Advanced Sterically Hindered Chiral Quaternary Ammonium Salts for High-Enantioselectivity Pharmaceutical Intermediates
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric phase transfer catalysis has relied heavily on cinchona alkaloid derivatives, evolving through distinct generations of structural modification. The first generation, pioneered by Dolling, utilized simple N-alkylation of the quinuclidine nitrogen, achieving respectable but limited enantioselectivity. Subsequent advancements led to the second generation, reported by O'Donnell, which introduced O-alkylation alongside N-alkylation, further refining the chiral environment. However, the third generation, characterized by the introduction of anthracene or large aromatic groups as seen in Lygo and Corey catalysts, still faces significant challenges when applied to harsh oxidative conditions. Specifically, when utilized in potassium permanganate oxidations, these conventional third-generation catalysts often exhibit poor stability, leading to decomposition and consequently moderate enantioselectivity and low yields. The inherent instability of the quaternary ammonium salt structure under strong oxidative conditions has long been a bottleneck, necessitating the use of stoichiometric amounts of catalyst to drive reactions, which is economically and environmentally unsustainable for large-scale pharmaceutical intermediate manufacturing.
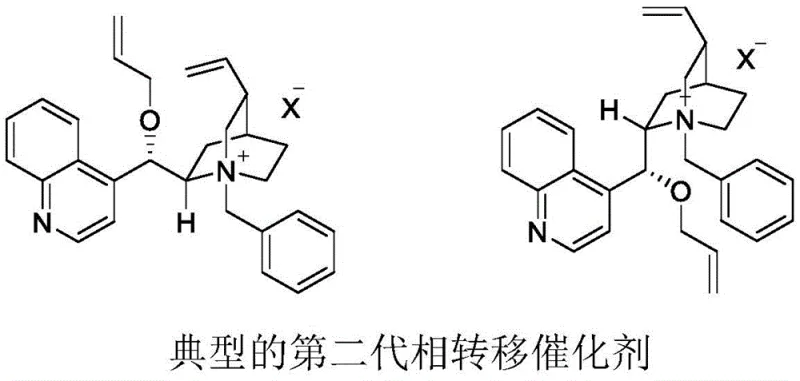
The Novel Approach
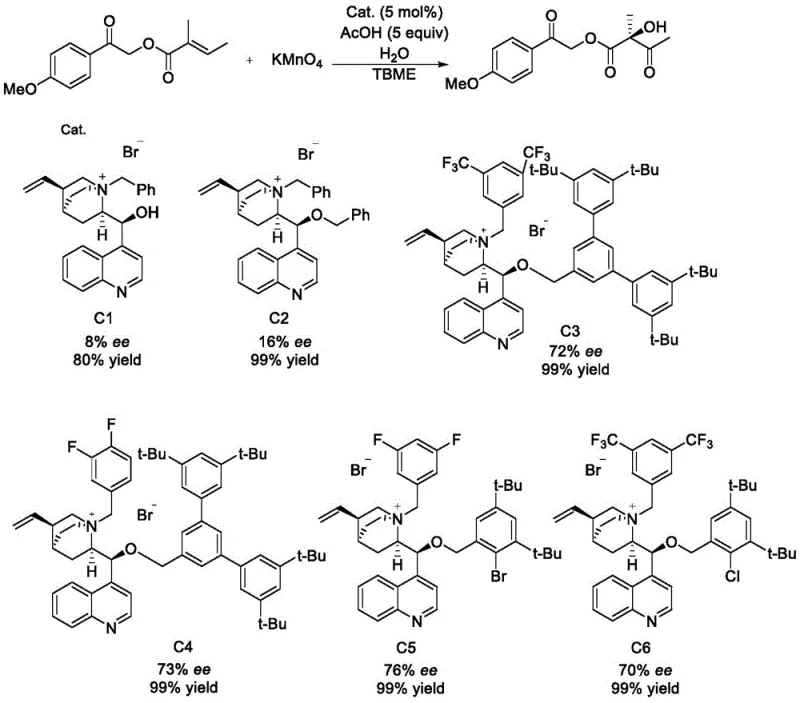
The present invention, detailed in patent CN113549062A, introduces a paradigm shift by engineering a new class of cinchona-derived quaternary ammonium salts featuring exceptionally large steric hindrance. Unlike previous iterations that relied on standard benzyl or anthracene methyl groups, this novel approach incorporates bulky aryl substituents, such as 3,5-di-tert-butylphenyl groups, at the oxygen position of the cinchona backbone. This strategic structural modification serves a dual purpose: it dramatically increases the steric bulk around the catalytic center, thereby enforcing a more rigid and defined chiral pocket for substrate binding, and it significantly enhances the chemical stability of the catalyst against oxidative degradation. By moving away from the fragile architectures of the past, this new methodology enables the use of catalytic rather than stoichiometric amounts of the phase transfer agent, representing a substantial leap forward in cost reduction in API manufacturing and process efficiency.
Mechanistic Insights into Sterically Hindered Phase Transfer Catalysis
The efficacy of the new catalyst lies in the precise manipulation of non-covalent interactions within the phase transfer interface. In the asymmetric oxidation of olefins by potassium permanganate, the reaction occurs at the interface of the organic and aqueous phases. The quaternary ammonium cation acts as a shuttle, transporting the permanganate anion into the organic phase where the olefin substrate resides. The introduction of massive steric groups, such as the bis(3,5-di-tert-butylphenyl)methyl moiety, creates a highly congested environment around the nitrogen center. This congestion forces the substrate to approach the oxidant from a specific trajectory, effectively blocking unfavorable reaction pathways that lead to racemic byproducts. Furthermore, the electron-donating nature of the tert-butyl groups may subtly influence the electron density of the aromatic rings, potentially stabilizing the transition state through pi-stacking or van der Waals interactions with the substrate. This level of stereocontrol is critical for producing high-purity pharmaceutical intermediates where even minor impurities can compromise drug safety.
Moreover, the stability mechanism is equally vital for practical application. Conventional catalysts often degrade via nucleophilic attack or oxidative cleavage of the benzyl groups attached to the nitrogen or oxygen. The bulky substituents in the new catalysts provide a physical shield, protecting the reactive centers from aggressive oxidants like KMnO4. This robustness ensures that the catalyst maintains its integrity throughout the reaction cycle, allowing for consistent turnover numbers. The result is a catalytic system that not only delivers superior enantioselectivity but also operates reliably under the rigorous conditions required for industrial synthesis, addressing the longstanding issue of catalyst decomposition that has plagued earlier generations of phase transfer catalysts in oxidative transformations.
How to Synthesize Bulky Chiral Quaternary Ammonium Salt Efficiently
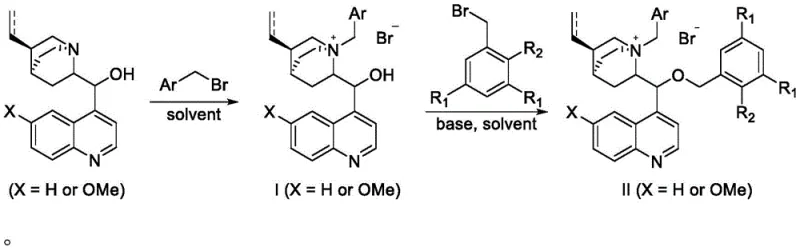
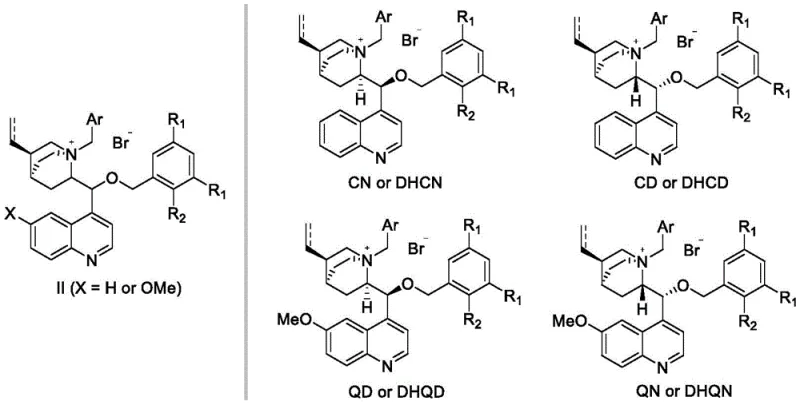
The synthesis of these advanced catalysts is designed for operational simplicity and scalability, avoiding the need for expensive transition metal catalysts or cryogenic conditions typically associated with fine chemical synthesis. The process begins with the readily available cinchona alkaloids, such as cinchonidine or quinine, which serve as the chiral pool starting materials. The first step involves a straightforward N-alkylation with a benzyl bromide derivative in a solvent like toluene, forming the intermediate quaternary salt. The second, and most critical step, involves the O-alkylation with a specifically designed, sterically demanding benzyl bromide. This step utilizes a biphasic system with aqueous base, leveraging the phase transfer properties of the intermediate itself to drive the reaction to completion. The entire process is amenable to commercial scale-up of complex pharmaceutical intermediates, requiring only standard filtration and chromatography for purification, thus minimizing waste and maximizing throughput.
- Dissolve cinchona alkaloid in an organic solvent like toluene and react with benzyl bromide at reflux to form the N-benzyl quaternary ammonium salt intermediate.
- Dissolve the intermediate in dichloromethane and react with a bulky substituted benzyl bromide (e.g., 3,5-di-tert-butylbenzyl bromide) in the presence of aqueous KOH.
- Purify the final crude product using silica gel column chromatography to isolate the high-steric-hindrance chiral catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers compelling strategic advantages beyond mere technical performance. The primary benefit is the drastic simplification of the supply chain for chiral building blocks. By enabling the direct asymmetric oxidation of simple olefins, this method bypasses the need for multi-step resolutions or the procurement of expensive chiral starting materials, which are often subject to volatile market pricing and limited availability. The reliance on potassium permanganate as the oxidant is particularly advantageous; it is a commodity chemical with a stable global supply chain, contrasting sharply with specialized oxidants that may face regulatory or logistical bottlenecks. This shift towards commodity reagents significantly de-risks the sourcing strategy for key intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the shift from stoichiometric to catalytic usage. Traditional methods often required equivalent amounts of expensive chiral auxiliaries or catalysts to achieve acceptable yields, inflating the raw material costs exponentially. In contrast, this new catalyst operates efficiently at low loading (e.g., 5 mol%), drastically reducing the cost per kilogram of the final product. Furthermore, the elimination of transition metals from the catalyst structure removes the need for costly and time-consuming heavy metal scavenging steps during downstream processing. This simplification of the purification workflow translates directly into lower operational expenditures and reduced solvent consumption, aligning with green chemistry principles while boosting the bottom line.
- Enhanced Supply Chain Reliability: The robustness of the catalyst synthesis ensures a reliable supply of the catalytic material itself. Since the precursors are derived from natural cinchona alkaloids and simple aromatic bromides, the risk of supply disruption is minimal compared to catalysts relying on rare earth metals or complex ligands. Additionally, the high stability of the catalyst under reaction conditions means that batch-to-batch variability is significantly reduced. For supply chain heads, this predictability is invaluable, as it allows for tighter production scheduling and reduces the need for safety stock buffers. The ability to consistently produce high-enantiopurity material reduces the risk of batch rejection, ensuring a steady flow of quality intermediates to downstream API synthesis units.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner alternative to traditional oxidation methods. The use of potassium permanganate generates manganese dioxide as a byproduct, which is a solid that can be easily filtered and potentially recycled, unlike liquid waste streams generated by other oxidants. The absence of toxic heavy metals in the catalyst formulation simplifies regulatory compliance and waste disposal protocols. The process is inherently scalable, having been demonstrated to work effectively in standard organic solvents like toluene and dichloromethane, which are well-understood in industrial settings. This ease of scale-up facilitates the reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond rapidly to market demands without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear picture of the operational benefits and performance metrics expected when transitioning to this new catalytic system.
Q: How does the new catalyst improve upon third-generation phase transfer catalysts?
A: Unlike third-generation catalysts which suffer from stability issues under potassium permanganate oxidation, the new bulky catalysts feature large steric groups (like di-tert-butylphenyl) that enhance structural stability and significantly boost enantioselectivity from moderate levels to over 70% ee.
Q: What are the key applications of this catalytic technology?
A: This technology is primarily applied to the asymmetric oxidation of olefins using potassium permanganate to synthesize chiral alpha-hydroxy-beta-keto esters, which are valuable intermediates in pharmaceutical and agrochemical manufacturing.
Q: Is the synthesis of these catalysts scalable for industrial production?
A: Yes, the synthesis involves a straightforward two-step alkylation process using commercially available cinchona alkaloids and benzyl bromides, making it highly suitable for commercial scale-up without requiring complex transition metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Quaternary Ammonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this sterically hindered phase transfer catalysis technology for the synthesis of high-value chiral intermediates. As a leading CDMO partner, we possess the technical expertise to adapt and optimize these patented routes for your specific production needs. Our facilities are equipped to handle the synthesis of complex quaternary ammonium salts with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key in pharmaceutical manufacturing, which is why our stringent purity specifications and rigorous QC labs ensure that every batch of catalyst meets the highest standards of quality and performance, ready for immediate integration into your asymmetric oxidation processes.
We invite you to explore how this advanced catalytic solution can streamline your synthesis of alpha-hydroxy-beta-keto esters and other chiral building blocks. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your current manufacturing workflow, demonstrating the tangible economic benefits of switching to this superior technology. Please contact our technical procurement team today to request specific COA data for our catalyst library and discuss route feasibility assessments for your target molecules, ensuring a seamless transition to more efficient and sustainable production methods.
