Advanced Palladium-Catalyzed Synthesis of Ibuprofen and Analogues for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of Ibuprofen and Analogues for Commercial Scale-up
Introduction to Green Chemistry in NSAID Manufacturing
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for producing non-steroidal anti-inflammatory drugs (NSAIDs), with Ibuprofen remaining a cornerstone of pain management therapy globally. A significant technological breakthrough in this domain is documented in patent CN102010323A, which outlines a novel method for synthesizing Ibuprofen and its structural analogues through a palladium-catalyzed decarboxylation coupling reaction. This approach represents a paradigm shift from traditional carbonylation methods that often rely on toxic carbon monoxide gas, offering a safer and more environmentally benign alternative for high-volume production. By utilizing 1-halo-4-isobutylbenzene and cyanoacetate derivatives as key starting materials, this process achieves high conversion rates while generating only inorganic salts and carbon dioxide as byproducts. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is critical for evaluating long-term supply chain stability and cost efficiency in the competitive landscape of pharmaceutical intermediates. The following analysis dissects the technical merits and commercial implications of this innovative synthetic strategy.
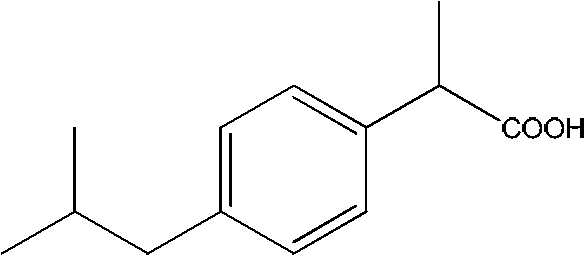
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ibuprofen has relied on processes such as the Boots or Hoechst methods, which involve multiple steps including Friedel-Crafts acylation, Darzens condensation, and hydrolysis, often requiring harsh reagents and generating substantial waste. Some newer carbonylation routes, while shorter, necessitate the use of high-pressure carbon monoxide, a highly toxic and flammable gas that imposes stringent safety regulations and expensive infrastructure requirements on manufacturing facilities. These traditional pathways often suffer from low atom economy and complex purification steps needed to remove heavy metal catalysts or acidic residues, leading to increased operational expenditures and environmental compliance burdens. Furthermore, the reliance on specific acyl chlorides and the generation of stoichiometric amounts of salt waste can create bottlenecks in supply chains, particularly when regulatory pressures on waste disposal intensify. For supply chain heads, these factors translate into higher risks of production delays and fluctuating costs associated with hazardous material handling and disposal protocols.
The Novel Approach
In contrast, the method described in patent CN102010323A introduces a streamlined pathway that leverages palladium-catalyzed decarboxylative coupling to construct the critical carbon-carbon bond directly. This novel approach utilizes readily available 1-halo-4-isobutylbenzene and cyanoacetate derivatives, bypassing the need for toxic carbon monoxide and reducing the overall step count significantly. The reaction conditions are robust, typically operating at temperatures between 120°C and 160°C in solvents like mesitylene or DMF, which are standard in fine chemical manufacturing and easy to recover. By eliminating the hazards associated with high-pressure CO gas, this method drastically simplifies the safety profile of the manufacturing plant, allowing for more flexible site selection and lower insurance premiums. The simplicity of the operation, combined with the generation of benign byproducts like CO2 and inorganic salts, positions this technology as a superior choice for cost reduction in Pharmaceutical Intermediates manufacturing, aligning perfectly with modern green chemistry mandates.
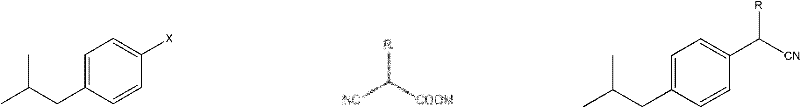
Mechanistic Insights into Pd-Catalyzed Decarboxylation Coupling
The core of this synthetic innovation lies in the palladium-catalyzed decarboxylation coupling mechanism, which facilitates the formation of the aryl-alkyl bond with high precision and selectivity. The reaction initiates with the oxidative addition of the aryl halide to the palladium center, followed by the coordination and subsequent decarboxylation of the cyanoacetate derivative, which acts as a nucleophilic partner. This decarboxylation step is crucial as it drives the reaction forward by releasing carbon dioxide, a thermodynamic sink that prevents the reverse reaction and ensures high yields, often exceeding 90% in optimized examples. The use of specific organic phosphine ligands, such as 9,9-dimethyl-4,5-bis(di-tert-butylphosphino)xanthene, stabilizes the palladium complex and enhances the catalytic turnover number, allowing for low catalyst loading of approximately 0.1% to 0.4% mol. For R&D teams, this mechanistic efficiency means fewer side reactions and a cleaner crude product profile, which significantly reduces the burden on downstream purification processes like chromatography or crystallization.
Impurity control is another critical aspect where this mechanism excels, particularly in avoiding the formation of regioisomers or over-alkylated byproducts common in Friedel-Crafts chemistries. The specificity of the palladium catalyst ensures that the coupling occurs exclusively at the halogenated position, preserving the integrity of other functional groups on the aromatic ring, which is vital when synthesizing analogues like Ketoprofen or Suprofen. Furthermore, the subsequent hydrolysis step, whether performed under acidic or basic conditions, is highly selective for the nitrile group, converting it to the carboxylic acid without affecting the aromatic core or the isobutyl side chain. This high level of chemoselectivity results in a final product with a superior purity profile, meeting the stringent specifications required for reliable Pharmaceutical Intermediates supplier status. The ability to control the stereochemistry and minimize impurities at the molecular level translates directly into reduced quality control costs and higher batch consistency for commercial clients.

How to Synthesize Ibuprofen Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, specifically the choice of solvent and the ratio of catalyst to ligand, to maximize efficiency and yield. The patent details a procedure where 1-halo-4-isobutylbenzene is reacted with potassium 2-cyanopropionate in mesitylene at 140°C for 20 hours, yielding the nitrile intermediate which is subsequently hydrolyzed. Detailed standardized synthesis steps see the guide below.
- Perform palladium-catalyzed decarboxylation coupling between 1-halo-4-isobutylbenzene and cyanoacetate derivatives in mesitylene at 140°C.
- If required, conduct methylation using methyl halides and strong base hydrides to form the propionitrile intermediate.
- Execute hydrolysis using strong acid or base at 100°C to 150°C to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed route offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for NSAID intermediates. The elimination of toxic carbon monoxide not only enhances safety but also removes the need for specialized high-pressure reactors and gas handling infrastructure, leading to significant capital expenditure savings and lower operational overheads. Additionally, the raw materials required, such as halo-benzene derivatives and cyanoacetates, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague more exotic synthetic routes. This stability ensures consistent production schedules and reliable delivery timelines, which are critical for maintaining the continuity of downstream API manufacturing operations. By simplifying the waste stream to primarily inorganic salts and CO2, manufacturers can also anticipate lower environmental compliance costs and faster regulatory approvals for new production lines.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates multiple steps associated with traditional methods, directly reducing labor, energy, and solvent consumption per kilogram of product. By avoiding the use of expensive and hazardous reagents like carbon monoxide, the process inherently lowers the cost of goods sold (COGS) while maintaining high yield efficiency. The low loading of palladium catalyst, combined with the potential for ligand recovery, further contributes to cost optimization, making this route economically viable for large-scale production. These factors collectively drive substantial cost savings without compromising on the quality or purity of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as the key precursors are widely produced bulk chemicals with multiple global suppliers, mitigating single-source dependency risks. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or pressure, ensuring high batch-to-batch consistency and reducing the rate of failed batches. This reliability translates into shorter lead times for high-purity Pharmaceutical Intermediates, allowing downstream partners to maintain leaner inventory levels and respond more agilely to market demand fluctuations. Consequently, supply chain heads can forecast production capacity with greater confidence and secure long-term supply agreements.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by benign byproducts and high atom economy, facilitates easier scale-up from pilot plant to commercial tonnage without encountering significant waste treatment bottlenecks. Regulatory bodies increasingly favor processes that minimize hazardous waste, meaning this route is well-positioned for rapid approval in stringent markets like the EU and North America. The simplicity of the workup procedure, often involving basic extraction and crystallization, allows for seamless integration into existing multipurpose chemical manufacturing facilities. This scalability ensures that the commercial scale-up of complex Pharmaceutical Intermediates can be achieved rapidly to meet surging global demand for pain management therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data and claims found within the patent documentation. These answers are designed to provide clarity on safety, scalability, and product quality for potential partners evaluating this route for their supply chain. Understanding these details is essential for making informed decisions about adopting this greener and more efficient manufacturing process.
Q: Does this synthesis route avoid toxic carbon monoxide?
A: Yes, unlike traditional carbonylation methods, this patent describes a decarboxylative coupling route that eliminates the need for hazardous carbon monoxide gas, significantly improving operational safety.
Q: What are the primary byproducts of this reaction?
A: The process generates only inorganic salts and carbon dioxide as byproducts, aligning with green chemistry principles and simplifying waste treatment protocols.
Q: Is this method suitable for large-scale manufacturing?
A: The patent explicitly states the method is suitable for industrial production due to simple operation, wide availability of raw materials, and robust reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibuprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the palladium-catalyzed decarboxylation route for producing high-quality Ibuprofen and its analogues efficiently. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless and risk-mitigated. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global regulatory agencies. We are committed to leveraging this advanced chemistry to deliver superior value to our partners in the pharmaceutical sector.
We invite you to engage with our technical procurement team to discuss how this specific synthetic route can be optimized for your unique production requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a robust and sustainable supply chain for your critical NSAID intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
