Advanced Visible Photochromic Spirooxazine Compounds for Optoelectronic Manufacturing
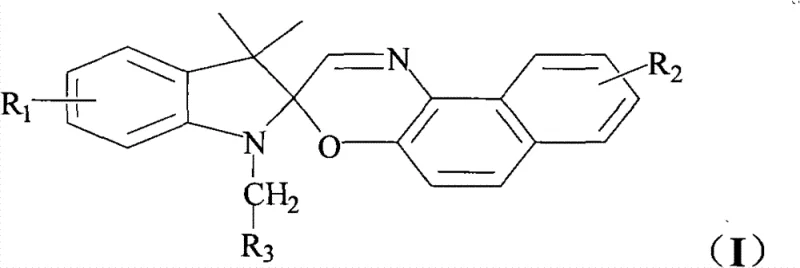
The chemical industry is witnessing a transformative shift in the development of smart materials, particularly within the realm of optoelectronics and data storage technologies. Patent CN102140120B introduces a groundbreaking class of visible photochromic compounds that overcome the historical limitations of traditional ultraviolet-responsive materials. By integrating a spirooxazine group with conjugated photosensitive chromogenic groups, this innovation enables precise molecular switching triggered by visible light wavelengths rather than harmful UV radiation. This structural modification facilitates charge transfer throughout the entire conjugated molecular system, inducing energy conversion between excited states that was previously unattainable with standard spirooxazine derivatives. The resulting compounds exhibit exceptional fatigue resistance and stability, making them ideal candidates for high-performance display materials and optical computing devices. For R&D directors and procurement specialists, this technology represents a significant opportunity to enhance product longevity and functionality in next-generation electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photochromic materials, particularly early-generation spirooxazines, have long suffered from critical performance bottlenecks that hinder their widespread commercial adoption in sensitive electronic environments. These conventional compounds typically rely on ultraviolet light to induce the ring-opening isomerization required for color change, which limits their utility in indoor applications or biological systems where UV exposure is restricted or damaging. Furthermore, the open-ring merocyanine forms generated during photo-excitation are often unstable in the presence of environmental oxygen, leading to rapid photodegradation and a loss of reversible switching capability over time. This inherent instability necessitates frequent replacement of materials in display devices, driving up long-term operational costs and complicating supply chain management for manufacturers. The lack of visible light sensitivity also restricts the design flexibility for engineers attempting to create multi-wavelength responsive systems for advanced optical data storage. Consequently, the industry has been in urgent need of a molecular architecture that can maintain robust photochromic cycles under visible light irradiation without compromising structural integrity.
The Novel Approach
The novel approach detailed in the patent data fundamentally reengineers the spirooxazine backbone by covalently linking it with organic chromophores that are specifically sensitive to visible light spectra. This conjugated connection creates a continuous electronic pathway that allows charge transfer to trigger the spirooxazine ring-opening mechanism directly through visible light absorption, bypassing the need for high-energy UV photons. By stabilizing the zwitterionic or diradical structures formed during the open state through this extended conjugation, the new compounds demonstrate markedly improved resistance to oxidative degradation and fatigue. This structural innovation ensures that the photochromic cycle can be repeated thousands of times with minimal loss of optical density or switching speed, a critical requirement for reliable memory storage and display technologies. Additionally, the modular nature of the synthesis allows for the customization of absorption wavelengths by varying the attached chromophoric groups, providing unparalleled flexibility for material scientists. This advancement effectively bridges the gap between laboratory curiosity and industrial viability for visible-light-activated smart materials.
Mechanistic Insights into Visible Light-Induced Spirooxazine Cyclization
The core mechanism driving the superior performance of these compounds lies in the intricate interplay between the spirooxazine moiety and the conjugated photosensitive groups attached to the molecular scaffold. Upon irradiation with visible light, the organic chromophore absorbs photon energy and enters an excited state, initiating a charge transfer process that propagates through the conjugated system to the spiro carbon-oxygen bond. This energy transfer weakens the spiro C-O bond, inducing heterolytic cleavage and causing the orthogonal ring system to rearrange into a planar, highly conjugated photomerocyanine structure. Unlike traditional systems where this open form is transient and unstable, the extended conjugation in this novel design delocalizes the charge density, significantly lowering the energy of the open state and protecting it from nucleophilic attack by oxygen. The reversible nature of this process is maintained through thermal relaxation or irradiation with a different wavelength, allowing the molecule to return to its closed spiro form without structural degradation. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize the response speed and contrast ratio of the final optoelectronic devices.
Impurity control is another critical aspect of the mechanistic profile that directly impacts the commercial viability of these photochromic materials. The synthesis route is designed to minimize side reactions that could generate non-photochromic byproducts, which would otherwise act as quenchers and reduce the overall efficiency of the optical switching. By utilizing specific reaction conditions such as controlled reflux temperatures and inert gas protection during the cyclization step, the formation of oxidative impurities is drastically reduced. The purification process, often involving silica gel column chromatography, effectively separates the desired trans-structure isomers from cis-isomers or unreacted starting materials that could interfere with the photochromic cycle. High purity is essential because even trace amounts of impurities can accelerate fatigue rates or shift the absorption maxima, rendering the material unsuitable for precision applications like optical computing. Therefore, the robustness of the synthetic mechanism in delivering high-purity products is a key value proposition for supply chain managers seeking consistent quality in raw materials.
How to Synthesize Visible Photochromic Spirooxazine Compounds Efficiently
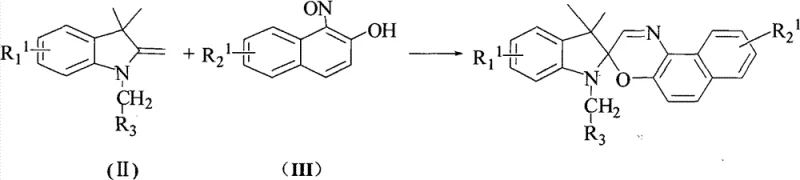
The synthesis of these advanced photochromic compounds follows a logical, multi-step pathway that balances chemical complexity with industrial practicality. The process begins with the preparation of key intermediates, specifically the indoline derivatives and nitroso-naphthol compounds, which serve as the building blocks for the spirooxazine core. These precursors are then subjected to a cyclization reaction under inert atmosphere to form the foundational spiro structure, followed by functionalization steps to introduce the visible-light-sensitive chromophores. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, molar ratios, and purification techniques required to achieve high yields and purity. This structured approach ensures reproducibility and safety, making it suitable for scale-up in a GMP-compliant manufacturing environment. By adhering to these optimized protocols, manufacturers can minimize waste and maximize the efficiency of their production lines.
- Perform cyclization reaction between indoline intermediate and nitroso-naphthol derivative under reflux in organic solvent.
- Execute halogenation and subsequent salt formation with triphenylphosphine to activate the hydroxymethyl group.
- Conduct condensation reaction with aldehyde derivatives under alkaline conditions to install conjugated chromophores.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis technology offers substantial benefits for procurement managers and supply chain heads looking to optimize costs and ensure material reliability. The process eliminates the need for expensive transition metal catalysts often required in alternative photochromic syntheses, thereby reducing raw material costs and simplifying the removal of metal residues from the final product. Furthermore, the use of common organic solvents such as ethanol, toluene, and tetrahydrofuran ensures that the supply chain is not dependent on scarce or highly regulated chemicals, enhancing supply continuity. The robustness of the reaction conditions, which operate at standard reflux temperatures without requiring extreme pressure or cryogenic cooling, translates to lower energy consumption and reduced equipment wear in large-scale reactors. These factors collectively contribute to a more sustainable and cost-effective manufacturing process that aligns with modern environmental compliance standards. For buyers, this means securing a reliable optoelectronic materials supplier who can deliver high-quality compounds without the volatility associated with complex catalytic systems.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers production expenses by avoiding the use of precious metal catalysts and reducing the number of purification steps required to achieve pharmaceutical-grade purity. By streamlining the halogenation and condensation stages, the process minimizes solvent usage and waste generation, leading to substantial cost savings in waste disposal and raw material procurement. The high yield of the cyclization step ensures that less starting material is wasted, further driving down the cost per kilogram of the final photochromic compound. Additionally, the stability of the intermediates allows for batch processing without the need for immediate downstream conversion, providing flexibility in production scheduling. These efficiencies make cost reduction in display material manufacturing a tangible reality for companies adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available chemical feedstocks such as aniline derivatives and naphthol compounds ensures that the supply chain remains resilient against market fluctuations and geopolitical disruptions. Since the synthesis does not depend on single-source proprietary catalysts, procurement teams can source reagents from multiple vendors, mitigating the risk of shortages. The robustness of the reaction also means that production timelines are predictable, reducing lead time for high-purity photochromic compounds and allowing for better inventory planning. This reliability is crucial for maintaining continuous operation in high-tech manufacturing facilities where material delays can halt entire production lines. Partnering with a supplier who utilizes this stable chemistry guarantees a steady flow of critical components for optoelectronic devices.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been designed with commercial scale-up of complex spirooxazines in mind, allowing for seamless transition from gram-scale laboratory batches to ton-scale industrial production. The reaction conditions are mild enough to be handled in standard glass-lined or stainless steel reactors, eliminating the need for specialized high-pressure equipment. Moreover, the process generates minimal hazardous byproducts, and the solvents used are easily recoverable and recyclable, supporting stringent environmental compliance and sustainability goals. This ease of scale-up ensures that supply can meet growing demand without compromising on quality or safety standards. For supply chain heads, this means a future-proof sourcing strategy that can adapt to increasing market volumes for smart materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochromic technology in industrial applications. These answers are derived directly from the patent specifications and practical synthesis experience, providing clarity on performance and integration. Understanding these details helps stakeholders make informed decisions about incorporating these materials into their product development roadmaps. The information covers stability, scalability, and application-specific performance metrics.
Q: How does this patent improve fatigue resistance compared to traditional spirooxazines?
A: The patent introduces conjugated organic chromophores that stabilize the zwitterionic structure formed during ring-opening, significantly reducing photodegradation caused by environmental oxygen.
Q: Can these compounds respond to visible light instead of just UV?
A: Yes, the integration of visible-light-sensitive chromophores allows the molecule to absorb visible wavelengths, triggering the spirooxazine ring-opening mechanism without requiring high-energy UV radiation.
Q: Is the synthesis process scalable for industrial production?
A: The method utilizes standard reflux conditions and common organic solvents like ethanol and toluene, avoiding exotic catalysts, which facilitates straightforward scale-up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Visible Photochromic Spirooxazine Compound Supplier
As a leader in fine chemical manufacturing, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of this advanced photochromic technology through our expert CDMO services. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is smooth and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of spirooxazine compound meets the exacting standards required for optoelectronic applications. We understand the critical nature of supply continuity in the electronics sector and have built a robust supply chain to support your long-term manufacturing needs. By leveraging our technical expertise, you can accelerate your product development cycles and bring innovative visible-light-responsive devices to market faster.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can optimize your supply chain for these high-value materials. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this novel synthesis route for your production lines. Our team is ready to provide specific COA data and route feasibility assessments tailored to your project's unique constraints and goals. Let us partner with you to drive innovation and efficiency in your optoelectronic manufacturing processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
