Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Drug Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups to enhance metabolic stability and lipophilicity. Patent CN113045503B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, utilizing a transition metal palladium-catalyzed carbonylation cascade reaction. This technology represents a significant leap forward in synthetic efficiency, moving away from traditional, cumbersome protocols towards a more streamlined and economically viable process. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride and various amines, this invention addresses critical pain points in the supply chain for high-value pharmaceutical intermediates. The method not only demonstrates exceptional substrate compatibility but also achieves high reaction efficiency, making it an ideal candidate for the commercial scale-up of complex drug molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinone compounds has been plagued by significant technical and economic hurdles. Conventional literature reports typically rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. These traditional pathways often suffer from harsh reaction requirements, necessitating extreme temperatures or pressures that complicate reactor design and increase energy consumption. Furthermore, many established methods depend on unstable intermediates like trifluoroacetamides or require expensive pre-activation steps using reagents such as T3P (propylphosphonic anhydride). These factors collectively result in lower overall yields, narrow substrate scopes, and increased production costs, rendering them less attractive for large-scale industrial applications where cost reduction in pharmaceutical intermediate manufacturing is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a sophisticated palladium-catalyzed carbonylation cascade that fundamentally simplifies the synthetic architecture. By utilizing trifluoroethylimidoyl chloride and amines as the foundational building blocks, the method bypasses the need for unstable or costly precursors. The reaction proceeds efficiently in an organic solvent like 1,4-dioxane at a moderate temperature of 110°C. A key innovation is the use of TFBen as a solid carbon monoxide surrogate, which releases CO in situ upon heating. This eliminates the safety risks and engineering complexities associated with handling gaseous carbon monoxide. The result is a highly versatile protocol capable of accommodating a wide range of functional groups, thereby enabling the rapid synthesis of diverse quinazolinone derivatives tailored for specific drug discovery programs.
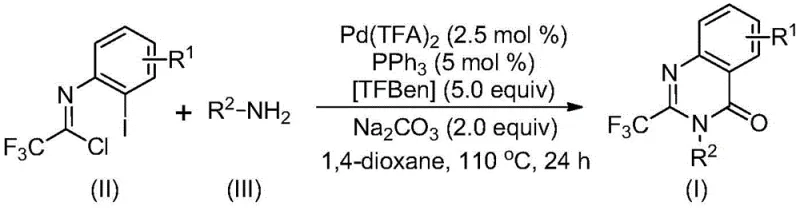
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation offers profound insights into its high efficiency and selectivity. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the trifluoroethylimidoyl chloride and the amine, generating a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, specifically palladium trifluoroacetate coordinated with triphenylphosphine, inserts into the carbon-iodine bond of the aromatic ring to form a divalent palladium species. As the temperature rises to 110°C, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to create an acyl-palladium intermediate. This step is crucial for introducing the carbonyl functionality required for the quinazolinone core.
Following the CO insertion, the presence of a base facilitates the formation of a palladium-nitrogen bond, leading to the construction of a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This intricate catalytic cycle ensures high atom economy and minimizes the formation of side products. From an impurity control perspective, the mild conditions and the specific ligand environment provided by triphenylphosphine help suppress unwanted side reactions, such as homocoupling or hydrolysis, ensuring that the resulting high-purity pharmaceutical intermediates meet stringent quality specifications required for downstream drug synthesis.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this synthesis makes it highly accessible for laboratory and pilot plant settings. The process involves charging a reaction vessel with the specified molar ratios of palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, the imidoyl chloride substrate, and the amine nucleophile in 1,4-dioxane. The mixture is then heated to 110°C for a duration of 16 to 30 hours. Post-reaction workup is straightforward, involving filtration to remove inorganic salts, followed by silica gel treatment and column chromatography purification. For a comprehensive, step-by-step guide including exact stoichiometric calculations and safety protocols, please refer to the standardized synthesis instructions below.
- Combine palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits regarding cost structure and supply reliability. The shift from expensive, specialized reagents to commodity chemicals like trifluoroethylimidoyl chlorides and simple amines drastically reduces the raw material cost base. Moreover, the use of a solid CO surrogate enhances process safety, potentially lowering insurance and facility compliance costs associated with high-pressure gas handling. The broad substrate scope means that a single manufacturing platform can produce a wide variety of derivatives, reducing the need for multiple dedicated production lines and increasing overall asset utilization.
- Cost Reduction in Manufacturing: The elimination of expensive coupling reagents like T3P and the use of catalytic amounts of palladium rather than stoichiometric quantities significantly lowers the direct material costs. Additionally, the simplified post-treatment process, which avoids complex extraction or distillation steps often required by older methods, reduces utility consumption and labor hours. This cumulative effect leads to substantial cost savings in the production of these valuable heterocyclic scaffolds, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including various substituted anilines and trifluoroacetic acid derivatives, are widely available from multiple global suppliers. This diversity in the supply base mitigates the risk of shortages that often plague processes dependent on single-source or highly specialized reagents. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results, thereby securing a steady flow of materials for downstream API synthesis.
- Scalability and Environmental Compliance: The protocol has been successfully demonstrated at the gram scale and is inherently designed for scale-up due to the use of standard organic solvents and manageable temperatures. The avoidance of hazardous gaseous carbon monoxide simplifies the engineering controls required for larger reactors. From an environmental standpoint, the high atom efficiency and reduced waste generation align with green chemistry principles, facilitating easier regulatory approval and waste disposal management, which is increasingly critical for sustainable chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this method for producing high-value chemical intermediates.
Q: What are the primary advantages of this Pd-catalyzed method over traditional quinazolinone synthesis?
A: Unlike conventional methods that require harsh conditions, expensive pre-activated substrates like trifluoroacetic anhydride, or unstable reagents, this novel approach utilizes cheap and readily available trifluoroethylimidoyl chlorides. It operates under milder conditions with a solid CO surrogate, significantly improving reaction efficiency and substrate compatibility.
Q: Can this synthetic route be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield synthesis of Rutaecarpine. The process achieved a total yield of 77% over three steps, proving its viability for constructing complex fused-ring nitrogen-containing heterocycles found in bioactive natural products.
Q: What is the role of TFBen in this reaction system?
A: TFBen acts as a solid carbon monoxide surrogate. Under heating conditions at 110°C, it releases carbon monoxide in situ, which then inserts into the carbon-palladium bond. This eliminates the need for handling hazardous high-pressure CO gas, enhancing operational safety and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation technology and is well-equipped to integrate it into our existing manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest international standards for pharmaceutical use.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific supply chain requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your project's volume and purity needs. We encourage you to request specific COA data and route feasibility assessments to verify the suitability of this advanced methodology for your next-generation drug candidates, ensuring a reliable and cost-effective partnership for the long term.
