Advanced Low-Temperature Carbonylation Technology for Commercial Scale Production of High-Purity 3,4-Dimethylbenzaldehyde
Introduction to Advanced Carbonylation Technology
The global demand for high-purity aromatic aldehydes, particularly 3,4-dimethylbenzaldehyde, has surged due to their critical role as precursors for polyolefin transparent nucleating agents such as DMDBS and various pharmaceutical intermediates. Addressing the historical challenges of selectivity and purification, patent CN100443456C introduces a groundbreaking low-temperature carbonylation methodology that redefines production standards. This innovative approach utilizes a sophisticated Lewis acid composite catalytic system to facilitate the formylation of o-xylene under remarkably mild conditions, specifically ranging from -10°C to 0°C at atmospheric pressure. By shifting away from harsh oxidation environments or high-pressure gas-phase reactions, this technology ensures a cleaner reaction profile with significantly reduced by-product formation. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols that guarantee product purity exceeding 99.5% without the need for complex, multi-stage purification trains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dimethylbenzaldehyde has been plagued by significant technical hurdles inherent to traditional oxidation and chloromethylation pathways. Conventional incomplete oxidation methods often suffer from poor selectivity, generating a complex cocktail of isomers and over-oxidized carboxylic acids that are notoriously difficult to separate due to similar boiling points. Furthermore, older Gattermann-Koch variations typically require elevated pressures around 0.7 MPa and temperatures between 20°C and 50°C, necessitating expensive high-pressure reactor infrastructure and posing substantial safety risks regarding the handling of gaseous hydrogen chloride. These legacy processes not only incur high operational costs due to energy-intensive separation units but also result in lower overall yields, often struggling to maintain single-pass conversion rates above 60% while producing substantial hazardous waste streams that complicate environmental compliance.
The Novel Approach
In stark contrast, the novel low-temperature carbonylation route detailed in the patent data leverages a unique in-situ generation of active formyl chloride species to drive the reaction with exceptional precision. By employing a composite catalyst system involving anhydrous aluminum trichloride and cuprous chloride within a 1,2-dichloroethane solvent matrix, the process achieves high conversion rates exceeding 70% under ambient pressure. This method effectively suppresses the formation of unwanted isomers and acidic by-products, streamlining the downstream workup to a simple extraction and vacuum distillation sequence. The ability to operate at sub-zero temperatures not only enhances the thermodynamic control over the electrophilic substitution but also drastically reduces the thermal load on the facility, translating to immediate energy savings and a smaller carbon footprint for the manufacturing site.
Mechanistic Insights into Lewis Acid-Catalyzed Formylation
The core of this technological breakthrough lies in the intricate interplay between the Lewis acid catalysts and the gaseous reagents, which creates a highly reactive electrophilic species capable of attacking the electron-rich aromatic ring of o-xylene. In the presence of the AlCl3 and CuCl composite, carbon monoxide and hydrogen chloride react to generate the transient formyl chloride (HCOCl) intermediate, which is immediately activated by the Lewis acid to form a potent acylium ion complex. This complex then undergoes electrophilic aromatic substitution preferentially at the position ortho to the methyl groups, driven by the steric and electronic directing effects of the substrate, ensuring the formation of the desired 3,4-isomer with high regioselectivity. The low-temperature environment is crucial here, as it stabilizes the reactive intermediates and prevents the decomposition of the formylating agent or the occurrence of side reactions such as polymerization or further oxidation.
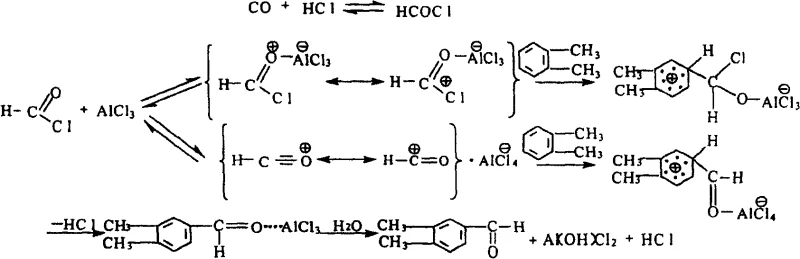
Furthermore, the mechanistic pathway includes a robust hydrolysis step that efficiently decomposes the aluminum complex post-reaction, facilitating the clean release of the aldehyde product and the regeneration of acid species that can be managed in the aqueous waste stream. This controlled decomposition is vital for impurity control, as it prevents the carryover of metal salts into the organic phase, thereby reducing the burden on the final distillation column. For quality assurance teams, understanding this mechanism confirms that the impurity profile will be dominated by unreacted starting materials rather than structurally similar isomers, making chromatographic purification highly effective. The result is a process that consistently delivers high-purity material suitable for sensitive applications in polymer science and pharmaceutical synthesis without the risk of trace metal contamination.
How to Synthesize 3,4-Dimethylbenzaldehyde Efficiently
Implementing this synthesis route requires precise control over gas flow rates and temperature gradients to maximize the efficiency of the catalytic cycle. The standardized protocol involves pre-cooling the solvent and substrate mixture before the sequential addition of the catalyst components to prevent localized exotherms that could degrade selectivity. Operators must maintain a strict 1:1 volume ratio of CO to HCl gas flow to ensure the stoichiometric generation of the formylating agent while avoiding the accumulation of excess acidic gas. Detailed standard operating procedures regarding the quenching of the reaction mass into ice-salt water and the subsequent vacuum distillation parameters are essential for reproducibility.
- Pre-cool a mixture of o-xylene and 1,2-dichloroethane solvent to between -10°C and 0°C using an ice-salt bath.
- Sequentially add the Lewis acid composite catalyst, specifically anhydrous aluminum trichloride and cuprous chloride, maintaining strict temperature control.
- Introduce carbon monoxide and hydrogen chloride gases at a 1: 1 volume ratio, allowing the reaction to proceed until conversion exceeds 70%.
- Quench the reaction mixture into ice-salt water, separate the organic layer, and purify the crude product via vacuum distillation to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this carbonylation technology offers profound advantages that directly impact the bottom line and supply chain resilience for buyers of fine chemical intermediates. The elimination of high-pressure equipment requirements significantly lowers the capital expenditure barrier for production scale-up, allowing for more flexible manufacturing footprints closer to end-user markets. Additionally, the use of commodity chemicals like o-xylene, carbon monoxide, and hydrochloric acid ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized or scarce reagents that often plague niche synthetic routes. The simplified purification process further reduces the consumption of auxiliary solvents and energy, contributing to a leaner operational model that is less susceptible to utility price volatility.
- Cost Reduction in Manufacturing: The transition to a low-temperature, atmospheric pressure process inherently reduces energy consumption associated with heating and pressurization, leading to substantial operational cost savings. By minimizing the formation of difficult-to-separate isomers, the process eliminates the need for extensive fractional distillation columns or complex crystallization steps, thereby lowering both utility usage and labor hours required for purification. The high catalytic activity of the Lewis acid composite system ensures efficient reagent utilization, reducing the overall raw material cost per kilogram of finished product and enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: Relying on bulk petrochemical feedstocks such as o-xylene and industrial gases like CO creates a robust supply chain that is less vulnerable to the disruptions often seen with specialized fine chemical precursors. The simplicity of the reaction setup allows for rapid turnaround times between batches, enabling manufacturers to respond quickly to fluctuations in market demand without lengthy campaign changeovers. This agility ensures a consistent flow of high-purity 3,4-dimethylbenzaldehyde to downstream users, securing the production schedules for critical polymer additives and pharmaceutical intermediates.
- Scalability and Environmental Compliance: The "clean" nature of this synthesis, characterized by minimal by-product generation and the absence of heavy metal oxidants, simplifies waste treatment and aligns with increasingly stringent environmental regulations. The ability to recover and recycle the 1,2-dichloroethane solvent further reduces the environmental footprint and raw material costs, supporting sustainability goals. Scalability is inherently supported by the mild reaction conditions, which reduce safety risks and allow for the use of standard glass-lined or stainless steel reactors, facilitating a smooth transition from pilot plant to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific carbonylation technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their supply chain. The answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for industrial application.
Q: What are the primary advantages of the low-temperature carbonylation method over traditional oxidation routes?
A: The low-temperature carbonylation method described in patent CN100443456C offers significantly higher selectivity and purity compared to oxidation methods. Traditional oxidation often yields complex mixtures of isomers and carboxylic acids that are difficult to separate, whereas this carbonylation route minimizes by-product formation, simplifying downstream purification and reducing overall processing costs.
Q: How does the Lewis acid composite catalyst system improve reaction efficiency?
A: The use of a composite catalyst system, typically comprising anhydrous aluminum trichloride and cuprous chloride, enhances the activation of carbon monoxide and facilitates the formation of the reactive formyl chloride intermediate at mild temperatures. This synergistic effect allows the reaction to proceed efficiently at atmospheric pressure and low temperatures (-10°C to 0°C), reducing energy consumption and equipment stress compared to high-pressure Gattermann-Koch variants.
Q: Is this synthesis method scalable for industrial production of polymer additives?
A: Yes, the process is highly scalable due to its operation at atmospheric pressure and the use of readily available raw materials like o-xylene and carbon monoxide. The mild reaction conditions and the ability to recover and recycle the solvent (1,2-dichloroethane) and unreacted starting materials make it economically viable for large-scale manufacturing of nucleating agents like DMDBS.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dimethylbenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and reliable supply for high-performance intermediates like 3,4-dimethylbenzaldehyde. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for polymer nucleating agents and pharmaceutical applications. We are committed to delivering not just a chemical product, but a comprehensive supply solution that supports your long-term growth and innovation.
We invite you to engage with our technical procurement team to discuss how this advanced low-temperature carbonylation route can optimize your current sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique volume requirements and quality targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
