Advanced Synthesis of High-Stability Spirooxazine Photochromic Compounds for Optical Applications
Advanced Synthesis of High-Stability Spirooxazine Photochromic Compounds for Optical Applications
The field of organic photochromic materials has witnessed significant advancements with the disclosure of patent CN102363620A, which introduces a novel class of 6'-azacyclo substituted-9'-acyloxy esterified spirooxazine compounds. These molecules represent a critical evolution in optical chemical engineering, addressing long-standing challenges regarding thermal stability and fatigue resistance inherent in earlier generations of spiropyrans and spirooxazines. The core innovation lies in the strategic dual-substitution on the naphthalene ring system, where a nitrogen-containing heterocycle is introduced at the 6'-position and an acyloxy ester group is installed at the 9'-position. This specific structural modification fundamentally alters the electronic environment of the spiro carbon center, thereby modulating the kinetics of the ring-opening and ring-closing processes that define photochromism. For R&D directors and procurement specialists in the optical materials sector, understanding the synthesis and properties of these compounds is essential for developing next-generation high-density storage media and security inks.
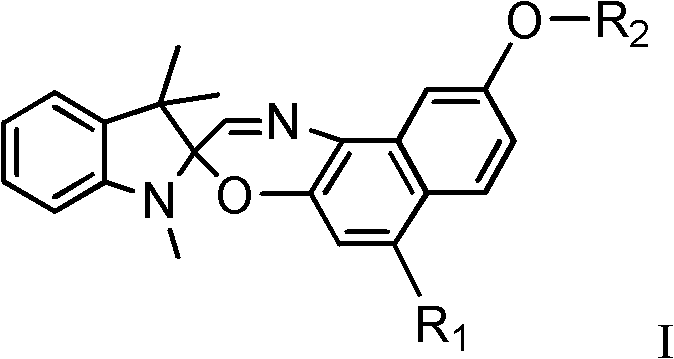
The general structure of these compounds, as depicted in the patent, allows for extensive tunability through the selection of R1 and R2 groups. R1 can be varied from morpholinyl to indolinyl derivatives, while R2 offers a range of acyl groups including methacryloyl and various substituted benzoyl moieties. This modularity provides a robust platform for creating a library of materials tailored to specific absorption wavelengths and solubility profiles required by different polymer matrices. The ability to fine-tune these parameters without compromising the core photochromic efficiency makes this technology highly attractive for commercial scale-up of complex electronic chemicals. Furthermore, the synthesis pathway described avoids the use of exotic transition metal catalysts, relying instead on classical organic transformations that are well-understood and easily scalable in a standard fine chemical manufacturing facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional spirooxazine derivatives, while superior to spiropyrans in terms of light fatigue resistance, often suffer from insufficient thermal stability and rapid thermal bleaching rates that limit their utility in certain optical recording applications. Conventional synthesis methods typically focus on modifying the indoline portion of the molecule or simple substitutions on the naphthalene ring, which yield diminishing returns in performance enhancement. Many existing commercial photochromic materials exhibit fading times that are too short for applications requiring persistent color states, such as specific types of holographic memory or slow-response optical switches. Additionally, the purification of conventional spirooxazines can be challenging due to the formation of closely related isomeric byproducts during the cyclization step, leading to lower overall yields and higher production costs. These limitations create a bottleneck for supply chain heads who require consistent, high-purity batches of materials that meet stringent specifications for electronic grade applications.
The Novel Approach
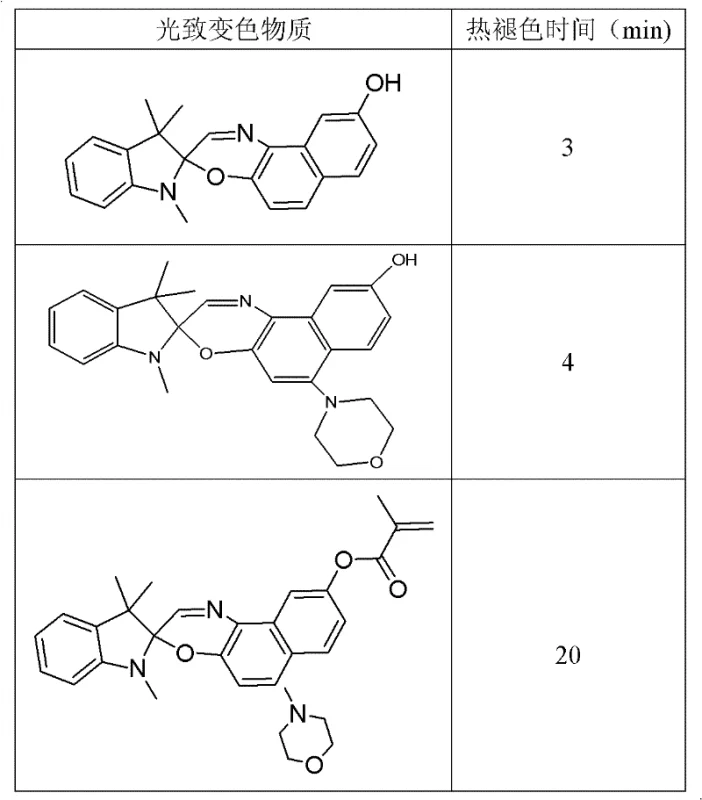
The approach detailed in CN102363620A overcomes these deficiencies by introducing bulky and electron-donating groups at specific positions that sterically and electronically stabilize the colored merocyanine form of the molecule. By incorporating a nitrogen heterocycle at the 6'-position, the electron density on the naphthalene ring is increased, which influences the energy barrier for the thermal reversion process. Simultaneously, the esterification at the 9'-position adds steric bulk that hinders the rotation necessary for the ring-closing reaction, effectively extending the lifetime of the colored state. Experimental data provided in the patent demonstrates that compounds synthesized via this method exhibit fading times significantly longer than unmodified analogs, with some derivatives maintaining their colored state for over 20 minutes under ambient conditions. This dramatic improvement in thermal stability translates directly to enhanced performance in end-use applications, making these materials a superior choice for reliable photochromic material supplier networks aiming to capture the high-end optical market.
Mechanistic Insights into Spirooxazine Cyclization and Esterification
The synthesis mechanism involves a sophisticated sequence of electrophilic aromatic substitution followed by nucleophilic addition and elimination reactions. The process begins with the nitrosation of 2,7-dihydroxynaphthalene, where the nitrosonium ion generated from sodium nitrite attacks the activated aromatic ring. This step is critical as it installs the nitrogen functionality required for the subsequent formation of the oxazine ring. The reaction conditions must be tightly controlled at low temperatures, typically between -5°C and 0°C, to prevent over-nitration or oxidation of the sensitive phenolic groups. Following isolation, the nitroso intermediate undergoes a condensation reaction with 1,3,3-trimethyl-2-methyleneindoline. This cyclization step is the heart of the spirooxazine formation, where the nucleophilic nitrogen of the indoline attacks the electrophilic carbon of the nitroso group, followed by intramolecular closure to form the oxazine ring. The presence of the nitrogen heterocycle substituent during this step requires careful solvent selection, such as trichloroethylene or methanol, to ensure solubility and drive the equilibrium towards the desired spiro product.
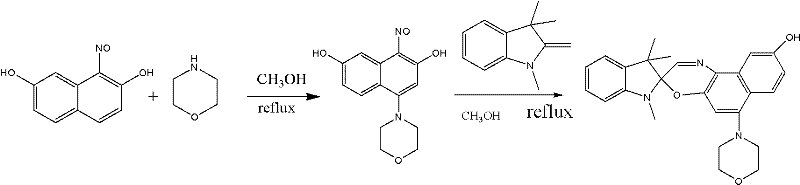
The final stage of the synthesis involves the esterification of the phenolic hydroxyl group at the 9'-position. This transformation is achieved using the corresponding acid chloride in the presence of a base like triethylamine and a solvent system comprising benzene and dioxane. The mechanism proceeds via a nucleophilic acyl substitution where the phenoxide ion attacks the carbonyl carbon of the acid chloride. The use of mild reaction conditions, often at room temperature or slightly elevated temperatures, ensures that the sensitive spiro C-O bond remains intact and does not undergo premature photolytic cleavage. This step is crucial for imparting the desired thermal stability, as the nature of the acyl group directly influences the steric environment around the spiro center. From a process chemistry perspective, this esterification is highly efficient and generates minimal waste, primarily consisting of triethylamine hydrochloride salt which can be easily filtered off. The robustness of this final step allows for the incorporation of a wide variety of functional groups, enabling the customization of the material's compatibility with different polymer hosts used in optical lenses and coatings.
How to Synthesize 6'-Azacyclo Substituted Spirooxazine Efficiently
The synthesis of these advanced photochromic compounds follows a logical three-step progression that balances yield optimization with operational simplicity. The process starts with the preparation of the nitroso-naphthalene intermediate, followed by the construction of the spirocyclic core, and concludes with the functionalization of the phenolic oxygen. Each step has been optimized in the patent examples to demonstrate feasibility on a laboratory scale, providing a clear roadmap for process engineers looking to adapt this chemistry for larger batch sizes. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions required to achieve high purity and consistent performance characteristics.
- Synthesize the intermediate 1-nitroso-2,7-dihydroxynaphthol by reacting 2,7-dihydroxynaphthalene with sodium nitrite under acidic conditions at low temperature.
- Perform the spiro-cyclization by condensing the nitroso intermediate with 1,3,3-trimethyl-2-methyleneindoline and a nitrogen heterocycle in a solvent like trichloroethylene under reflux.
- Complete the synthesis by esterifying the hydroxy-spirooxazine intermediate with the corresponding acid chloride using triethylamine and dioxane as catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers distinct strategic advantages over sourcing traditional photochromic materials. The primary benefit stems from the use of readily available starting materials such as 2,7-dihydroxynaphthalene and common acid chlorides, which reduces dependency on specialized or scarce reagents. This accessibility translates into a more resilient supply chain with reduced risk of disruption due to raw material shortages. Furthermore, the synthetic pathway eliminates the need for expensive transition metal catalysts or complex purification techniques like preparative HPLC, relying instead on standard crystallization and column chromatography methods that are cost-effective at scale. The operational simplicity of the esterification step, which can be conducted at room temperature, also contributes to lower energy consumption during manufacturing, aligning with sustainability goals and reducing overall utility costs.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of commodity chemicals for the acylating agents significantly lowers the direct material costs associated with production. By avoiding complex catalytic cycles that require rigorous removal of metal residues to meet electronic grade standards, the downstream processing costs are drastically simplified. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, thereby improving the overall mass balance and reducing the volume of solvent waste that requires treatment. These factors combine to create a manufacturing process that is inherently more economical, allowing for competitive pricing strategies in the global optical materials market without sacrificing quality or performance metrics.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates ensures that production can be scheduled flexibly without the degradation issues often associated with reactive nitroso compounds stored for long periods. The modular nature of the synthesis, where different R1 and R2 groups can be swapped in the final steps, allows manufacturers to respond quickly to changing customer demands for specific absorption profiles. This agility is crucial for maintaining service levels in industries like ophthalmics and security printing where trends can shift rapidly. Additionally, the robustness of the chemical bonds formed in the final product ensures a long shelf life for the finished goods, reducing inventory write-offs and enhancing the reliability of supply to downstream formulators and integrators.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the use of common organic solvents and ambient temperature steps, are highly amenable to scale-up from kilogram to multi-ton production volumes. The process avoids the generation of hazardous heavy metal waste streams, simplifying environmental compliance and wastewater treatment requirements. The byproduct of the esterification step, triethylamine hydrochloride, is a benign salt that can be easily managed or potentially recycled. This clean profile supports the growing demand for green chemistry practices in the fine chemical industry, making the technology attractive for facilities operating under strict environmental regulations while ensuring consistent commercial scale-up of complex photochromic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these novel spirooxazine derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the material's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating these compounds into new product lines or optimizing existing formulations for better durability and color intensity.
Q: What are the key structural features of this novel spirooxazine?
A: The compound features a unique dual-substitution pattern with a nitrogen heterocycle at the 6'-position of the naphthalene ring and an acyloxy ester group at the 9'-position, significantly enhancing thermal stability and fatigue resistance compared to traditional spirooxazines.
Q: How does the thermal stability compare to existing materials?
A: Experimental data indicates that the fading time for these new compounds exceeds 18 minutes, demonstrating superior thermal stability and slower thermal reversion rates suitable for long-duration optical recording.
Q: What are the primary industrial applications for these compounds?
A: These materials are designed for high-density optical information storage, holographic anti-counterfeiting, nonlinear optical devices, and military camouflage due to their reversible color change and high fatigue resistance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirooxazine Photochromic Compounds Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring these advanced photochromic materials from the laboratory to the global market. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the identity, potency, and impurity profile of every batch. This commitment to quality assurance guarantees that our clients receive materials that consistently meet the high performance standards required for optical storage and security applications, minimizing the risk of batch-to-batch variability.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to our manufactured spirooxazine derivatives can optimize your bill of materials. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific application needs. Whether you require custom substitution patterns or large-volume supply agreements, our dedicated support team is ready to provide the technical backing and commercial flexibility necessary to drive your project forward successfully.
