Scalable Palladium-Catalyzed Carbonylation for High-Purity Dihydroquinolone Derivatives
Scalable Palladium-Catalyzed Carbonylation for High-Purity Dihydroquinolone Derivatives
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly the 2,3-dihydroquinolone scaffold, which serves as a critical backbone for numerous bioactive molecules ranging from analgesics to antitumor agents. As illustrated in the structural diversity of known bioactive compounds, the precise installation of substituents on this core is paramount for optimizing biological activity. Patent CN113735826A introduces a transformative preparation method for 3-benzylidene-2,3-dihydroquinolone compounds, leveraging a sophisticated palladium-catalyzed carbonylation strategy. This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing N-pyridinesulfonyl-o-iodoaniline and allenes as key building blocks, offering a pathway that is not only chemically elegant but also practically viable for large-scale operations. The significance of this development lies in its ability to bypass traditional limitations, providing a reliable pharmaceutical intermediate supplier with a tool to access complex molecular architectures efficiently.
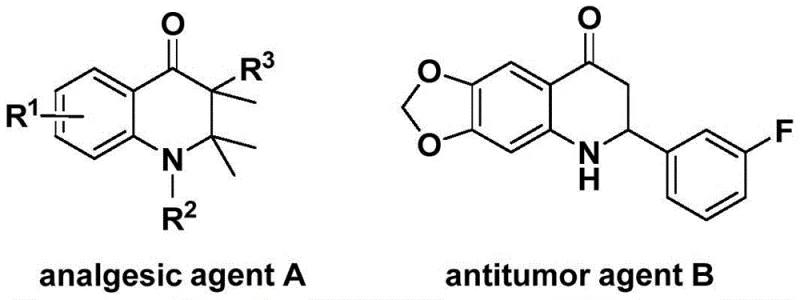
The strategic value of this patent extends beyond mere academic interest; it represents a tangible solution for cost reduction in API manufacturing where the quinolone motif is prevalent. By employing a solid carbon monoxide substitute instead of hazardous gaseous CO, the process inherently improves safety profiles while maintaining high reaction efficiency. For R&D directors focused on purity and impurity profiles, the method's high substrate compatibility ensures that diverse functional groups can be tolerated without compromising the integrity of the final product. This level of control is essential when developing candidates for clinical evaluation, where batch-to-batch consistency and chemical purity are non-negotiable standards. Consequently, this technology positions itself as a cornerstone for the commercial scale-up of complex pharmaceutical intermediates, bridging the gap between discovery chemistry and industrial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone derivatives has relied on methodologies that often suffer from significant drawbacks regarding operational complexity and safety. Traditional carbonylation reactions frequently necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks and requires specialized, expensive equipment that many standard synthesis laboratories lack. Furthermore, conventional routes may involve multi-step sequences with poor atom economy, leading to excessive waste generation and higher overall production costs. The reliance on harsh reaction conditions can also limit the scope of compatible substrates, particularly those containing sensitive functional groups that might degrade under extreme temperatures or pressures. These factors collectively create bottlenecks in the supply chain, extending lead times for high-purity pharmaceutical intermediates and increasing the financial burden on drug development projects. Additionally, the purification of products from such reactions can be cumbersome, often requiring extensive chromatographic separation to remove metal residues and side products.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed system that operates under remarkably mild and manageable conditions, fundamentally shifting the paradigm for synthesizing these valuable heterocycles. The reaction employs bis(acetylacetone)palladium as the catalyst and 1,3-bis(diphenylphosphino)propane (DPPP) as the ligand, facilitating a smooth cyclization process at temperatures between 80°C and 100°C. A critical innovation is the use of 1,3,5-trimesic acid phenol ester (TFBen) as a safe, solid carbon monoxide substitute, which releases CO in situ, thereby eliminating the need for high-pressure gas cylinders. This modification not only enhances safety but also simplifies the reactor setup, making the process accessible for broader adoption. The reaction scheme below highlights the convergence of N-pyridinesulfonyl-o-iodoaniline and various allenes to form the target 3-benzylidene-2,3-dihydroquinolone structure with high precision.
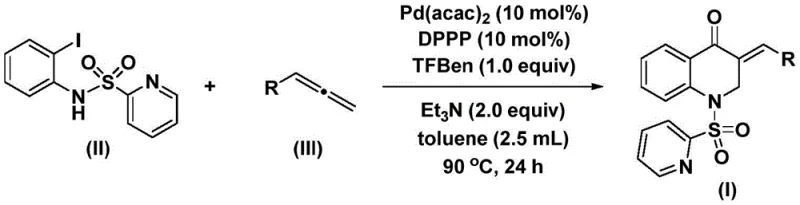
Furthermore, the novel approach demonstrates exceptional versatility, accommodating a wide range of allene substrates with different electronic and steric properties. Whether the allene bears electron-donating groups like methoxy and methyl or electron-withdrawing halogens, the system maintains high conversion rates and yields, often surpassing 80% and reaching up to 93% in optimized cases. This broad substrate scope is a testament to the robustness of the catalytic cycle, ensuring that medicinal chemists can rapidly generate libraries of analogs for structure-activity relationship studies. The simplicity of the post-treatment procedure, involving basic filtration and standard column chromatography, further underscores the practical advantages of this method over legacy techniques. By streamlining the synthesis workflow, this approach directly contributes to reducing lead time for high-purity pharmaceutical intermediates, allowing companies to accelerate their drug discovery timelines significantly.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific applications. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridinesulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is facilitated by the electron-rich DPPP ligand, which stabilizes the metal center and promotes the insertion process. Subsequently, carbon monoxide, released thermally from the TFBen additive, inserts into the aryl-palladium bond to form an acyl-palladium species. This migratory insertion is the key carbonylation step that introduces the ketone functionality essential for the quinolone core. The precision of this step ensures that the carbonyl group is installed regioselectively, minimizing the formation of regioisomeric impurities that could complicate downstream purification.
Following the CO insertion, the allene substrate coordinates to the palladium center and undergoes insertion into the acyl-palladium bond, forming a new alkyl-palladium intermediate. This step dictates the stereochemistry and substitution pattern of the exocyclic double bond in the final product. The cycle concludes with a reductive elimination step, which releases the 3-benzylidene-2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst to continue the cycle. The presence of triethylamine as an additive likely plays a role in neutralizing acidic byproducts and maintaining the basicity required for the catalytic turnover. The ability of this system to tolerate diverse substituents on the allene, as shown in the examples below, suggests that the steric bulk of the ligand and the electronic nature of the palladium center are well-balanced to accommodate various transition states.
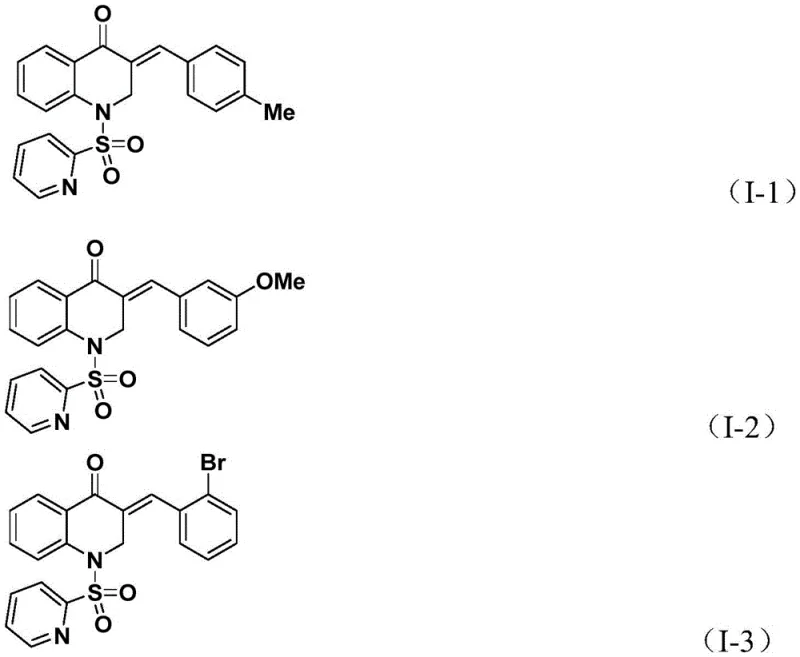
From an impurity control perspective, the mechanism offers inherent advantages. The use of a specific sulfonyl protecting group on the aniline nitrogen directs the cyclization pathway, preventing unwanted side reactions such as polymerization of the allene or homocoupling of the aryl iodide. The high yields observed across different substrates indicate that the reductive elimination step is fast and efficient, preventing the accumulation of stable palladium intermediates that could lead to catalyst deactivation. For quality control teams, this means a cleaner reaction profile with fewer unknown impurities, simplifying the analytical validation process. The mechanistic clarity provided by this patent allows process chemists to fine-tune parameters such as temperature and catalyst loading to maximize throughput while maintaining stringent purity specifications required for GMP manufacturing environments.
How to Synthesize 3-Benzylidene-2,3-dihydroquinolone Efficiently
The execution of this synthesis protocol is designed to be straightforward, enabling seamless translation from bench-scale experiments to pilot plant operations. The procedure begins with the precise weighing of the palladium catalyst, ligand, and solid CO source, ensuring the correct stoichiometric ratios are maintained to drive the reaction to completion. The choice of toluene as the solvent is critical, as it provides the optimal solubility for both the organic substrates and the organometallic intermediates while withstanding the reaction temperature of 90°C. Operators should ensure that the reaction vessel is properly sealed to prevent solvent loss, although the use of a solid CO source negates the need for high-pressure autoclaves. Detailed standardized synthesis steps see the guide below for specific operational parameters.
- Combine Pd(acac)2 catalyst, DPPP ligand, TFBen CO-source, triethylamine, N-pyridinesulfonyl-o-iodoaniline, and the specific allene substrate in toluene solvent within a Schlenk tube.
- Heat the reaction mixture to 90°C and maintain stirring for 24 to 48 hours to ensure complete conversion of the starting materials into the target heterocycle.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the high-purity 3-benzylidene-2,3-dihydroquinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend far beyond the laboratory. The primary advantage lies in the substantial cost savings achieved through the use of readily available and inexpensive starting materials. N-pyridinesulfonyl-o-iodoaniline and various allenes are commercially accessible or can be synthesized in few steps from commodity chemicals, reducing the raw material cost burden significantly. Moreover, the elimination of high-pressure carbon monoxide gas removes the need for specialized gas handling infrastructure and safety certifications, which translates to lower capital expenditure and reduced operational overheads. This streamlined approach allows manufacturers to allocate resources more efficiently, focusing on capacity expansion rather than complex safety compliance measures associated with toxic gases.
- Cost Reduction in Manufacturing: The economic viability of this process is further enhanced by the high catalytic efficiency and the ability to run reactions at relatively low catalyst loadings without sacrificing yield. By avoiding expensive and hazardous reagents, the overall cost of goods sold (COGS) for the final intermediate is drastically reduced. The simplified workup procedure, which involves basic filtration and standard purification techniques, minimizes solvent consumption and waste disposal costs. This lean manufacturing approach ensures that the production of high-purity pharmaceutical intermediates remains financially sustainable even when scaling to multi-ton quantities. Additionally, the robustness of the reaction reduces the risk of batch failures, thereby protecting profit margins and ensuring consistent supply.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on stable, solid reagents like TFBen and commercially available palladium complexes ensures a steady flow of materials without the volatility associated with gas supply chains. The short reaction times of 24 to 48 hours allow for rapid turnover of production batches, enabling manufacturers to respond quickly to fluctuating market demands. This agility is crucial in the pharmaceutical sector, where delays in intermediate supply can stall entire drug development programs. The method's compatibility with standard glass-lined reactors means that existing manufacturing facilities can be utilized without major retrofitting, further securing supply continuity. Consequently, partners can rely on a dependable source of critical building blocks, mitigating the risk of production bottlenecks.
- Scalability and Environmental Compliance: The environmental footprint of this process is notably smaller compared to traditional methods, aligning with modern green chemistry principles and regulatory requirements. The use of toluene, a common industrial solvent, facilitates easy recovery and recycling, minimizing volatile organic compound (VOC) emissions. The absence of heavy metal waste streams associated with stoichiometric reagents simplifies wastewater treatment and disposal protocols. As the process scales from grams to kilograms and eventually to tons, these environmental advantages become increasingly significant, reducing the liability and cost associated with environmental compliance. The ability to scale up smoothly without encountering exothermic runaway risks or pressure hazards makes this technology an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for potential adopters. Understanding these nuances helps stakeholders make informed decisions about integrating this methodology into their existing production workflows. The answers reflect the practical realities of running this chemistry at scale, covering aspects from catalyst selection to product isolation.
Q: What is the advantage of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-trimethoxybenzene phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents on the aryl ring of the allene, including methyl, tert-butyl, methoxy, and halogens like fluorine, chlorine, and bromine, with yields often exceeding 80%.
Q: Can this process be scaled for commercial API intermediate production?
A: Yes, the protocol utilizes commercially available reagents and standard organic solvents like toluene, and the simple post-treatment involving filtration and chromatography makes it highly suitable for scaling from gram-level laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2,3-dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has thoroughly evaluated the technology described in CN113735826A and is fully equipped to leverage this palladium-catalyzed carbonylation route for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs protocols to guarantee the highest quality intermediates.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to discuss your specific project requirements,索取 specific COA data, and obtain comprehensive route feasibility assessments. Let us help you secure a competitive edge in the market with our reliable supply of high-performance pharmaceutical intermediates.
