Advanced Spermidine Synthesis: Overcoming Purification Challenges for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for polyamines like Spermidine, a critical molecule involved in cellular proliferation and autophagy regulation. A significant breakthrough in this domain is detailed in Chinese Patent CN116239475A, which discloses a novel preparation method that fundamentally alters the impurity profile and scalability of Spermidine production. Unlike traditional approaches that struggle with separation efficiencies, this patented methodology employs a strategic BOC-protection strategy coupled with catalytic hydrogenation to achieve exceptional purity levels exceeding 99%. For R&D directors and procurement specialists, understanding this shift from Michael addition chemistry to protected amide coupling represents a pivotal opportunity to optimize supply chains for high-value pharmaceutical intermediates. The process not only mitigates the risks associated with toxic acrylonitrile handling but also streamlines the downstream processing, offering a compelling value proposition for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Spermidine has relied heavily on the nucleophilic addition of 1,4-diaminobutane to acrylonitrile, followed by the reduction of the resulting nitrile group. While conceptually straightforward, this legacy pathway is plagued by severe thermodynamic and kinetic limitations that hinder efficient manufacturing. The primary bottleneck lies in the difficulty of removing the large excess of 1,4-diaminobutane required to drive the reaction, as its boiling point and polarity closely mimic the desired product. Furthermore, the reactivity of acrylonitrile often leads to uncontrolled polymerization and the formation of oligomeric byproducts that are chemically similar to Spermidine. These impurities possess physicochemical properties nearly identical to the target molecule, rendering standard purification techniques like simple distillation ineffective and necessitating costly and time-consuming chromatographic separations. Consequently, the overall yield is compromised, and the environmental footprint is expanded due to the generation of hazardous waste streams that are difficult to treat.
The Novel Approach
In stark contrast, the methodology outlined in CN116239475A introduces a sophisticated three-step sequence that elegantly bypasses these purification nightmares through the use of orthogonal protecting groups. The process initiates with the coupling of N-BOC-1,4-butanediamine and cyanoacetic acid, utilizing a condensing agent to form a stable amide intermediate. This strategic installation of the tert-butoxycarbonyl (BOC) group masks the reactive amine, preventing the self-polymerization and side reactions typical of free amines in the presence of electrophiles. The subsequent step involves a catalytic hydrogenation in the presence of BOC anhydride, which simultaneously reduces the nitrile and protects the newly formed amine, ensuring a clean conversion to the poly-BOC substituted precursor. This approach transforms the purification challenge from a complex separation of similar amines into a straightforward isolation of protected intermediates, culminating in a final deprotection and distillation step that yields pharmaceutical-grade Spermidine with remarkable efficiency.
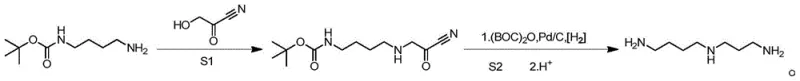
Mechanistic Insights into BOC-Protected Amide Coupling and Hydrogenation
The core of this synthetic innovation lies in the precise control of reactivity during the initial amide bond formation and the subsequent reduction phase. In the first step, the reaction between N-BOC-1,4-butanediamine and cyanoacetic acid is mediated by a carbodiimide coupling agent, preferably DCC (dicyclohexylcarbodiimide), in a solvent system such as dichloromethane. The mechanism proceeds through the activation of the carboxylic acid to form an O-acylisourea intermediate, which is then attacked by the free amine of the protected diamine. The presence of the BOC group is crucial here; it electronically deactivates the protected nitrogen, ensuring that the nucleophilic attack occurs exclusively at the terminal primary amine. This regioselectivity is vital for preventing the formation of bis-alkylated byproducts or cyclic ureas, which are common failure modes in unprotected diamine couplings. The use of DCC facilitates this transformation under mild conditions, typically at room temperature, minimizing thermal degradation of the sensitive cyano group.
Following the isolation of the cyano-amide intermediate, the process advances to a high-pressure hydrogenation step that serves a dual purpose: reduction and protection. Under a hydrogen pressure of 2.0MPa and at an elevated temperature of 80°C, the palladium on carbon (Pd/C) catalyst facilitates the reduction of the nitrile moiety to a primary amine. However, in the presence of excess BOC anhydride, this newly generated amine is immediately trapped as a carbamate. This in situ protection is a masterstroke of process chemistry, as it prevents the reduced amine from reacting with remaining nitrile groups or undergoing intermolecular condensation. The result is a crude product consisting primarily of poly-BOC substituted Spermidine, which is chemically distinct from the starting materials and side products. This distinctness allows for the effective removal of catalyst residues and organic impurities prior to the final acidic deprotection, ensuring that the final distillation yields a product with a purity profile suitable for sensitive biological applications without the need for preparative HPLC.
How to Synthesize Spermidine Efficiently
The operational protocol derived from this patent offers a clear roadmap for laboratory and pilot-scale synthesis, emphasizing safety and reproducibility. The procedure begins with the careful stoichiometric control of the coupling reagents, specifically maintaining a molar ratio of cyanoacetic acid to the protected diamine near 1.1:1 to ensure complete consumption of the more valuable amine component. Following the coupling, the hydrogenation step requires specialized equipment capable of withstanding 2.0MPa pressure, highlighting the need for rigorous safety protocols when handling hydrogen gas at elevated temperatures. The final workup involves a pH-swing extraction, where the protected intermediate is deprotected under acidic conditions (pH 1) and then liberated as the free base at pH 9 before distillation. For a comprehensive, step-by-step guide including specific quantities, stirring rates, and workup details, please refer to the standardized synthesis instructions below.
- Couple N-BOC-1,4-butanediamine with cyanoacetic acid using DCC in DCM to form the protected intermediate.
- Perform catalytic hydrogenation using Pd/C and BOC anhydride in methanol under 2.0MPa pressure at 80°C.
- Deprotect the crude product using acidic methanol followed by basification and fractional distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this BOC-protected synthesis route offers transformative benefits for supply chain stability and cost structures in the manufacturing of polyamine intermediates. By eliminating the reliance on acrylonitrile, a volatile and highly toxic monomer, manufacturers can significantly reduce the regulatory burden and safety costs associated with raw material handling and storage. Furthermore, the ability to purify the final product via simple fractional distillation, rather than complex chromatography, drastically reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in waste disposal and raw material procurement. The high conversion rates reported in the patent examples suggest that raw material utilization is optimized, minimizing the loss of expensive protected starting materials and improving the overall atom economy of the process.
- Cost Reduction in Manufacturing: The streamlined purification process inherent to this method eliminates the need for expensive and time-consuming chromatographic columns, which are often the bottleneck in polyamine production. By relying on distillation for final purification, the process reduces both the capital expenditure on equipment and the operational expenditure on consumables. Additionally, the use of standard reagents like DCC and Pd/C, which are readily available in bulk quantities, ensures that the cost of goods sold remains competitive even at smaller batch sizes. The high purity achieved directly from distillation also reduces the need for recrystallization cycles, further lowering energy consumption and labor costs associated with extended processing times.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including N-BOC-1,4-butanediamine and cyanoacetic acid, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. Unlike the traditional route which relies on the price-volatile acrylonitrile market, this pathway utilizes stable carboxylic acid derivatives that are less susceptible to petrochemical feedstock fluctuations. The robustness of the reaction conditions, which tolerate slight variations in temperature and pressure without significant yield loss, ensures consistent batch-to-batch quality. This reliability is critical for long-term supply agreements with pharmaceutical clients who require stringent consistency in their active ingredient supply chains.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as hydrogenation and distillation that are standard in modern chemical plants. The absence of heavy metal catalysts other than the recoverable Pd/C, and the avoidance of chlorinated solvents in the final steps, aligns well with increasingly strict environmental regulations regarding VOC emissions and heavy metal residuals. The simplified waste stream, primarily consisting of aqueous salts and recoverable organic solvents, facilitates easier treatment and compliance with green chemistry principles. This environmental compatibility not only future-proofs the manufacturing site against regulatory changes but also enhances the brand value for customers seeking sustainable sourcing options for their pharmaceutical portfolios.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Spermidine synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing capabilities. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: Why is the traditional acrylonitrile route for Spermidine problematic?
A: The conventional method using 1,4-diamine and acrylonitrile suffers from difficult removal of excess starting materials and generates impurities with similar physicochemical properties, making purification industrially challenging.
Q: What is the key advantage of the BOC-protection strategy in this patent?
A: The BOC group protects the amine functionality during the coupling and reduction steps, preventing polymerization and side reactions, which significantly simplifies the final purification via distillation.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reagents like DCC and Pd/C, operates at manageable temperatures (80°C), and avoids complex chromatography, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spermidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN116239475A requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and precision distillation columns, allowing us to meet the stringent purity specifications required for pharmaceutical grade Spermidine. With our rigorous QC labs and commitment to continuous process improvement, we guarantee a supply of high-quality intermediates that support your drug development timelines without compromise.
We invite you to engage with our technical team to explore how this optimized synthesis route can enhance your project's economics and supply security. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential ROI associated with switching to this BOC-protected methodology. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to bring this innovative chemistry to life, ensuring a reliable and cost-effective supply of Spermidine for your critical applications.
