Scalable Visible Light Catalysis for High-Purity Deuterated Aldehyde Manufacturing
Scalable Visible Light Catalysis for High-Purity Deuterated Aldehyde Manufacturing
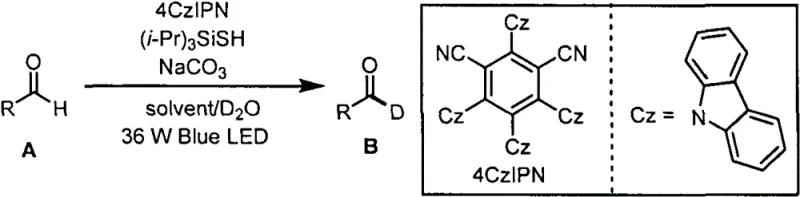
The landscape of isotopic labeling in pharmaceutical research is undergoing a significant transformation, driven by the urgent need for safer, more cost-effective, and environmentally benign synthetic methodologies. Patent CN114075108A introduces a groundbreaking approach to the preparation of deuterated aldehydes, utilizing a synergistic catalytic system powered by visible light and small organic molecules. This technology represents a paradigm shift away from traditional stoichiometric reductions or precious metal-catalyzed processes, offering a robust pathway for introducing deuterium atoms directly into the formyl C-H bond of aromatic and aliphatic aldehydes. By leveraging the unique photophysical properties of the organic photocatalyst 4CzIPN in conjunction with triisopropylsilyl mercaptan, this method achieves exceptional deuteration rates under mild conditions. For R&D directors and process chemists, this innovation provides a critical tool for enhancing metabolic stability in drug candidates while simplifying the supply chain for deuterated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated aldehydes has been fraught with significant operational challenges and economic inefficiencies that hinder large-scale adoption. Traditional routes often rely on the reduction of esters or amides using expensive and hazardous deuterated reagents such as lithium aluminum deuteride (LiAlD4) or Schwartz's reagent, followed by a subsequent oxidation step to regenerate the aldehyde functionality. These multi-step sequences not only suffer from poor atom economy but also introduce substantial safety risks due to the pyrophoric nature of the reagents involved. Furthermore, alternative methods employing transition metal catalysis, such as iridium or ruthenium complexes for hydrogen-isotope exchange (HIE), are often plagued by low regioselectivity, leading to unwanted deuteration on the aromatic ring rather than the desired formyl position. The reliance on scarce precious metals also creates supply chain vulnerabilities and necessitates rigorous downstream purification to meet stringent residual metal specifications required for pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN114075108A offers a streamlined, single-step solution that operates under exceptionally mild conditions. By employing a dual catalytic system consisting of the metal-free organic photocatalyst 4CzIPN and triisopropylsilyl mercaptan ((i-Pr)3SiSH), the process facilitates a direct hydrogen-deuterium exchange at the aldehyde position using inexpensive deuterated water (D2O) as the deuterium source. This approach eliminates the need for cryogenic temperatures or inert atmosphere gloveboxes, as the reaction proceeds efficiently at ambient temperatures (20-50°C) under standard blue LED irradiation. The use of common organic solvents like ethyl acetate or dichloromethane further enhances the practicality of the method, allowing for easy workup and purification via standard column chromatography. This technological leap not only improves the safety profile of the operation but also drastically reduces the cost of goods by replacing expensive deuterated reagents and precious metal catalysts with abundant, commodity chemicals.
Mechanistic Insights into 4CzIPN-Catalyzed H/D Exchange
The success of this deuteration strategy lies in the intricate interplay between the excited state dynamics of the 4CzIPN photocatalyst and the hydrogen atom transfer (HAT) capabilities of the silyl thiol co-catalyst. Upon irradiation with 470nm blue light, 4CzIPN enters a long-lived excited state capable of engaging in single electron transfer (SET) processes or energy transfer events. In this specific catalytic cycle, the excited photocatalyst likely activates the thiol species to generate a thiyl radical, which acts as a potent hydrogen abstractor. This thiyl radical selectively abstracts the weakly bonded hydrogen atom from the formyl group of the aldehyde substrate, generating an acyl radical intermediate. The presence of deuterated water and base (sodium carbonate) facilitates the rapid quenching of this acyl radical with a deuterium atom, effectively completing the H/D exchange cycle. The regeneration of the thiol catalyst ensures the turnover of the catalytic cycle, allowing for high efficiency with minimal catalyst loading (0.001-0.2 equivalents).
From an impurity control perspective, this radical-mediated mechanism offers superior regioselectivity compared to electrophilic aromatic substitution pathways often seen with acidic or basic conditions. The bond dissociation energy (BDE) of the formyl C-H bond is significantly lower than that of aromatic C-H bonds, ensuring that the thiyl radical preferentially targets the aldehyde proton. This intrinsic selectivity minimizes the formation of ring-deuterated byproducts, which are common contaminants in transition metal-catalyzed HIE reactions. Additionally, the mild neutral to slightly basic conditions provided by sodium carbonate prevent acid-catalyzed side reactions such as aldol condensation or acetal formation, which are prevalent risks when handling reactive aldehyde intermediates. The result is a clean reaction profile that yields high-purity deuterated aldehydes, simplifying the downstream purification burden and ensuring consistent quality for sensitive pharmaceutical applications.
How to Synthesize Deuterated Aldehydes Efficiently
The operational simplicity of this photochemical protocol makes it highly accessible for both laboratory-scale optimization and pilot-plant production. The procedure typically involves charging a reaction vessel with the aldehyde substrate, the 4CzIPN photocatalyst, the silane thiol co-catalyst, and sodium carbonate in a biphasic mixture of organic solvent and deuterated water. After a brief purge with argon to exclude oxygen, which can quench the excited state of the photocatalyst, the mixture is subjected to continuous stirring under 36W blue LED illumination. The reaction progress is monitored to ensure complete conversion, typically achieved within 36 to 48 hours at optimal temperatures. Following the reaction, the organic layer is separated, dried, and concentrated, with the final pure product isolated via flash column chromatography. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific substrate, please refer to the standardized synthesis steps provided below.
- Mix aldehyde substrate with photocatalyst 4CzIPN, triisopropylsilyl mercaptan, sodium carbonate, organic solvent, and deuterated water in a reaction flask.
- Purge the reaction mixture with argon gas for approximately 30 seconds to remove oxygen.
- Irradiate the mixture with 36W 470nm blue LED light at 20-50°C for 36-48 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible-light catalytic technology presents a compelling value proposition centered on cost stability and supply security. By transitioning away from precious metal catalysts like iridium and ruthenium, manufacturers can insulate their production costs from the volatile fluctuations of the commodities market associated with these rare earth elements. Furthermore, the substitution of specialized deuterated reducing agents with bulk deuterated water significantly lowers the raw material expenditure per kilogram of product. This shift not only reduces the direct cost of manufacturing but also mitigates the regulatory and logistical complexities associated with handling hazardous pyrophoric materials, thereby lowering insurance and compliance overheads. The robustness of the process ensures a reliable supply of critical deuterated intermediates, essential for maintaining continuity in drug development pipelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and stoichiometric deuterated reagents drives a substantial decrease in production costs. The use of catalytic amounts of an organic dye (4CzIPN) and a simple silane thiol, coupled with inexpensive D2O, creates a highly economical reagent profile. Additionally, the mild reaction conditions reduce energy consumption related to heating or cooling, contributing to overall operational efficiency and a lower carbon footprint for the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as sodium carbonate, ethyl acetate, and commercially available organic catalysts reduces dependency on single-source suppliers of exotic reagents. This diversification of the supply base enhances resilience against global supply chain disruptions. The scalability of the photochemical setup, which can be adapted from batch to flow chemistry systems, ensures that production capacity can be rapidly expanded to meet surging demand without the long lead times associated with sourcing specialized metal catalysts.
- Scalability and Environmental Compliance: The metal-free nature of this process simplifies waste stream management, as there is no need for complex heavy metal scavenging steps or disposal of toxic metal residues. This aligns perfectly with green chemistry principles and increasingly stringent environmental regulations. The ability to run the reaction at ambient pressure and moderate temperatures also reduces the engineering controls required for scale-up, facilitating a smoother transition from gram-scale discovery to multi-kilogram commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this deuteration technology. These insights are derived directly from the experimental data and scope limitations defined in the patent literature, providing a realistic overview of what process chemists can expect when adopting this methodology. Understanding these parameters is crucial for effective project planning and risk assessment in early-stage drug development.
Q: What are the optimal reaction conditions for aldehyde deuteration using this method?
A: The optimal conditions involve using ethyl acetate or dichloromethane as the solvent, a reaction temperature between 20-50°C, and irradiation with 36W 470nm blue light for 36-48 hours.
Q: Can this method be applied to complex drug molecules?
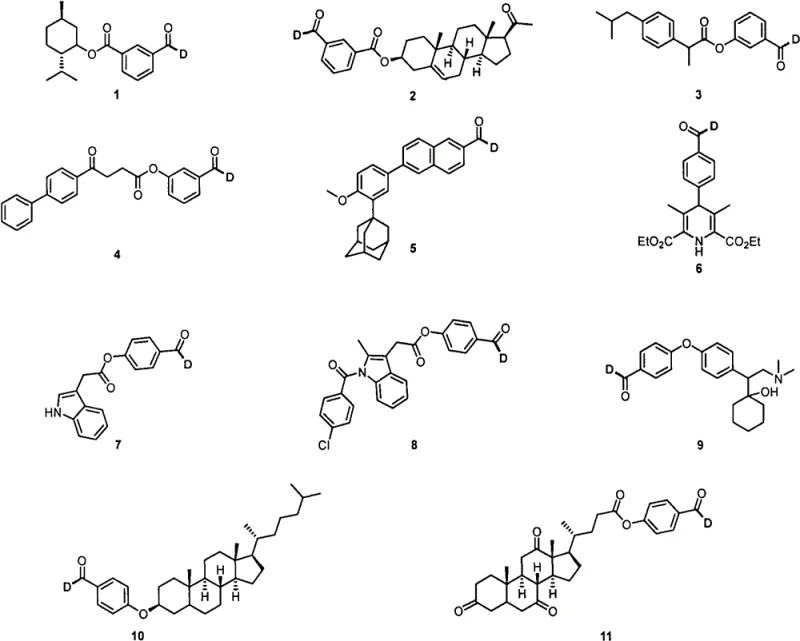
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully deuterating complex structures including menthol esters, ibuprofen derivatives, and adamantane-containing aldehydes with high regioselectivity.
Q: Is the use of expensive transition metals required for this deuteration?
A: No, this novel approach utilizes an organic photocatalyst (4CzIPN) and a silane-based co-catalyst, completely eliminating the need for costly iridium or ruthenium transition metals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality deuterated intermediates in modern drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of executing the precise 470nm irradiation protocols required for this novel synthesis. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of deuterated aldehyde meets the highest standards of isotopic enrichment and chemical purity, free from residual metal contaminants.
We invite you to collaborate with our technical team to evaluate the feasibility of this cost-effective route for your specific pipeline candidates. By leveraging our expertise in visible light catalysis, we can help you achieve significant process improvements and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project requirements. Let us help you secure a sustainable and economical supply of critical deuterated building blocks for your next breakthrough therapy.
