Strategic Analysis of Pitavastatin Calcium Manufacturing: High-Yield Routes for Global API Sourcing
Introduction to Advanced Pitavastatin Calcium Synthesis
The global demand for effective lipid-lowering agents continues to drive innovation in the pharmaceutical sector, with Pitavastatin Calcium standing out as a potent HMG-CoA reductase inhibitor. This comprehensive technical analysis focuses on the preparation method disclosed in patent CN103508947A, which offers a robust alternative to traditional synthetic pathways. The invention details a streamlined process for treating hyperlipidemia, addressing critical pain points such as cost, yield, and environmental impact. By leveraging a rational design that avoids severe toxicity and utilizes market-available reagents, this method represents a significant leap forward in API manufacturing efficiency. The structural integrity and stereochemistry of the final product are paramount, as illustrated by the precise molecular architecture required for therapeutic efficacy.
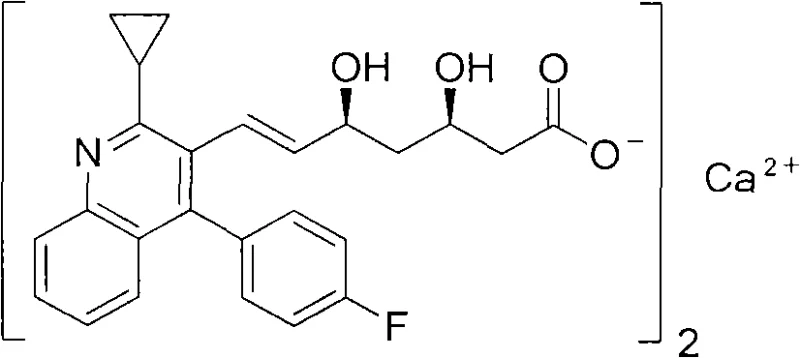
Furthermore, the strategic importance of this synthesis lies in its ability to bypass the limitations of earlier achiral and chiral induction methods. While previous art often struggled with difficult product isolation and low practical applicability, this novel approach ensures a reasonable design with simple process flows. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply chain. The method not only promises high yields but also simplifies the purification of intermediates, thereby reducing the overall burden on quality control laboratories and production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pitavastatin Calcium has been categorized into achiral and chiral synthetic routes, both of which present substantial challenges for industrial scale-up. Achiral methods, such as those cited in US5011930 and Tetrahedron Letter 1993, often encounter significant difficulties in product separation, rendering them impractical for large-scale commercial application without extensive downstream processing. More critically, chiral synthetic routes, while attempting to address stereochemistry, frequently rely on the use of precious metal catalysts or complex boron compounds. These reagents not only inflate raw material costs but also introduce severe environmental and safety hazards due to their toxicity and the difficulty of removing trace metal residues from the final API.
Additionally, conventional chiral induction strategies typically necessitate reaction systems operating at extremely low temperatures, often around -70°C. Maintaining such cryogenic conditions on a multi-ton scale requires specialized refrigeration equipment and immense energy consumption, drastically driving up operational expenditures (OPEX). The combination of expensive catalysts, hazardous reagents, and energy-intensive conditions creates a bottleneck for manufacturers aiming to reduce the cost of goods sold (COGS). Consequently, there is a pressing industry need for a method that can achieve high stereoselectivity under mild conditions without compromising on yield or purity.
The Novel Approach
The patented method described in CN103508947A introduces a transformative approach that mitigates these historical inefficiencies through a carefully engineered eight-step sequence. By utilizing 2-amino-4'-fluorobenzophenone and ethyl 3-cyclopropyl-3-oxo-propanoate as starting materials, the process establishes the quinoline core through a cyclization reaction that is both robust and scalable. Unlike prior art, this route avoids the use of reagents with severe toxicity and serious pollution, aligning with modern green chemistry principles. The elimination of precious metals and the avoidance of extreme cryogenic temperatures significantly lower the barrier to entry for manufacturing, making the process economically viable for a broader range of producers.
Moreover, the novelty of this approach is evident in its post-treatment simplicity. The intermediates generated throughout the synthesis, such as the quinoline methanol and the phosphonium salts, are designed to be easily purified, often through straightforward crystallization or extraction techniques. This reduces the reliance on complex chromatographic columns, which are often a bottleneck in batch processing. The final steps involve a highly efficient salt formation and purification using chiral amines, ensuring that the optical purity meets stringent pharmacopeial standards. This holistic optimization of the synthetic route translates directly into enhanced process reliability and reduced production lead times.
Mechanistic Insights into Quinoline Cyclization and Wittig Olefination
The core of this synthetic strategy relies on the efficient construction of the 2-cyclopropyl-4-(4-fluorophenyl)quinoline scaffold, which serves as the lipophilic anchor of the molecule. The mechanism begins with the cyclization of 2-amino-4'-fluorobenzophenone with ethyl 3-cyclopropyl-3-oxo-propanoate, likely proceeding through a condensation followed by intramolecular ring closure catalyzed by p-toluenesulfonic acid. This step is critical as it establishes the heterocyclic system that defines the drug's class. Subsequent reduction with Lithium Aluminum Hydride (LiAlH4) converts the ester functionality into the corresponding methanol, a transformation that must be carefully controlled to prevent over-reduction or side reactions on the fluorophenyl ring. The use of LiAlH4, while reactive, is a well-understood industrial reagent that allows for high conversion rates when managed correctly.
Following the formation of the quinoline methanol, the pathway employs a classic Wittig reaction strategy to attach the chiral side chain. The methanol is first converted to a bromomethyl derivative using phosphorus tribromide, which is then reacted with triphenylphosphine to generate the phosphonium salt. This salt is subsequently treated with a strong base, such as n-Butyl Lithium, to form the reactive ylide. The condensation of this ylide with the chiral ester (3R, 5S)-6-oxo-3,5-isopropylidene-dioxo-6-heptenoic acid tert-butyl ester is the stereochemistry-defining step. The reaction conditions, specifically the temperature control during ylide formation and addition, are optimized to maximize the E-isomer formation, which is crucial for the biological activity of the final statin. This mechanistic precision ensures that the complex chiral centers are installed with high fidelity.
How to Synthesize Pitavastatin Calcium Efficiently
The execution of this synthesis requires precise adherence to the reaction parameters outlined in the patent embodiments to ensure reproducibility and high yield. The process is divided into distinct stages: the construction of the quinoline core, the preparation of the phosphonium coupling partner, and the final assembly and resolution of the chiral acid. Each stage builds upon the previous one, with intermediate purification steps designed to remove specific byproducts before they can interfere with subsequent transformations. For process chemists, the key to success lies in the careful control of stoichiometry during the Wittig coupling and the rigorous management of pH during the hydrolysis and salt formation steps.
- Cyclization of 2-amino-4'-fluorobenzophenone with ethyl 3-cyclopropyl-3-oxo-propanoate followed by LiAlH4 reduction to form the quinoline methanol intermediate.
- Conversion of the methanol intermediate to a phosphonium salt via bromination and reaction with triphenylphosphine.
- Wittig condensation with a chiral ester followed by hydrolysis, deprotection, and calcium salt formation to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing route offers compelling advantages that directly address the priorities of procurement managers and supply chain heads. The primary benefit is the substantial reduction in manufacturing costs driven by the elimination of expensive and scarce reagents. By avoiding precious metal catalysts and complex boron compounds, the raw material bill of materials (BOM) is significantly optimized. Furthermore, the simplified post-treatment procedures reduce the consumption of solvents and the labor hours associated with complex purification workflows. This efficiency translates into a more competitive pricing structure for the final API, allowing pharmaceutical companies to improve their margins or offer more affordable treatments to the market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commercially available starting materials and the avoidance of extreme reaction conditions. Traditional methods requiring -70°C operations incur massive energy costs for refrigeration; by contrast, this method operates under much milder thermal conditions, drastically lowering utility expenses. Additionally, the high yields reported in key steps, particularly the final salt formation which approaches quantitative levels, minimize material waste. The removal of toxic reagents also reduces the costs associated with hazardous waste disposal and environmental compliance, further enhancing the overall cost-effectiveness of the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals rather than specialized, single-source catalysts. Reagents such as triphenylphosphine, lithium aluminum hydride, and common organic solvents are widely available from multiple global suppliers, reducing the risk of supply disruptions. The robustness of the synthetic route means that production schedules are less likely to be impacted by failed batches or difficult purifications. This reliability is crucial for maintaining continuous inventory levels and meeting the demanding delivery timelines of downstream pharmaceutical formulators who depend on a steady stream of high-quality active ingredients.
- Scalability and Environmental Compliance: The design of this process inherently supports commercial scale-up, moving seamlessly from kilogram to multi-ton production without fundamental changes to the chemistry. The absence of severe toxicity and serious pollution reagents simplifies the regulatory approval process for manufacturing sites, as it aligns with increasingly stringent environmental, health, and safety (EHS) regulations. The simplified waste stream, devoid of heavy metals, makes effluent treatment more straightforward and cost-effective. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the corporate social responsibility (CSR) profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Pitavastatin Calcium using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this synthetic route for their own supply chains or for assessing the quality of potential vendors.
Q: How does this synthesis method improve upon conventional chiral induction routes?
A: Unlike conventional methods that rely on expensive precious metal catalysts or boron compounds and require extreme low temperatures (e.g., -70°C), this patented route utilizes commercially available raw materials and milder reaction conditions, significantly reducing operational complexity and equipment costs.
Q: What are the key purity advantages of the described purification process?
A: The process features simplified post-treatment steps, including direct crystallization and specific chiral amine resolution, which effectively remove impurities without requiring complex chromatographic separations at every stage, ensuring high optical purity and chemical integrity.
Q: Is this manufacturing route scalable for commercial API production?
A: Yes, the route is designed for scalability with high yields reported in key steps (up to 98% in final salt formation) and avoids reagents with severe toxicity or serious pollution, making it compliant with modern environmental and safety standards for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pitavastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying efficient and compliant synthetic routes for high-value APIs like Pitavastatin Calcium. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103508947A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for global pharmaceutical registration. Our commitment to process excellence means we can deliver this complex molecule with the consistency and quality necessary for your drug development and commercialization needs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of our production methods compared to traditional sources. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability. Let us collaborate to optimize your API sourcing strategy and drive value for your organization.
