Revolutionizing Diindolylmethane Production with Solvent-Free Ionic Liquid Catalysis for Global Pharma Supply Chains
Introduction to Green Catalytic Breakthroughs in Heterocyclic Synthesis
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste. A pivotal development in this arena is documented in patent CN103880728A, which discloses an efficient, environmentally friendly method for preparing diindolylmethane compounds. These compounds are critical scaffolds in medicinal chemistry, known for their potent biological and pharmacological activities, particularly in anticancer applications. The patent describes a novel approach utilizing a specifically designed acidic functional ionic liquid as a catalyst, enabling the reaction between indole derivatives and aldehydes or ketones under mild, solvent-free conditions at room temperature. This technological advancement addresses long-standing challenges in heterocyclic synthesis, such as the reliance on volatile organic compounds (VOCs) and the generation of hazardous heavy metal waste, positioning it as a cornerstone for modern green chemistry initiatives in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diindolylmethane derivatives has relied heavily on a variety of catalytic systems, including Bronsted acids like HBF4-SiO2 and Lewis acids such as ZnO, I2, FeCl3·6H2O, ZrCl4, CAN, RuCl3, and LiClO4. While these traditional methodologies have achieved varying degrees of success in laboratory settings, they are fraught with significant industrial drawbacks that hinder scalable and sustainable production. A primary concern is the frequent requirement for excessive amounts of catalyst to drive the reaction to completion, which not only inflates raw material costs but also complicates the purification process. Furthermore, many of these protocols necessitate the use of toxic organic solvents to solubilize reactants and facilitate mixing, creating substantial environmental liabilities and requiring expensive solvent recovery infrastructure. Perhaps most critically for pharmaceutical applications, the use of metal-containing catalysts introduces the risk of heavy metal residues in the final active pharmaceutical ingredient (API), necessitating rigorous and costly purification steps to meet strict regulatory limits on metal impurities, thereby extending lead times and increasing the overall cost of goods sold.
The Novel Approach
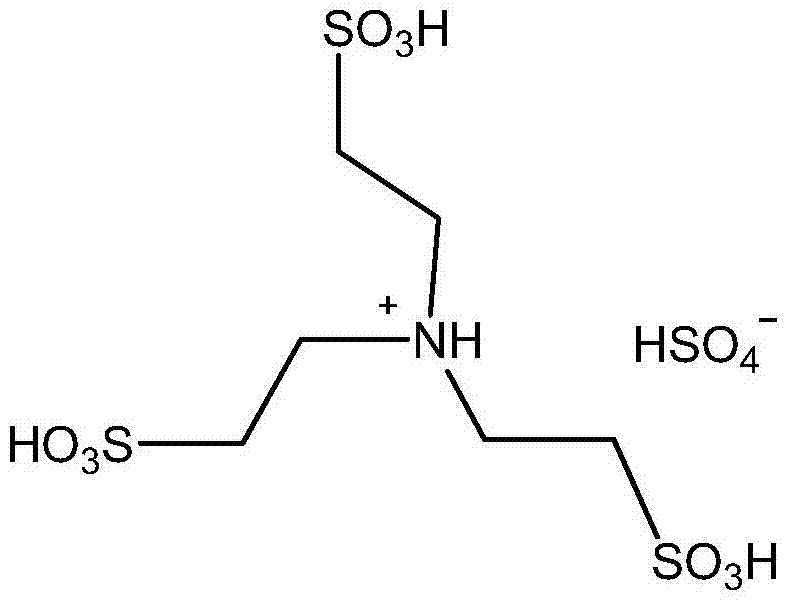
In stark contrast to these legacy methods, the technology outlined in CN103880728A introduces a transformative solvent-free protocol that utilizes a triethanolamine-derived acidic functional ionic liquid. This innovative catalyst system operates effectively at room temperature and normal pressure, eliminating the need for energy-intensive heating or cooling cycles that characterize conventional processes. The absence of organic solvents not only drastically reduces the environmental footprint by removing VOC emissions but also simplifies the work-up procedure, as the product can often be isolated through simple extraction without complex distillation steps. The molecular structure of the ionic liquid, featuring multiple sulfonic acid groups, provides strong Brønsted acidity that efficiently activates the carbonyl group of the aldehyde or ketone, facilitating rapid electrophilic substitution on the indole ring. This results in exceptionally high yields, often exceeding 90%, within reaction times as short as 2 to 30 minutes, demonstrating a level of efficiency and operational simplicity that far surpasses traditional metal-catalyzed routes while ensuring the final product is free from toxic metal contaminants.
Mechanistic Insights into Ionic Liquid Catalyzed Electrophilic Substitution
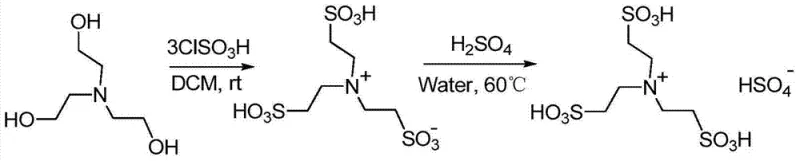
The efficacy of this synthesis route lies in the unique physicochemical properties of the triethanolamine-based ionic liquid, which acts as a dual-function medium providing both catalytic activity and a structured reaction environment. The catalyst is synthesized by the sulfonation of triethanolamine with chlorosulfonic acid, followed by an ion exchange with sulfuric acid to generate a highly acidic species containing both sulfonic acid groups and bisulfate anions. In the reaction mechanism, the protons from the sulfonic acid groups of the ionic liquid protonate the oxygen atom of the aldehyde or ketone carbonyl group, significantly increasing its electrophilicity. This activation allows the electron-rich C3 position of the indole nucleus to attack the carbonyl carbon more readily, forming a transient carbocation intermediate. The subsequent loss of a water molecule and a second nucleophilic attack by another indole molecule leads to the formation of the diindolylmethane skeleton. The ionic liquid stabilizes these charged intermediates through electrostatic interactions, lowering the activation energy of the rate-determining steps and allowing the reaction to proceed rapidly even at ambient temperatures without the need for external thermal energy input.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Traditional Lewis acid catalysts can sometimes promote side reactions such as polymerization of the indole or over-alkylation due to uncontrolled acidity or metal coordination effects. However, the tunable acidity of the functional ionic liquid allows for precise control over the reaction kinetics, minimizing the formation of polymeric byproducts and ensuring high selectivity for the desired 3,3'-substituted product. Furthermore, because the catalyst is non-volatile and remains in the polar phase during the extraction of the organic product with ethyl acetate, there is minimal risk of catalyst leaching into the product stream. This inherent phase separation capability ensures that the final diindolylmethane compound possesses a clean impurity profile, devoid of heavy metals or residual catalyst fragments, which is a critical quality attribute for pharmaceutical intermediates intended for downstream API synthesis where strict purity specifications must be met.
How to Synthesize Diindolylmethane Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and scalability, making it highly attractive for contract development and manufacturing organizations (CDMOs) looking to optimize their production workflows. The process begins with the preparation of the acidic functional ionic liquid, which serves as the reusable catalytic engine for the transformation. Once the catalyst is ready, the synthesis of the target diindolylmethane involves a straightforward mixing of stoichiometric amounts of indole (or its derivative) and the chosen aldehyde or ketone in the presence of a catalytic quantity of the ionic liquid. The reaction proceeds spontaneously at room temperature, monitored conveniently by thin-layer chromatography (TLC) to ensure complete conversion of the starting materials. Upon completion, the reaction mixture is subjected to a liquid-liquid extraction using ethyl acetate, which selectively partitions the organic product into the organic phase while retaining the ionic liquid catalyst in the aqueous or residue phase. This elegant separation strategy not only isolates the high-purity product but also facilitates the immediate recovery and regeneration of the catalyst for subsequent batches, embodying the principles of circular chemistry in industrial practice.
- Prepare the acidic functional ionic liquid catalyst by reacting triethanolamine with chlorosulfonic acid followed by ion exchange with sulfuric acid.
- In a reaction vessel, mix indole or its derivatives with aldehydes or ketones and add the ionic liquid catalyst (0.01 to 0.3 molar equivalent) under solvent-free conditions.
- Stir the mixture at room temperature (15-35°C) for 2 to 30 minutes, monitor by TLC, then extract with ethyl acetate and recover the catalyst from the raffinate phase for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid catalyzed technology represents a strategic opportunity to enhance supply security while simultaneously driving down the total cost of ownership for key pharmaceutical intermediates. The elimination of organic solvents from the reaction matrix removes the logistical burden and cost associated with purchasing, storing, and disposing of large volumes of volatile and often hazardous chemicals. This solvent-free approach significantly reduces the complexity of the manufacturing process, leading to shorter batch cycle times and higher throughput capacity without the need for capital-intensive solvent recovery units. Moreover, the ability to reuse the catalyst for multiple cycles without significant loss of activity translates directly into reduced raw material consumption, as the expensive catalytic species does not need to be replenished for every single batch. This durability of the catalyst system ensures a more stable and predictable cost structure, shielding the supply chain from fluctuations in the prices of specialty chemical reagents and providing a robust foundation for long-term supply agreements.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the drastic simplification of the unit operations involved. By operating under solvent-free conditions, manufacturers eliminate the substantial energy costs associated with heating large volumes of solvent and the subsequent energy-intensive distillation required for solvent removal and recovery. Additionally, the absence of heavy metal catalysts removes the need for specialized scavenging resins or complex purification steps designed to reduce metal content to ppm levels, which are often costly and result in product yield losses. The high atom economy and rapid reaction times further contribute to cost efficiency by maximizing the utilization of reactor volume and labor hours, allowing facilities to produce more product in less time with fewer resources, thereby achieving substantial cost savings in diindolylmethane manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and simplicity of this synthetic route. The starting materials, such as triethanolamine, indole, and various benzaldehydes, are commodity chemicals with well-established global supply chains, reducing the risk of raw material shortages that can plague processes relying on exotic or proprietary reagents. The mild reaction conditions (room temperature and normal pressure) mean that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, increasing the number of qualified manufacturing sites capable of producing the material. This flexibility in manufacturing location and equipment requirements diversifies the supply base and mitigates the risk of production disruptions due to equipment failure or regional constraints, ensuring a continuous and reliable flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is inherently safer and more straightforward due to the absence of exothermic risks associated with large-scale solvent handling and the elimination of toxic metal waste streams. The process aligns perfectly with increasingly stringent environmental, health, and safety (EHS) regulations, as it generates minimal waste and avoids the discharge of heavy metals into the environment. This compliance advantage reduces the regulatory burden and potential liability for manufacturers, facilitating faster approval times for new drug applications that utilize these intermediates. Furthermore, the ease of catalyst recovery and reuse minimizes the volume of chemical waste requiring treatment or disposal, supporting corporate sustainability goals and enhancing the overall environmental profile of the supply chain, which is becoming a key differentiator in vendor selection processes for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries based on the detailed experimental data and technical specifications provided in the patent literature. These questions address critical aspects of the process, including catalyst longevity, reaction scope, and product quality, providing a clear picture of the operational realities of implementing this green synthesis method. Understanding these technical nuances is essential for R&D directors and process engineers who are tasked with validating new routes and ensuring that they meet the rigorous standards required for Good Manufacturing Practice (GMP) production of pharmaceutical ingredients.
Q: Can the ionic liquid catalyst be reused in diindolylmethane synthesis?
A: Yes, according to patent CN103880728A, the acidic functional ionic liquid can be recovered from the raffinate phase after ethyl acetate extraction, vacuum-dried at 80°C, and reused for at least five consecutive batches without a significant decrease in reaction yield.
Q: What are the advantages of this solvent-free method over traditional Lewis acid catalysis?
A: Traditional methods often require toxic organic solvents, excessive amounts of metal catalysts like FeCl3 or ZnO, and harsh conditions leading to heavy metal residues. This novel method operates at room temperature without solvents, eliminating heavy metal contamination risks and simplifying post-treatment.
Q: What is the typical reaction time for this ionic liquid catalyzed process?
A: The reaction is remarkably fast, typically completing within 2 to 30 minutes at room temperature and normal pressure, depending on the specific indole and aldehyde substrates used, which significantly enhances throughput compared to conventional heating methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diindolylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid catalyzed synthesis of diindolylmethane compounds and are fully equipped to leverage this technology for our global partners. As a premier CDMO specializing in complex organic synthesis, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle solvent-free and green chemistry processes with the highest regard for safety and quality, supported by our rigorous QC labs that enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the pharmaceutical industry. We understand that consistency and reliability are paramount, and our dedicated technical team is committed to optimizing this route to deliver cost-effective, high-quality intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this advanced synthesis method can benefit your specific project requirements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this greener, more efficient process can impact your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our diindolylmethane portfolio and to discuss route feasibility assessments for your custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge chemical technologies and a supply chain partner dedicated to driving innovation and value in the global pharmaceutical market.
