Advanced Synthesis of Asymmetric Cyanine Dyes for High-Performance DVD-R Recording Media
Advanced Synthesis of Asymmetric Cyanine Dyes for High-Performance DVD-R Recording Media
The rapid evolution of optical storage technology has necessitated the development of recording media capable of higher data densities and faster write speeds, specifically targeting the DVD-R format. Patent CN1148368C discloses a groundbreaking preparation method for asymmetric cyanine dyes that serve as the critical recording layer in these discs. Unlike traditional CD-R dyes which operate at 780nm, DVD-R systems require recording media with strong absorption in the 630-690nm range to match the shorter wavelength semiconductor lasers used in DVD drives. This patent introduces a novel synthetic route utilizing 1,1-dimethyl-2-formylmethylene-3-alkyl-1H-benzo[e]indole as a key intermediate, reacting directly with substituted 1-alkyl-2,3,3-trimethylindole salts. The resulting asymmetric trimethine cyanine dyes demonstrate exceptional optical properties, including a large extinction coefficient exceeding 10^5 M^-1 cm^-1 and high stability, making them ideal candidates for next-generation optical storage solutions.
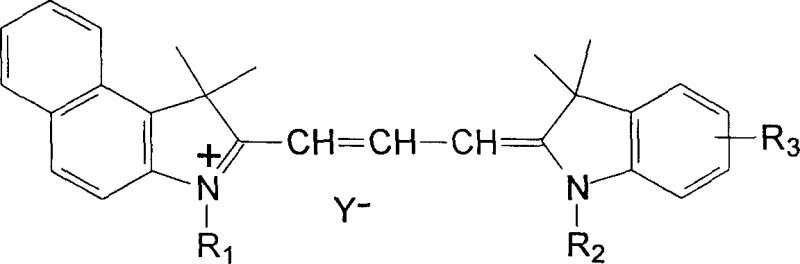
For procurement managers and supply chain directors seeking a reliable electronic chemical supplier, understanding the structural versatility of these compounds is paramount. The general formula allows for significant modulation of solubility and thermal stability by varying the alkyl chains (R1, R2) and substituents (R3) on the indole rings. The patent specifies that R1 and R2 can be linear or branched alkyl chains ranging from C1 to C20, while R3 can be hydrogen, methyl, methoxy, or halogen groups. This flexibility ensures that manufacturers can tailor the dye properties to specific spin-coating processes, particularly ensuring excellent solubility in organic solvents like alcohols. Such adaptability is crucial for maintaining consistent film quality during the high-speed manufacturing of recordable discs, thereby reducing waste and improving yield in the final assembly line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric cyanine dyes relied on a multi-step process involving the formation of a hemicyanine intermediate. As detailed in prior art such as Brit. Pat. 344,409 and US 4,847,385, this conventional approach reacts an indole quaternary ammonium salt with a condensing agent to generate the hemicyanine, which is subsequently reacted with another indole salt. This methodology suffers from inherent chemical inefficiencies, primarily the unavoidable formation of symmetric dye byproducts alongside the desired asymmetric target. These symmetric impurities possess similar physical properties to the target molecule, making purification extremely difficult and often requiring multiple, labor-intensive recrystallization steps. Consequently, the final purity often fails to meet the stringent requirements for high-quality DVD-R recording media, leading to lower reflectivity and potential data integrity issues in the finished optical discs.
The Novel Approach
The innovative method described in CN1148368C circumvents these historical bottlenecks by employing a direct condensation strategy. By utilizing 1,1-dimethyl-2-formylmethylene-3-alkyl-1H-benzo[e]indole as the starting material, the reaction proceeds without generating the problematic symmetric dye impurities associated with hemicyanine intermediates. Since the aldehyde intermediate itself does not generate dye byproducts during its own preparation, the reaction mixture remains chemically cleaner from the outset. This fundamental shift in synthetic strategy simplifies the downstream purification process drastically; high-purity dye (>99%) can be achieved through a single recrystallization step. For industrial partners, this translates to a streamlined workflow that reduces solvent usage, minimizes processing time, and significantly lowers the cost of goods sold (COGS) while ensuring the high optical performance required for commercial DVD-R production.
Mechanistic Insights into Condensation Reaction and Purification
The core of this technological advancement lies in the efficient condensation reaction between the formylmethylene indole derivative and the quaternary indole salt. The reaction is typically conducted in acetic anhydride, which serves a dual purpose as both the solvent and a dehydrating agent to drive the equilibrium towards the formation of the polymethine chain. The reaction conditions are remarkably mild yet effective, operating at temperatures ranging from 50°C to the reflux temperature of acetic anhydride (approximately 105-110°C). The molar ratio of the reactants is maintained at a stoichiometric 1:1, ensuring maximal atom economy and minimizing the presence of unreacted starting materials that could complicate purification. This precise control over reaction parameters allows for the consistent production of the trimethine bridge (-CH=CH-CH=) that connects the two heterocyclic nuclei, which is responsible for the dye's specific absorption characteristics in the red spectrum.
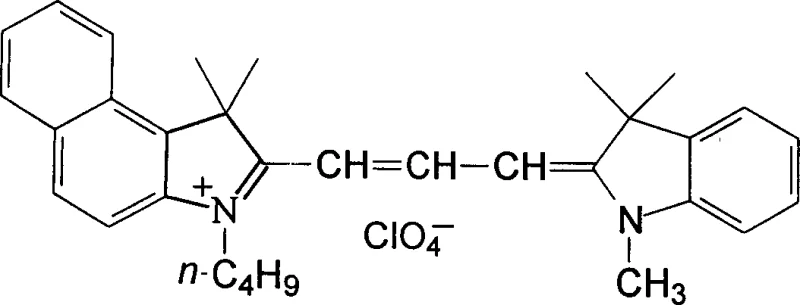
Following the reaction, the workup procedure is designed for scalability and environmental compliance. The reaction mixture is poured into water, causing the hydrophobic dye to precipitate as a loose red solid, which is then filtered and washed. The elimination of symmetric byproducts means that the subsequent recrystallization step—using solvents such as methanol, ethanol, or isopropanol—is highly efficient. In contrast to older methods where chromatography or repeated crystallizations were necessary to separate symmetric impurities, this method achieves pharmaceutical-grade purity levels suitable for electronic applications in a single pass. This mechanistic simplicity not only enhances the purity profile but also ensures that the batch-to-batch variability is minimized, a critical factor for R&D directors validating processes for commercial scale-up of complex electronic chemicals.
How to Synthesize Asymmetric Cyanine Dye Efficiently
To implement this synthesis in a pilot or production environment, operators must adhere to strict control over the addition rates and temperature profiles to maximize yield and crystal quality. The process begins with the precise weighing of the 1,1-dimethyl-2-formylmethylene-3-alkyl-1H-benzo[e]indole and the substituted indole salt, followed by their suspension in acetic anhydride. Heating is applied gradually to reach the target range of 105-110°C, where the mixture is stirred for approximately one hour to ensure complete conversion. Upon completion, the quenching step involves pouring the hot reaction mass into agitated water, inducing rapid precipitation. The detailed standardized synthesis steps, including specific workup protocols and recrystallization parameters for various derivatives, are outlined below to guide technical teams in replicating these high-yield results.
- React 1,1-dimethyl-2-formylmethylene-3-alkyl-1H-benzo[e]indole with a substituted 1-alkyl-2,3,3-trimethylindole salt in acetic anhydride solvent.
- Heat the reaction mixture to a temperature between 50°C and the reflux temperature of acetic anhydride (approx. 105-110°C) for 5 minutes to 5 hours.
- Pour the reaction liquid into water to precipitate the solid, filter, wash, and purify the crude product via a single recrystallization step using alcohol solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthesis route offers substantial benefits for cost reduction in display material manufacturing. The primary driver of value is the drastic simplification of the purification train. By eliminating the need for complex separation techniques to remove symmetric dye impurities, manufacturers can significantly reduce the consumption of high-purity solvents and the energy costs associated with extended heating and cooling cycles. Furthermore, the high yield reported in the patent examples, consistently exceeding 85% and reaching up to 92.5% in optimized cases, ensures that raw material utilization is maximized. This efficiency directly impacts the bottom line, allowing procurement managers to negotiate better margins or pass savings on to downstream disc manufacturers, thereby enhancing competitiveness in the global optical media market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts or complex condensing agents required in older hemicyanine routes results in a leaner bill of materials. Additionally, the ability to achieve >99% purity through a single recrystallization step reduces the operational expenditure related to waste treatment and solvent recovery. The process relies on commodity chemicals like acetic anhydride and simple indole salts, which are readily available from bulk chemical suppliers, ensuring stable pricing and reducing the risk of supply chain disruptions caused by specialty reagent shortages.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes to a more resilient supply chain for high-purity electronic chemicals. Because the reaction tolerances are relatively broad (5 minutes to 5 hours reaction time, 50°C to reflux), the process is less susceptible to minor fluctuations in plant conditions, leading to higher first-pass success rates. This reliability reduces lead time for high-purity asymmetric cyanine dyes, enabling suppliers to respond more quickly to fluctuating demand from the consumer electronics sector. The use of stable intermediates also allows for the stocking of key precursors, further buffering against raw material volatility.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to metric tons is facilitated by the straightforward unit operations involved: stirring, heating, filtration, and crystallization. There are no exothermic hazards or toxic gas evolutions that would require specialized containment equipment, making it suitable for standard multipurpose chemical reactors. Moreover, the reduced solvent load and the absence of heavy metal contaminants simplify wastewater treatment, helping manufacturers meet increasingly stringent environmental regulations without incurring prohibitive compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric cyanine dyes. The answers are derived directly from the experimental data and technical specifications provided in patent CN1148368C, ensuring accuracy for R&D and procurement stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this material into existing DVD-R manufacturing lines or for developing new optical storage formulations that require precise wavelength tuning.
Q: Why is this new synthesis method superior to conventional hemicyanine routes?
A: Conventional methods involve forming a hemicyanine intermediate which often leads to the formation of symmetric dye byproducts that are difficult to remove. This novel direct condensation method uses a pure aldehyde intermediate, preventing symmetric dye formation and allowing high purity (>99%) via simple recrystallization.
Q: What are the critical optical properties of these dyes for DVD-R applications?
A: The synthesized asymmetric cyanine dyes exhibit a maximum absorption wavelength between 650nm and 700nm, matching the laser wavelength of DVD-R systems. They possess a large extinction coefficient (ε > 10^5 M^-1 cm^-1) and high reflectivity after film formation, ensuring reliable data recording.
Q: How does the purification process impact manufacturing costs?
A: The process eliminates the need for complex chromatographic separation or multiple recrystallizations required by older methods. Achieving high purity through a single recrystallization step significantly reduces solvent consumption, processing time, and overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Cyanine Dye Supplier
As the demand for high-performance optical recording media continues to evolve, partnering with an experienced chemical manufacturer is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC analysis and spectral characterization, to guarantee that every batch of asymmetric cyanine dye meets the exacting standards required for DVD-R applications. We understand the nuances of electronic chemical manufacturing and are committed to delivering products that enhance the reflectivity and stability of your final recording discs.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your production costs and improve product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D initiatives, ensuring a seamless transition to this superior dye technology for your next generation of optical storage solutions.
