Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
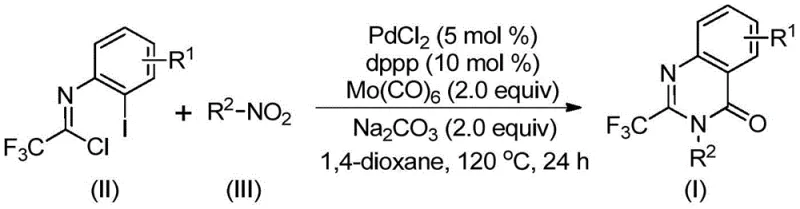
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity. Patent CN112480015B introduces a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a core structure found in numerous bioactive molecules ranging from antifungals to anticancer agents. This technology leverages a palladium-catalyzed carbonylation cascade that transforms inexpensive nitro compounds and trifluoroethylimidoyl chlorides into high-value intermediates without the need for hazardous high-pressure carbon monoxide gas. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, more cost-effective manufacturing of complex nitrogen-containing heterocycles.
The significance of this invention lies in its ability to streamline the supply chain for critical drug intermediates. By utilizing a solid carbon monoxide substitute, specifically molybdenum hexacarbonyl, the process circumvents the logistical and safety challenges associated with gaseous CO handling. Furthermore, the reaction exhibits exceptional substrate compatibility, tolerating various functional groups such as halogens, alkyls, and trifluoromethyl groups on both the aromatic ring and the nitrogen substituent. This versatility ensures that a reliable pharmaceutical intermediate supplier can offer a diverse library of derivatives to support early-stage drug discovery and late-stage process optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the quinazolinone core often suffer from severe operational constraints that hinder commercial viability. Historically, methods have relied on ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides, which necessitate high-pressure carbon monoxide conditions that pose significant safety risks and require specialized reactor infrastructure. Other approaches involve iron-catalyzed condensations or palladium-catalyzed cyclizations using expensive pre-activated substrates like 2-bromoformylaniline or acid anhydrides. These conventional routes are frequently plagued by low yields, narrow substrate scope, and the need for harsh reaction conditions that limit functional group tolerance. Consequently, scaling these processes for industrial application often results in prohibitive costs and complex purification challenges due to the formation of difficult-to-remove impurities.
The Novel Approach
In stark contrast, the methodology disclosed in CN112480015B offers a streamlined, one-pot solution that dramatically simplifies the synthetic workflow. By employing trifluoroethylimidoyl chloride and nitro compounds as起始 materials, the process utilizes a palladium catalyst system with a phosphine ligand to drive a tandem reduction-carbonylation-cyclization sequence. The use of Mo(CO)6 as an in situ source of carbon monoxide allows the reaction to proceed at atmospheric pressure equivalents within a sealed vessel at 120°C, eliminating the need for high-pressure gas lines. This novel approach not only enhances safety but also improves atom economy and reaction efficiency, enabling the synthesis of diverse 2-trifluoromethyl quinazolinones with yields often exceeding 90%.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogs. The reaction likely initiates with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which acts as both a reducing agent and a CO source. Subsequently, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the generated amine and the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine derivative intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a divalent palladium species. As the temperature rises to 120°C, Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to form an acyl-palladium intermediate.
Following CO insertion, the base facilitates the formation of a palladium-nitrogen bond, leading to a seven-membered cyclic palladium intermediate. The final step involves reductive elimination, which releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This intricate cascade highlights the importance of ligand selection, specifically 1,3-bis(diphenylphosphino)propane (dppp), in stabilizing the palladium center and promoting the necessary oxidative addition and reductive elimination steps. The mechanism ensures high purity by minimizing side reactions, as the sequential nature of the transformations directs the pathway firmly toward the cyclized product.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it ideal for both laboratory scale-up and pilot plant production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an aprotic solvent such as 1,4-dioxane. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal profiles required to achieve optimal conversion. Adhering to these parameters ensures consistent quality and maximizes the yield of the target pharmaceutical intermediate.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from high-pressure gas systems to solid reagents fundamentally alters the risk profile and capital expenditure requirements for manufacturing facilities. By removing the dependency on specialized high-pressure infrastructure, companies can utilize standard glass-lined or stainless steel reactors, thereby significantly reducing capital investment and maintenance costs. Furthermore, the use of commercially available nitro compounds and simple imidoyl chlorides ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical markets.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated substrates and high-pressure equipment leads to substantial cost savings in API manufacturing. The use of cheap nitro compounds as starting materials drastically lowers the raw material cost per kilogram compared to traditional bromo- or iodo-aniline precursors. Additionally, the simplified post-treatment process, which involves basic filtration and column chromatography, reduces solvent consumption and labor hours associated with complex workups. This economic efficiency allows for more competitive pricing strategies when supplying high-purity pharmaceutical intermediates to global clients.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved due to the widespread availability of the key reagents. Nitro compounds and trifluoroethylimidoyl chlorides are commodity chemicals produced by multiple vendors worldwide, ensuring that supply chain disruptions are minimized. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that minor variations in raw material quality do not compromise the final product integrity. This resilience is critical for maintaining continuous production schedules and meeting tight delivery deadlines for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated from milligram to gram scales with consistent performance. The absence of toxic carbon monoxide gas emissions aligns with increasingly stringent environmental regulations, simplifying the permitting process for new production lines. Moreover, the high atom economy and reduced waste generation contribute to a greener manufacturing footprint, supporting corporate sustainability goals. The ability to scale up complex pharmaceutical intermediates safely and efficiently positions this technology as a preferred choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this technology for their pipeline.
Q: What are the key advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that replace costly pre-activated substrates, significantly lowering raw material costs while maintaining high reaction efficiency.
Q: How does this method improve safety compared to traditional carbonylation?
A: By utilizing solid Mo(CO)6 as a carbon monoxide substitute instead of high-pressure CO gas, the process eliminates the need for specialized high-pressure equipment, enhancing operational safety.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-treatment procedures, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain for quinazolinone-based therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of patent CN112480015B can deliver superior value, reduced lead time for high-purity pharmaceutical intermediates, and unmatched supply security for your organization.
