Advanced Cinchonidine-Derived Catalysts for High-Purity Pharmaceutical Intermediate Manufacturing
Advanced Cinchonidine-Derived Catalysts for High-Purity Pharmaceutical Intermediate Manufacturing
The landscape of asymmetric synthesis is undergoing a transformative shift driven by the demand for higher purity and greener processes in the production of active pharmaceutical ingredients (APIs). A pivotal development in this field is documented in patent CN113549062A, which introduces a novel class of large sterically hindered chiral quaternary ammonium salt phase transfer catalysts derived from cinchonadine. This technology addresses critical bottlenecks in the asymmetric oxidation of olefins, specifically targeting the synthesis of chiral α-hydroxy-β-ketoesters, which are vital building blocks for numerous therapeutic agents. By leveraging the inherent chirality of natural cinchona alkaloids and enhancing it through strategic structural modification, this innovation offers a robust pathway for achieving superior enantioselectivity. For R&D directors and process chemists, this represents a significant opportunity to optimize synthetic routes, reducing the reliance on expensive chiral auxiliaries or resolution steps that often plague traditional manufacturing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the evolution of cinchona alkaloid-based phase transfer catalysts has been marked by incremental improvements, yet significant challenges remain in achieving high stereocontrol under mild conditions. First-generation catalysts, typically formed by simple N-alkylation of the bridgehead nitrogen, often lack the necessary steric bulk to effectively differentiate between enantiotopic faces of a substrate. As illustrated in early models, these structures provide a relatively open chiral pocket, resulting in modest enantiomeric excess (ee) values that frequently necessitate costly downstream purification. Furthermore, second-generation catalysts, which introduced O-alkylation, improved performance but still struggled with stability under harsh oxidative conditions. When applied to potassium permanganate oxidations, earlier catalysts often decomposed or exhibited poor turnover numbers, requiring stoichiometric amounts rather than catalytic quantities. This inefficiency not only drives up raw material costs but also complicates the removal of catalyst residues from the final product, posing a risk to the stringent impurity profiles required for pharmaceutical intermediates.
The Novel Approach
The breakthrough presented in the patent data lies in the rational design of catalysts featuring massive steric hindrance groups attached to the oxygen atom of the cinchona scaffold. Unlike previous iterations that utilized simple allyl or benzyl groups, this new approach employs bulky aryl substituents, such as 3,5-di-tert-butylphenyl or halogenated phenyl rings. 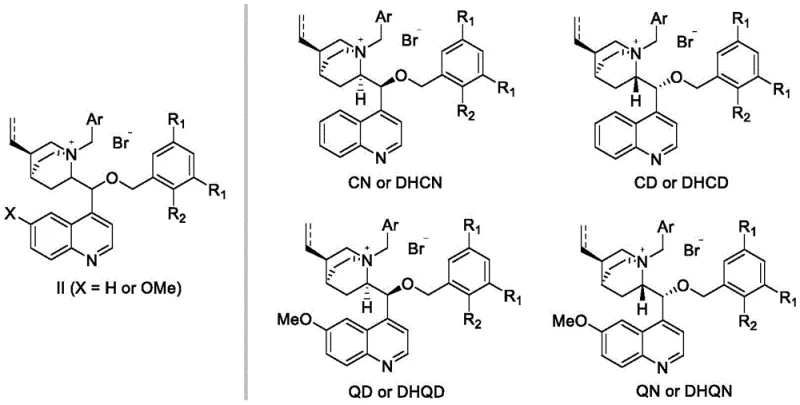 . This structural reinforcement creates a highly defined chiral environment that rigidly locks the substrate orientation during the reaction. The result is a dramatic enhancement in catalytic efficiency and stereoselectivity. By shifting from small, flexible substituents to these voluminous aromatic systems, the catalyst effectively shields one side of the reactive center, forcing the oxidant to approach from a single, specific trajectory. This design philosophy transforms the catalyst from a passive participant into a highly selective molecular machine, capable of delivering products with significantly higher optical purity while maintaining stability under the rigorous conditions of permanganate oxidation.
. This structural reinforcement creates a highly defined chiral environment that rigidly locks the substrate orientation during the reaction. The result is a dramatic enhancement in catalytic efficiency and stereoselectivity. By shifting from small, flexible substituents to these voluminous aromatic systems, the catalyst effectively shields one side of the reactive center, forcing the oxidant to approach from a single, specific trajectory. This design philosophy transforms the catalyst from a passive participant into a highly selective molecular machine, capable of delivering products with significantly higher optical purity while maintaining stability under the rigorous conditions of permanganate oxidation.
Mechanistic Insights into Asymmetric Phase Transfer Oxidation
The mechanistic superiority of these bulky catalysts stems from their ability to form tight ion pairs at the interface of the organic and aqueous phases. In the context of potassium permanganate oxidation, the quaternary ammonium cation acts as a phase transfer agent, shuttling the permanganate anion into the organic layer where the olefin substrate resides. The presence of the large steric groups near the chiral center influences the conformation of the transition state. Specifically, the bulky aryl groups prevent the substrate from adopting low-energy conformations that would lead to the undesired enantiomer. Instead, they force the olefin into a high-energy, yet stereochemically defined orientation that favors the formation of the target chiral α-hydroxy-β-ketoester. 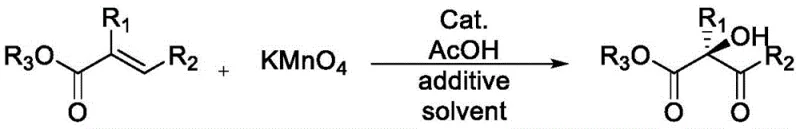 . This precise control over the transition state geometry is the key to the observed leap in enantioselectivity. Moreover, the electron-withdrawing or donating properties of the substituents on the aryl rings can be tuned to further modulate the electrophilicity of the catalyst, allowing for fine-tuning of reaction rates and selectivity for different substrate classes.
. This precise control over the transition state geometry is the key to the observed leap in enantioselectivity. Moreover, the electron-withdrawing or donating properties of the substituents on the aryl rings can be tuned to further modulate the electrophilicity of the catalyst, allowing for fine-tuning of reaction rates and selectivity for different substrate classes.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional methods often suffer from over-oxidation or non-selective background reactions that generate racemic byproducts. The high selectivity of the bulky catalyst minimizes these side reactions by accelerating the desired pathway so effectively that competing non-catalyzed reactions become negligible. This kinetic resolution ensures that the crude reaction mixture contains a much higher proportion of the desired enantiomer, simplifying the purification process. For quality control teams, this means fewer chromatographic steps are required to meet specification limits, directly translating to higher overall yields and reduced solvent consumption. The stability of the catalyst under oxidative stress also means that degradation products of the catalyst itself do not contaminate the product stream, a common issue with less robust phase transfer agents.
How to Synthesize Cinchonidine-Derived Catalysts Efficiently
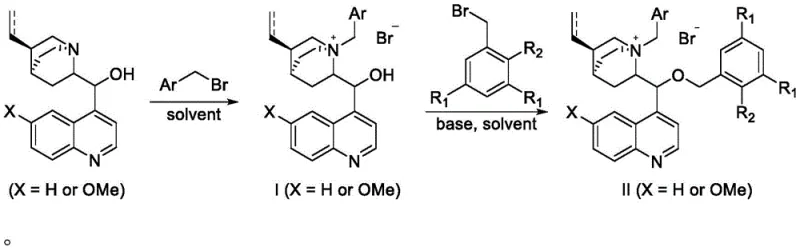
The synthesis of these advanced catalysts is designed for practicality and scalability, utilizing a straightforward two-step sequence that avoids exotic reagents or extreme conditions. The process begins with the quaternization of the naturally abundant cinchona alkaloid, followed by a selective alkylation of the hydroxyl group with a sterically demanding benzyl bromide derivative. This modular approach allows manufacturers to easily swap out the aryl groups to create a library of catalysts tailored for specific substrates. The robustness of the synthetic route ensures that the catalyst can be produced in multi-kilogram quantities without compromising quality, addressing a major concern for supply chain managers looking for reliable sources of specialized reagents.
- Dissolve cinchona alkaloid in an organic solvent such as toluene, add benzyl bromide, and reflux to obtain the N-benzyl cinchona alkaloid quaternary ammonium salt intermediate.
- Dissolve the intermediate in dichloromethane, add a sterically hindered substituted benzyl bromide and an inorganic base aqueous solution like 50% KOH.
- React the mixture at controlled temperatures between -20°C and 60°C, followed by column chromatography separation to isolate the final bulky catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The shift towards this novel catalyst system addresses several pain points associated with traditional asymmetric oxidation methods, particularly regarding safety, waste management, and raw material availability. By replacing hazardous oxidants like lead tetraacetate or molybdenum-based reagents with potassium permanganate, the process aligns with modern green chemistry principles, significantly reducing the regulatory burden and disposal costs associated with toxic heavy metal waste. This transition not only lowers the environmental footprint of the manufacturing site but also mitigates the risk of supply disruptions caused by tightening regulations on hazardous chemicals.
- Cost Reduction in Manufacturing: The implementation of this bulky catalyst system drives cost efficiency through multiple mechanisms. Primarily, the enhanced enantioselectivity reduces the need for expensive chiral resolution steps, such as preparative chiral HPLC or diastereomeric crystallization, which are often the most cost-intensive part of producing chiral intermediates. Furthermore, the catalyst operates effectively at low loadings (e.g., 5 mol%), and its stability allows for potential recycling or extended use in continuous flow setups. The use of potassium permanganate as a stoichiometric oxidant is also economically favorable compared to proprietary oxidizing agents, as it is a commodity chemical with a stable global supply chain and low price point. These factors combine to drastically lower the cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The synthesis of the catalyst relies on cinchona alkaloids, which are extracted from renewable plant sources, and common aromatic building blocks that are widely available from bulk chemical suppliers. This reduces dependency on single-source suppliers for exotic ligands or metals that are prone to geopolitical supply shocks. The two-step synthesis is robust and tolerant of minor variations in reaction conditions, ensuring consistent batch-to-batch quality. For supply chain planners, this reliability translates to predictable lead times and the ability to scale production rapidly in response to market demand without the lengthy validation periods often required for biocatalytic or enzymatic processes.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to large-scale industrial reactors. The use of common solvents like toluene and dichloromethane, along with aqueous base solutions, fits well within existing infrastructure in most fine chemical plants. The byproduct of the oxidation, manganese dioxide, is a solid that can be easily removed by filtration, simplifying the workup procedure and minimizing aqueous waste volume. This ease of separation is crucial for scaling up, as complex extractions or distillations often become bottlenecks at the tonne scale. Additionally, the avoidance of heavy metals simplifies the environmental permitting process, facilitating faster site approvals and reducing long-term liability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a realistic view of the technology's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing portfolio.
Q: How does the steric hindrance of this new catalyst improve enantioselectivity compared to first-generation models?
A: The introduction of large steric groups, such as 3,5-di-tert-butylphenyl moieties, creates a more rigid chiral environment around the active nitrogen center. This effectively blocks one face of the substrate during the phase transfer process, forcing the reaction to proceed through a single stereochemical pathway, thereby significantly boosting enantiomeric excess (ee) values from single digits to over 70%.
Q: Is the potassium permanganate oxidation method scalable for industrial production?
A: Yes, the method utilizes potassium permanganate, a stable and commercially available oxidant, avoiding toxic heavy metals like lead or molybdenum. The byproduct, manganese dioxide, is easily filtered and can be recycled, making the process environmentally compliant and suitable for large-scale manufacturing without complex waste treatment protocols.
Q: What are the storage and stability conditions for these cinchona-derived catalysts?
A: These quaternary ammonium salts exhibit high thermal and chemical stability under standard storage conditions. Unlike earlier generations that suffered from decomposition under strong oxidative conditions, the bulky substituents protect the catalytic core, ensuring a long shelf-life and consistent performance across multiple batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinchonidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug development and optimizing commercial manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of the catalyst itself or the downstream chiral intermediates generated using this methodology, our infrastructure is designed to support your timeline and quality requirements.
We invite you to collaborate with us to evaluate the potential of this bulky phase transfer catalyst technology for your specific projects. Our technical sales team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential reductions in processing time and waste disposal costs for your specific molecule. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us partner with you to drive efficiency and innovation in your supply chain.
