Revolutionizing Clomazone Production: A Deep Dive into Green Aqueous Catalysis and Commercial Scalability
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, with Clomazone standing out as a critical active ingredient for pre-emergence weed control in soybean and corn cultivation. A pivotal advancement in this sector is detailed in Chinese Patent CN113024479B, which discloses a novel, environmentally benign preparation method that fundamentally restructures the synthetic pathway. This patent introduces a groundbreaking approach utilizing hydroxylamine hydrochloride as the starting material, facilitated by a proprietary ether catalyst system, to achieve high-yield production of the key intermediate 3-chloro-N-hydroxy-2,2-dimethylpropionamide. Unlike traditional methods that rely on hazardous organic solvents and complex purification steps, this invention leverages a fully aqueous reaction medium, demonstrating a sophisticated balance between chemical efficiency and ecological responsibility. For R&D directors and procurement strategists, this technology represents a significant leap forward, offering a robust framework for scaling production while adhering to increasingly stringent global environmental regulations. The integration of a self-made ether catalyst not only optimizes reaction kinetics but also ensures exceptional product purity, addressing the persistent challenge of impurity profiles in complex heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clomazone has been plagued by significant technical and economic bottlenecks that hinder efficient large-scale manufacturing. One prevalent conventional route, often referred to as the o-chlorobenzaldehyde method, initiates with the condensation of o-chlorobenzaldehyde and hydroxylamine to form an oxime, followed by a reduction step to generate o-chlorobenzyl hydroxylamine. As illustrated in the reaction scheme below, this reduction phase is notoriously difficult to control, often failing to stop selectively at the hydroxylamine stage, which leads to substantial byproduct formation and limits yields to approximately 80%. Furthermore, this pathway necessitates the use of expensive reducing agents and vast quantities of organic solvents, resulting in a cumbersome purification process that inflates production costs and generates excessive chemical waste. Another common approach involves the ammonolysis of chloropivaloyl chloride, but existing literature indicates that the synthesis of the crucial 3-chloro-N-hydroxy-2,2-dimethylpropionamide intermediate in this route typically suffers from suboptimal yields. These inefficiencies compound through subsequent cyclization and benzylation steps, ultimately rendering the overall process economically unviable for modern, cost-sensitive agrochemical supply chains.
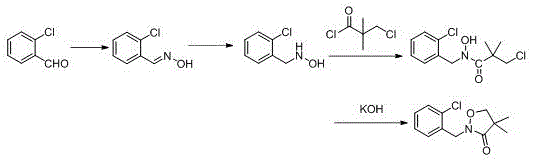
The Novel Approach
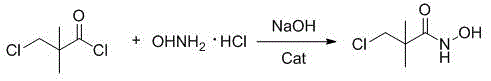
In stark contrast to these legacy methods, the technology disclosed in CN113024479B introduces a streamlined, high-efficiency pathway that circumvents the inherent flaws of previous generations. The core innovation lies in the direct acylation of hydroxylamine hydrochloride with chloropivaloyl chloride, mediated by a specifically engineered ether catalyst system composed of tetrahydrofuran and 18-crown-6 ether. This catalytic environment dramatically accelerates the reaction rate and drives the conversion to near completion, achieving yields exceeding 95% for the initial amidation step. Crucially, the entire process is conducted in water, eliminating the safety hazards and disposal costs associated with volatile organic solvents. The subsequent cyclization to form 4,4-dimethyl-3-isoxazolone and the final benzylation are performed in a telescoped manner, meaning the intermediate does not require isolation before proceeding to the next stage. This "one-pot" philosophy significantly reduces unit operations, minimizes material handling losses, and simplifies the overall workflow, making it exceptionally suitable for continuous or batch processing in a commercial plant setting.
Mechanistic Insights into Ether-Catalyzed Amidation and Cyclization
The success of this novel synthesis hinges on the precise mechanistic role of the self-made ether catalyst during the amidation phase. The catalyst mixture, consisting of tetrahydrofuran and 18-crown-6 ether in a specific molar ratio, functions as a highly effective phase transfer and activation system. In the aqueous medium, the crown ether component complexes with sodium cations from the base, effectively stripping the solvation shell around the hydroxylamine anion. This "naked" anion exhibits significantly enhanced nucleophilicity, allowing it to attack the carbonyl carbon of chloropivaloyl chloride with much greater vigor than in uncatalyzed systems. This mechanism not only suppresses side reactions such as hydrolysis of the acid chloride but also ensures that the reaction proceeds rapidly even at mild temperatures ranging from -5°C to 0°C. The result is a clean transformation that yields 3-chloro-N-hydroxy-2,2-dimethylpropionamide with a purity of 99.0% as determined by HPLC, setting a superior foundation for the downstream steps.
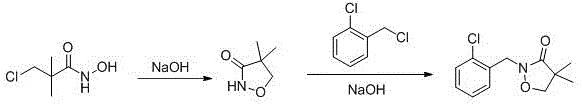
Following the formation of the hydroxamic acid intermediate, the process transitions seamlessly into cyclization and benzylation, driven by careful pH control and the use of flake base. The cyclization step involves an intramolecular nucleophilic substitution where the hydroxamic oxygen attacks the alkyl chloride moiety, closing the isoxazolone ring. By maintaining the pH at approximately 9.0 using sodium hydroxide, the reaction environment remains optimal for ring closure while preventing the degradation of the sensitive heterocyclic structure. The final benzylation utilizes o-chlorobenzyl chloride and is catalyzed by flake base in the presence of a phase transfer catalyst like tetrabutylammonium bromide. This step attaches the essential 2-chlorobenzyl group to the nitrogen atom of the isoxazolone ring. The use of water as the solvent here is particularly advantageous, as it facilitates the easy separation of the organic product upon cooling and extraction, while the inorganic salts remain dissolved in the aqueous phase. This mechanistic elegance ensures that the final Clomazone product is obtained with high optical purity and minimal impurity burden, satisfying the rigorous quality standards required for agrochemical registration.
How to Synthesize Clomazone Efficiently
The practical implementation of this synthesis route requires strict adherence to the optimized conditions regarding temperature, pH, and catalyst loading to maximize the benefits of the aqueous system. The process begins with the preparation of the ether catalyst mixture, followed by the controlled addition of reagents to manage the exothermic nature of the amidation. Subsequent steps involve precise pH adjustments to trigger cyclization and finally heating for the benzylation reaction. The ability to telescope these steps without intermediate isolation is a key operational advantage that reduces cycle time and equipment footprint. For detailed standard operating procedures and specific parameter ranges validated by patent data, please refer to the technical guide below.
- React hydroxylamine hydrochloride with chloropivaloyl chloride in water using a THF and 18-crown-6 ether catalyst mixture at controlled pH to form 3-chloro-N-hydroxy-2,2-dimethylpropionamide.
- Perform cyclization by adjusting pH with sodium hydroxide to generate 4,4-dimethyl-3-isoxazolone without isolating the intermediate.
- Complete the synthesis via benzylation with o-chlorobenzyl chloride using flake base and a phase transfer catalyst in water at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous catalytic technology translates into tangible strategic advantages that extend far beyond simple yield metrics. The shift from organic solvents to water as the primary reaction medium fundamentally alters the cost structure of manufacturing, removing the volatility associated with petrochemical-derived solvent pricing and availability. Furthermore, the elimination of solvent recovery distillation columns and the reduction in hazardous waste generation significantly lower the operational expenditure related to environmental compliance and waste disposal. This process design inherently de-risks the supply chain by relying on commodity chemicals like hydroxylamine hydrochloride and chloropivaloyl chloride, which are widely available and less susceptible to supply disruptions compared to specialized reducing agents or exotic reagents used in older methods. The robustness of the reaction conditions also implies a higher tolerance for minor variations in raw material quality, ensuring consistent output even when sourcing from diverse suppliers.
- Cost Reduction in Manufacturing: The implementation of this water-based system eliminates the capital and operational costs associated with solvent recovery and recycling infrastructure. By avoiding the use of expensive reducing agents and minimizing the number of isolation steps, the overall consumption of utilities such as steam and electricity is drastically reduced. The high yield of the initial amidation step ensures that raw material utilization is maximized, directly lowering the cost of goods sold per kilogram of active ingredient produced. Additionally, the simplified work-up procedure, which relies on filtration and extraction rather than complex chromatography or crystallization from organic media, reduces labor hours and processing time, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials like hydroxylamine hydrochloride and chloropivaloyl chloride ensures a stable and resilient supply chain that is not dependent on niche chemical vendors. The mild reaction conditions and the use of water as a solvent reduce the safety risks associated with transportation and storage of hazardous flammable liquids, simplifying logistics and insurance requirements. The telescoped nature of the synthesis reduces the lead time for production batches, allowing for more responsive inventory management and faster fulfillment of customer orders. This agility is crucial for meeting seasonal demand spikes in the agrochemical sector without the need for excessive safety stock.
- Scalability and Environmental Compliance: The aqueous nature of the process makes it inherently safer and easier to scale from pilot plant to multi-ton commercial production without the thermal runaway risks often associated with organic solvent reactions. The reduction in volatile organic compound (VOC) emissions aligns perfectly with global trends towards greener manufacturing and helps facilities meet stringent environmental regulations with minimal additional investment in abatement technology. The simplicity of the effluent stream, primarily consisting of inorganic salts and water, simplifies wastewater treatment processes, further enhancing the sustainability profile of the manufacturing site. This environmental compatibility future-proofs the production asset against tightening regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Clomazone synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this method. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition.
Q: What is the primary advantage of the ether catalyst system in Clomazone synthesis?
A: The self-made ether catalyst, comprising tetrahydrofuran and 18-crown-6 ether, significantly enhances the nucleophilicity of hydroxylamine, driving the amidation yield from typical levels up to over 95% while maintaining high purity.
Q: How does this process address environmental concerns in agrochemical manufacturing?
A: By utilizing water as the sole solvent throughout the reaction sequence, the process eliminates the need for volatile organic compounds (VOCs), drastically reducing waste treatment costs and environmental impact compared to traditional organic solvent-based routes.
Q: Is the intermediate isolation required between cyclization and benzylation?
A: No, the process is designed as a telescoped sequence where the 4,4-dimethyl-3-isoxazolone intermediate generated from cyclization is directly subjected to benzylation without separation, streamlining operations and improving overall throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clomazone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, green synthesis routes like the one described in CN113024479B requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO specializing in complex agrochemical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous catalytic systems, ensuring that the theoretical benefits of high yield and purity are fully realized in commercial batches. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of Clomazone meets the exacting standards required for global herbicide formulation.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this aqueous route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of high-quality Clomazone for the upcoming growing season.
