Scalable Production of High-Purity Deuterated Aldehydes Using Novel Triazole Carbene Catalysts
The landscape of deuterated pharmaceutical development has shifted dramatically since the FDA approval of Austedo, creating an urgent demand for efficient, scalable methods to introduce deuterium isotopes into complex molecular scaffolds. Patent CN111039767B introduces a groundbreaking methodology for the preparation of deuterated aldehydes, utilizing a novel triazole carbene catalytic system that fundamentally alters the economic and technical feasibility of isotopic labeling. This innovation addresses the critical bottleneck in synthesizing deuterated building blocks by replacing traditional, costly transition metal catalysts with an organocatalytic approach that leverages deuterated methanol as both solvent and deuterium source. For R&D directors and process chemists, this represents a paradigm shift towards greener, more atom-economical synthesis routes that maintain exceptionally high deuteration rates of up to 98% while drastically simplifying the purification workflow. The technology is particularly relevant for the production of high-purity pharmaceutical intermediates where isotopic purity is directly correlated with metabolic stability and pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated aldehydes has been plagued by significant operational inefficiencies and economic burdens associated with traditional stoichiometric reductions or transition metal catalysis. Conventional routes often rely on the reduction of esters using expensive deuterated lithium aluminum hydride followed by re-oxidation, a multi-step sequence that inherently suffers from low atom economy and generates substantial chemical waste. Alternatively, the Rosenmund reduction utilizes palladium catalysts under deuterium gas atmospheres, which introduces severe safety hazards related to high-pressure gas handling and requires rigorous removal of toxic palladium residues to meet pharmaceutical standards. Furthermore, existing transition metal-catalyzed C-H activation methods, while regioselective, frequently depend on scarce and volatile precious metals such as iridium or ruthenium complexes. These metal-dependent processes not only inflate the raw material costs exponentially but also create complex downstream processing challenges, as trace metal contamination can compromise the safety profile of the final active pharmaceutical ingredient, necessitating additional scavenging steps that reduce overall yield and extend production lead times.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111039767B employs a metal-free hydrogen-deuterium exchange mechanism driven by triazole carbene catalysts. This approach utilizes readily available deuterated methanol as the deuterium donor, effectively bypassing the need for hazardous deuterium gas or stoichiometric deuterated reducing agents. The core innovation lies in the use of specific triazole salt precursors, which generate active carbene species in situ under basic conditions to activate the aldehyde substrate for isotopic exchange. This strategy not only streamlines the synthetic route into a single-pot operation but also ensures that the reaction environment remains free from heavy metal contaminants. By eliminating the reliance on noble metals, the process achieves a substantial reduction in manufacturing costs and environmental impact, aligning perfectly with modern green chemistry principles. The versatility of this system is further demonstrated by its broad substrate scope, successfully accommodating aryl, alkyl, and alkenyl aldehydes with moderate to excellent deuteration efficiency, thereby offering a universal solution for diverse chemical architectures.
Mechanistic Insights into Triazole Carbene-Catalyzed Hydrogen-Deuterium Exchange
The efficacy of this deuteration protocol is rooted in the unique electronic and steric properties of the triazole carbene catalyst, which facilitates the activation of the aldehyde C-H bond through a non-metallic pathway. The catalyst precursors, depicted in the structural formulas below, feature bulky substituents such as 2,6-diisopropylphenyl (Dipp) or 2,4,6-trimethylphenyl (Mes) groups that provide essential steric protection to the carbene center, enhancing its stability and longevity under reflux conditions. Upon treatment with a base such as potassium tert-butoxide or DBU, the triazole salt undergoes deprotonation to generate the nucleophilic carbene species, which then interacts with the aldehyde substrate to form a transient zwitterionic intermediate. This activation lowers the energy barrier for proton abstraction at the formyl position, allowing for rapid exchange with the deuterium atoms supplied by the deuterated methanol solvent. The catalytic cycle is sustained by the continuous regeneration of the active carbene species, ensuring that only catalytic amounts of the precursor are required to drive the reaction to completion.
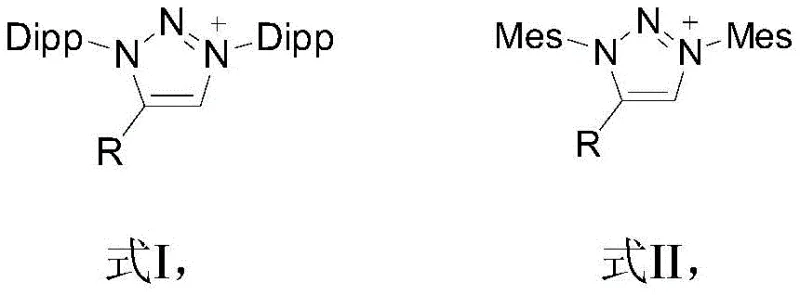
From an impurity control perspective, the metal-free nature of this mechanism offers distinct advantages for the production of high-purity pharmaceutical intermediates. Traditional transition metal catalysis often leaves behind trace amounts of iridium, ruthenium, or palladium, which are strictly regulated under ICH Q3D guidelines due to their toxicity. The triazole carbene system completely circumvents this issue, as the organic catalyst and its byproducts can be easily separated from the product via standard aqueous workup or silica gel chromatography. Furthermore, the mild reaction conditions minimize the formation of side products such as over-reduced alcohols or oxidized carboxylic acids, which are common pitfalls in stoichiometric reduction methods. The high selectivity of the carbene activation ensures that the deuteration occurs specifically at the aldehyde position without affecting other sensitive functional groups on the substrate, preserving the integrity of complex molecular scaffolds required for advanced drug synthesis.
How to Synthesize Deuterated Aldehydes Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, requiring standard laboratory equipment and avoiding the need for specialized high-pressure reactors or glovebox exclusivity beyond initial setup. The process begins with the precise weighing of the aldehyde substrate and the triazole carbene salt precursor, followed by the addition of a stoichiometric amount of base to initiate carbene generation. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the target aldehyde substrate, triazole carbene salt precursor (such as Formula I or II), and a strong base like potassium tert-butoxide in a sealed vessel under inert atmosphere.
- Add deuterated methanol (CD3OD) as the deuterium source and solvent, ensuring a molar ratio that favors excess deuterium donor to drive the equilibrium towards full deuteration.
- Heat the reaction mixture under reflux conditions for 9 to 24 hours, then cool, remove solvent, and purify the resulting deuterated aldehyde via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this triazole carbene-mediated deuteration technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of precious metal catalysts removes a major source of price volatility and supply risk, as the market for iridium and ruthenium is often subject to geopolitical constraints and fluctuating demand from other industries. By shifting to an organocatalytic model based on abundant nitrogen heterocycles and common bases, manufacturers can secure a more stable and predictable cost of goods sold (COGS). Additionally, the simplified workflow reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media, further driving down operational expenditures. The ability to use deuterated methanol, a commodity isotopic reagent, instead of custom-synthesized deuterium gas or complex hydrides, streamlines the procurement process and reduces inventory complexity.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of expensive noble metal catalysts, which traditionally account for a significant portion of raw material expenses in deuteration reactions. Without the need for iridium or palladium complexes, the direct material costs are drastically lowered, and the associated costs of metal recovery and waste disposal are virtually eliminated. Furthermore, the high atom economy of the exchange reaction means that less deuterium source is wasted, maximizing the utility of every mole of deuterated methanol purchased. This efficiency gain allows for competitive pricing strategies when supplying deuterated intermediates to downstream pharmaceutical clients, enhancing margin potential without compromising quality.
- Enhanced Supply Chain Reliability: Relying on organocatalysts derived from readily available chemical feedstocks significantly mitigates the risk of supply chain disruptions caused by rare metal shortages. The precursors for the triazole salts can be synthesized from bulk chemicals with established global supply networks, ensuring consistent availability even during market fluctuations. Moreover, the robustness of the reaction conditions, which tolerate standard reflux temperatures and do not require cryogenic cooling or high-pressure infrastructure, simplifies logistics and storage requirements. This operational resilience ensures that production schedules can be maintained with high predictability, reducing lead times for high-purity pharmaceutical intermediates and enabling just-in-time delivery models for key clients.
- Scalability and Environmental Compliance: The transition from batch to commercial scale is facilitated by the inherent safety and simplicity of the metal-free protocol, which avoids the hazards associated with pyrophoric reagents or high-pressure hydrogenation. Scaling up this process does not require exponential increases in safety engineering controls, making it easier to move from pilot plant to multi-ton production facilities. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly stringent global environmental regulations, reducing the burden of effluent treatment and hazardous waste management. This compliance advantage not only lowers regulatory overhead but also enhances the corporate sustainability profile, a critical factor for partnerships with major multinational pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented deuteration technology, based on the specific data and embodiments disclosed in the patent literature. These insights are intended to clarify the operational parameters and quality outcomes expected from this novel synthetic route.
Q: What are the advantages of using triazole carbene over transition metal catalysts for deuteration?
A: Triazole carbene catalysts eliminate the need for expensive and toxic precious metals like iridium or ruthenium. This metal-free approach significantly reduces raw material costs, simplifies downstream purification by removing heavy metal clearance steps, and ensures the final product meets stringent regulatory limits for elemental impurities in pharmaceutical applications.
Q: What is the achievable deuteration rate with this method?
A: According to patent CN111039767B, this method can achieve deuteration rates as high as 98%. The process is effective across a broad range of substrates, including aryl, alkyl, and alkenyl aldehydes, providing consistent isotopic labeling efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes simple reflux conditions and commercially available reagents like deuterated methanol and common bases. The absence of sensitive transition metal catalysts and the robustness of the organocatalytic system make it highly amenable to commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the triazole carbene catalytic system described in CN111039767B for advancing the synthesis of next-generation deuterated therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying isotopic purity and stringent purity specifications, guaranteeing that every batch of deuterated aldehyde meets the exacting standards required for clinical and commercial drug manufacturing. We are committed to leveraging this metal-free technology to deliver superior value to our partners through enhanced efficiency and quality.
We invite you to engage with our technical procurement team to discuss how this advanced deuteration methodology can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this organocatalytic route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless path from discovery to commercial supply.
