical characteristics with the target API, separation typically requires multiple recrystallizations or preparative chromatography, leading to substantial product loss and extended production cycles.
The Novel Approach
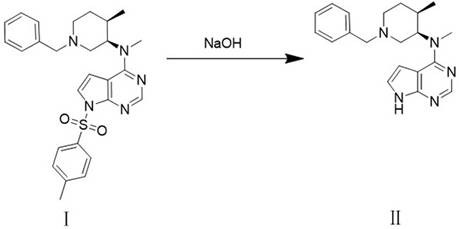
The methodology outlined in patent CN112679508A offers a decisive break from these constraints by introducing a purification-in-process strategy. Instead of passively accepting impurities from the start, the new route actively suppresses their formation and facilitates their removal during the conversion of Compound I to Compound II. This is achieved through a carefully calibrated reaction environment using sodium hydroxide in a tetrahydrofuran and water system, followed by a specific crystallization step using a methanol and water mixed solvent. This dual-action mechanism ensures that the enantiomeric impurity content in Compound II is reduced by a factor of five to ten compared to the starting material, effectively "cleaning" the stream before it enters the critical hydrogenation phase.
Mechanistic Insights into Chiral Impurity Control and Hydrogenation
The core of this technological advancement lies in the nuanced manipulation of solubility and reaction kinetics during the hydrolysis and crystallization phases. When Compound I is treated with aqueous sodium hydroxide under controlled thermal conditions (30-55°C), the reaction proceeds to form Compound II. However, the true innovation is the subsequent crystallization where the crude product is dissolved in methanol and precipitated by the addition of water at low temperatures (0-10°C). This specific solvent ratio (methanol to water ranging from 1:6 to 1:10) creates a thermodynamic environment where the desired enantiomer crystallizes preferentially, leaving the unwanted enantiomeric impurity (Compound II-1) in the mother liquor. This physical separation is far more efficient than chemical derivatization and sets a high purity baseline for the subsequent steps.
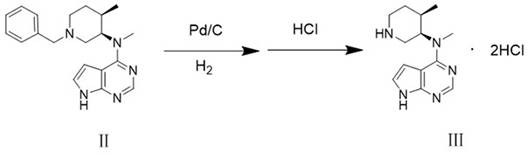
Following the purification of Compound II, the process moves to a catalytic hydrogenation step to generate the final intermediate, Compound III. This step is equally critical, as improper hydrogenation conditions can lead to the formation of specific byproducts like Compound III-1 and Compound III-2. The patent specifies a narrow operating window for hydrogen pressure (0.7-0.8 MPa) and temperature (50-55°C) using palladium on carbon catalysts. Maintaining these parameters is essential to ensure complete deprotection without inducing side reactions that could compromise the integrity of the piperidine ring or the pyrrolo-pyrimidine core. The synergy between the high-purity input from the previous step and the optimized hydrogenation conditions results in a final product with exceptional purity profiles.
How to Synthesize Tofacitinib Intermediate Efficiently
The synthesis of the target Tofacitinib intermediate involves a streamlined sequence that prioritizes impurity management at every juncture. The process begins with the alkaline treatment of the protected precursor, followed immediately by a targeted crystallization to lock in optical purity. Once the intermediate is secured, a controlled hydrogenolysis removes the protecting groups to reveal the active amine functionality required for the final coupling. This route is designed to be operationally simple, avoiding exotic reagents while delivering pharmaceutical-grade quality. For detailed operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- Perform alkaline hydrolysis of Compound I using sodium hydroxide in a THF/water system at controlled temperatures (30-55°C) to generate crude Compound II.
- Purify crude Compound II via crystallization in a methanol/water mixed solvent (1: 6-10 ratio) at 0-10°C to significantly reduce enantiomeric impurities.
- Conduct catalytic hydrogenation of purified Compound II using Pd/C under 0.7-0.8 MPa hydrogen pressure at 50-55°C, followed by salt formation with HCl to yield Compound III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic benefits beyond mere technical specifications. The primary advantage is the decoupling of final product quality from the extreme cost premiums associated with ultra-high-purity chiral starting materials. By demonstrating the capability to upgrade lower-purity inputs through in-process crystallization, manufacturers can diversify their supplier base and negotiate better pricing on raw materials without compromising the quality of the final intermediate. This flexibility acts as a buffer against market volatility and supply disruptions, ensuring a more resilient production schedule.
- Cost Reduction in Manufacturing: The elimination of complex, multi-stage purification procedures at the end of the synthesis line leads to significant operational savings. By shifting the burden of purity control to an earlier, simpler crystallization step, the process reduces solvent consumption, energy usage, and labor hours associated with repetitive recrystallizations or chromatography. Furthermore, the higher overall yield resulting from minimized product loss during purification directly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in starting material quality means that supply chains are less fragile. Manufacturers are not held hostage by a single supplier capable of providing 99.9% ee starting materials; instead, they can source from a broader range of vendors who meet a slightly relaxed specification, knowing the process will correct the deficiency. This redundancy is crucial for maintaining continuous production lines and meeting tight delivery deadlines for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the use of common solvents like methanol, water, and THF simplifies waste management and solvent recovery systems. The process avoids the generation of heavy metal waste streams often associated with alternative chiral resolution techniques, aligning with modern green chemistry principles. The straightforward nature of the unit operations—mixing, heating, cooling, and filtering—ensures that the technology transfers seamlessly from pilot plant to multi-ton commercial reactors with minimal risk of failure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific Tofacitinib intermediate synthesis pathway. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what potential partners can expect regarding quality and performance.
Q: How does this method control enantiomeric impurities compared to conventional routes?
A: Unlike traditional methods that rely heavily on expensive high-optical-purity starting materials, this process actively reduces enantiomeric impurities during the conversion of Compound I to Compound II through optimized reaction conditions and a specific methanol/water crystallization step, achieving a reduction of impurity content by over 5 to 10 times.
Q: What are the critical parameters for the hydrogenation step to ensure low impurity levels?
A: The hydrogenation of Compound II to Compound III requires precise control of hydrogen pressure (0.7-0.8 MPa) and temperature (50-55°C). Additionally, ensuring the residual content of Compound I in the starting material is below 0.2% is crucial to preventing the formation of specific byproduct impurities like Compound III-2.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common solvents like THF, methanol, and water, avoids complex chromatographic separations by relying on efficient crystallization, and employs standard catalytic hydrogenation equipment, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tofacitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of JAK inhibitor synthesis demands a partner with both technical depth and manufacturing agility. Our team has extensively analyzed the pathways described in CN112679508A and possesses the expertise to implement these advanced impurity control strategies effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of Tofacitinib intermediate meets the exacting standards required for global regulatory filings.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific supply chain needs. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your project. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to quality and innovation can drive value for your organization.
