Revolutionizing Alkyl Amino Acryl Amides Manufacturing via Continuous Aminolysis Technology
Revolutionizing Alkyl Amino Acryl Amides Manufacturing via Continuous Aminolysis Technology
The landscape of fine chemical manufacturing is undergoing a significant transformation, driven by the urgent need for more efficient, scalable, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN101027276B, which discloses a robust method for the continuous production of alkyl amino acryl amides. This technology addresses longstanding inefficiencies in the synthesis of these critical intermediates, which are widely utilized in the formulation of pharmaceuticals, agrochemicals, and functional polymers. By shifting from traditional discontinuous batch processes to a sophisticated continuous operation, manufacturers can achieve unprecedented space-time yields and product consistency. The core innovation lies in the seamless integration of reaction and separation units, specifically designed to manage the equilibrium of the aminolysis reaction while protecting sensitive catalytic systems from deactivation. For global procurement and R&D teams, understanding this technological leap is essential for securing a reliable supply of high-purity intermediates in an increasingly competitive market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted acrylamides has relied heavily on batch-wise transesterification or aminolysis processes, which are fraught with operational and economic disadvantages. Conventional literature describes numerous discontinuous methods that often require the use of stoichiometric amounts of catalysts or harsh reaction conditions that degrade product quality. A significant drawback of these legacy batch processes is the inability to efficiently remove the alcohol by-product generated during the reaction, which limits conversion rates and necessitates complex downstream purification steps. Furthermore, batch operations inherently involve sequential steps such as charging, heating, reacting, separating, and discharging, which create bottlenecks in production capacity and lead to significant variability between batches. The thermal stress placed on catalysts, particularly titanium-based compounds, during the prolonged heating and cooling cycles of batch reactors often results in catalyst decomposition and the formation of unwanted solid precipitates. These solids not only reduce catalytic activity but also foul reactor internals and distillation columns, leading to increased maintenance downtime and higher operational expenditures.
The Novel Approach
In stark contrast, the novel continuous process described in the patent data offers a paradigm shift by enabling the continuous feeding of reactants and the continuous withdrawal of products, thereby eliminating the inefficiencies of batch cycling. This method utilizes a cascade of continuously operating stirred tank reactors, allowing for precise control over residence time and temperature profiles, which are critical for maximizing yield and minimizing side reactions. A key feature of this approach is the strategic removal of the liberated alcohol by-product directly from the reaction mixture, often as an azeotrope, which drives the equilibrium towards the desired amide product according to Le Chatelier's principle. By integrating the reaction zone with immediate separation technologies, such as distillation columns and falling film evaporators, the process achieves a level of automation and reproducibility that is unattainable with batch methods. This continuous flow architecture not only enhances the overall throughput of the plant but also ensures that the product quality remains consistent over extended production runs, providing a distinct competitive advantage for suppliers aiming to serve large-scale industrial clients.
Mechanistic Insights into Continuous Aminolysis Catalysis
The chemical foundation of this process is the nucleophilic attack of a primary or secondary amine on the carbonyl carbon of an alkyl acrylate ester, resulting in the formation of an amide bond and the release of an alcohol molecule. This aminolysis reaction is reversible, and its efficiency is heavily dependent on the choice of catalyst and the management of reaction by-products. The patent specifies the use of advanced catalyst systems, preferably a mixture of dialkyltin oxides and alkyl titanates, which act as Lewis acids to activate the carbonyl group for nucleophilic attack. The synergy between tin and titanium species enhances the reaction rate significantly compared to single-component catalysts, allowing the reaction to proceed at moderate temperatures ranging from 110°C to 160°C. Crucially, the mechanism involves the formation of a transient coordination complex between the catalyst and the ester substrate, which lowers the activation energy barrier for the substitution reaction. However, these metal-alkoxide catalysts are highly susceptible to hydrolysis; even trace amounts of water can irreversibly convert the active catalytic species into inactive metal oxides or hydroxides, leading to process failure.
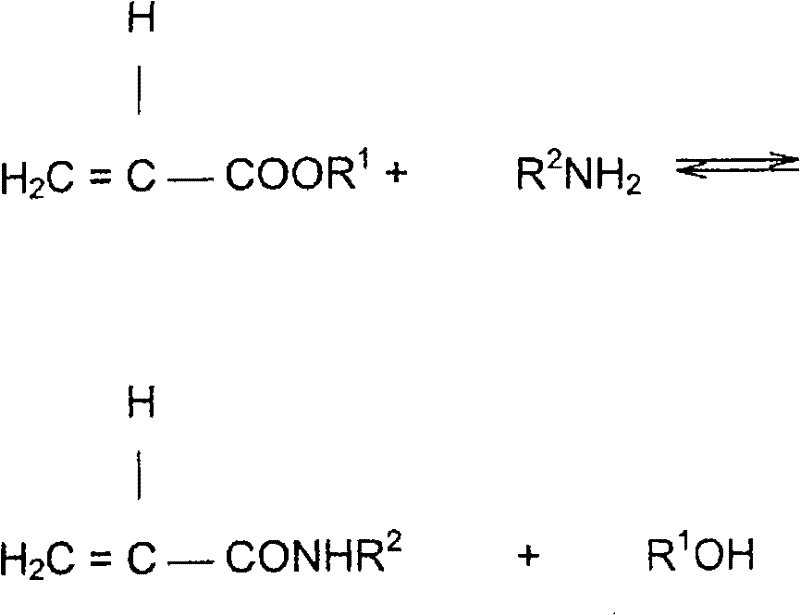
To mitigate this risk, the process incorporates a rigorous dehydration protocol where the amine feed is distilled prior to entering the reactor cascade. This step ensures that water content is reduced to parts-per-million levels, preserving the integrity of the catalyst throughout the continuous operation. Furthermore, the inclusion of polymerization inhibitors, such as phenothiazine or hydroquinone derivatives, is mechanistically essential to prevent the radical polymerization of the acrylic double bond, which is a common side reaction under thermal stress. The careful balance of catalyst concentration, typically between 0.2% and 7% by weight, alongside the precise control of the molar ratio of acrylate to amine, ensures that the reaction proceeds with high selectivity towards the mono-amide product, minimizing the formation of crosslinked oligomers or Michael addition by-products. This deep understanding of the reaction kinetics and thermodynamics allows for the optimization of the process parameters to achieve yields that were previously unattainable in industrial settings.
How to Synthesize N-Dimethylaminopropylacrylamide Efficiently
The practical implementation of this continuous synthesis route requires a meticulously engineered setup that harmonizes fluid dynamics with chemical kinetics. Operators must establish a steady state where the inflow of fresh reactants matches the outflow of products and by-products, maintaining constant levels within the reactor cascade. The process begins with the preparation of the catalyst solution, which is metered continuously into the first reactor of the series alongside the alkyl acrylate feed. Simultaneously, the amine stream undergoes pre-treatment in a dedicated dehydration column to strip moisture, ensuring the longevity of the catalytic system. As the reaction mixture progresses through the串联 (series) of reactors, the temperature is gradually increased to drive the conversion to completion while the evolved alcohol is continuously stripped off via the overhead vapor line connected to a distillation column.
- Continuously feed alkyl acrylate and a specific catalyst mixture (e.g., dioctyltin oxide and isopropyl titanate) into a cascade of stirred tank reactors while maintaining temperatures between 110-160°C.
- Simultaneously introduce the amine reactant through a dedicated dehydration distillation column to remove moisture and prevent catalyst hydrolysis before it enters the reaction zone.
- Separate the crude product mixture using a falling film evaporator and subsequent vacuum distillation to isolate the high-purity amide while recycling unreacted low-boiling components.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this continuous manufacturing technology represents a strategic opportunity to optimize costs and secure supply reliability. The elimination of batch-to-batch variability reduces the need for extensive quality control testing and rework, leading to substantial cost savings in laboratory operations and waste management. By removing the need for frequent reactor cleaning and catalyst replacement associated with batch processes, the continuous method significantly lowers maintenance expenditures and extends the campaign life of production assets. The ability to automate the entire production line reduces the dependency on manual labor, thereby mitigating risks associated with human error and enhancing operational safety in handling reactive chemical intermediates. Moreover, the improved space-time yield means that existing infrastructure can produce significantly higher volumes of material without the need for capital-intensive expansion, offering a scalable solution that can adapt to fluctuating market demands. This flexibility is crucial for maintaining supply continuity in the face of global disruptions, ensuring that downstream customers receive their orders on time and within specification.
- Cost Reduction in Manufacturing: The continuous nature of the process eliminates the downtime associated with charging and discharging batch reactors, leading to a drastic increase in asset utilization and overall productivity. By optimizing the catalyst system to prevent deactivation, the consumption of expensive metal catalysts is minimized, directly reducing the raw material cost per kilogram of product. The integrated separation steps allow for the efficient recycling of unreacted starting materials, further enhancing the atom economy of the process and reducing waste disposal costs. Additionally, the energy efficiency of continuous distillation and evaporation units is superior to batch heating and cooling cycles, resulting in lower utility bills and a reduced carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: Continuous manufacturing provides a steady, predictable output stream that simplifies inventory management and logistics planning for both the producer and the buyer. The robustness of the process against feedstock variations ensures that product specifications remain tight, reducing the risk of supply rejection and the need for safety stock buffers. The modular design of the reactor cascade allows for easy scale-up or capacity adjustment, enabling suppliers to respond rapidly to surges in demand from the pharmaceutical or agrochemical sectors. This reliability fosters stronger long-term partnerships between chemical suppliers and their clients, as the risk of production stoppages due to equipment fouling or catalyst failure is significantly mitigated.
- Scalability and Environmental Compliance: The closed-loop nature of the continuous system minimizes the release of volatile organic compounds (VOCs) and hazardous vapors, ensuring strict compliance with environmental regulations and safety standards. The efficient recovery and recycling of solvents and reactants reduce the volume of chemical waste generated, aligning with green chemistry principles and sustainability goals. The process is inherently scalable, meaning that technology transfer from pilot plant to full commercial production is straightforward and low-risk, facilitating rapid market entry for new derivatives. This scalability ensures that the supply chain can grow in tandem with the customer's business, supporting the commercialization of new drugs or crop protection agents without supply bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous production of alkyl amino acryl amides, based on the detailed disclosures within the patent literature. Understanding these aspects is vital for technical teams evaluating the feasibility of adopting this technology for their specific supply chain needs. The answers provided reflect the consensus on best practices for managing catalyst stability, product purity, and process safety in an industrial setting.
Q: Why is amine dehydration critical in this continuous aminolysis process?
A: Water present in the amine feed can cause irreversible hydrolysis of the titanium-based catalysts, leading to solid precipitates that foul equipment and deactivate the catalytic system. The patented process integrates a dedicated distillation step to reduce water content to ppm levels, ensuring stable long-term operation.
Q: How does this continuous method improve upon traditional batch transesterification?
A: Traditional batch methods suffer from lower space-time yields and inconsistent product quality due to sequential charging and discharging steps. This continuous approach utilizes a cascade of reactors with integrated alcohol removal, significantly increasing throughput, automating operations, and reducing personnel requirements while achieving higher conversion rates.
Q: What catalyst system is preferred for minimizing side reactions?
A: The patent highlights a synergistic mixture of dialkyltin oxides (such as dioctyltin oxide) and alkyl titanates (like isopropyl titanate). This combination effectively drives the aminolysis equilibrium forward while minimizing the formation of crosslinkers and addition products on the double bond, resulting in superior product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Amino Acryl Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced manufacturing technologies to meet the evolving needs of the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of continuous aminolysis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of alkyl amino acryl amides meets the highest international standards for impurity profiles and physical properties. Our commitment to technical excellence allows us to offer customized solutions that align with your specific process requirements, whether you are developing a new API intermediate or optimizing an existing agrochemical formulation.
We invite you to engage with our technical procurement team to discuss how our continuous manufacturing capabilities can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to our high-efficiency supply model. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to build a more resilient and efficient supply chain for your critical chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
