Advanced Catalytic Synthesis of 4-Hydroxyindole for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry constantly seeks robust synthetic routes that balance efficiency, safety, and economic viability, particularly for critical building blocks like 4-hydroxyindole. A recent technological breakthrough documented in patent CN113321609A introduces a transformative approach to synthesizing this key intermediate, which is essential for the production of beta-receptor blocking drugs such as pindolol. Unlike traditional methods that rely on hazardous high-pressure conditions and expensive chlorinated precursors, this novel protocol utilizes a dehydrogenative aromatization strategy starting from 1,3-cyclohexanedione and 2-aminoethanol. For R&D directors and procurement managers alike, this shift represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. By fundamentally altering the reaction mechanism to avoid pressurized ammonia and corrosive byproducts, the process not only enhances operational safety but also streamlines the purification workflow. This report provides a deep technical analysis of this innovation, evaluating its potential to redefine cost reduction in pharmaceutical intermediate manufacturing while ensuring the stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4-hydroxyindole has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. The conventional synthetic route typically involves the reaction of 1,3-cyclohexanedione with 2-chloroacetaldehyde to form a furan intermediate, which subsequently requires treatment with ammonia water in a closed system under high temperature and pressure. This reliance on pressurized ammonia creates severe engineering challenges, as ammonia possesses a strong complexing ability with various metals, leading to serious corrosion of reaction vessels and associated piping infrastructure. Furthermore, the use of 2-chloroacetaldehyde introduces substantial raw material costs and safety hazards due to its toxicity and reactivity. The multi-step nature of this traditional pathway, combined with the need for specialized pressure-rated equipment, results in extended lead times and increased capital expenditure, making it a less attractive option for modern, agile supply chains seeking reliable pharmaceutical intermediate supplier partnerships.
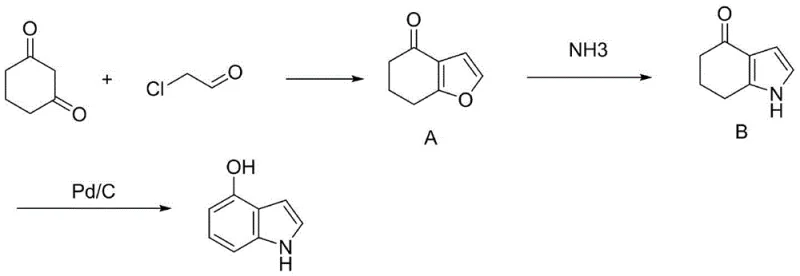
The Novel Approach
In stark contrast to the legacy methods, the innovative pathway disclosed in the patent data offers a streamlined, two-step synthesis that fundamentally resolves the safety and cost issues associated with the old process. By substituting 2-chloroacetaldehyde with 2-aminoethanol, the new method utilizes a much more economical and readily available starting material that reacts with 1,3-cyclohexanedione to form an enamine intermediate. This condensation reaction produces water as the only byproduct, eliminating the generation of corrosive hydrochloric acid or salt waste typical of chlorinated precursors. The subsequent conversion of the enamine to 4-hydroxyindole is achieved through a catalytic dehydrogenation process that operates without the need for high-pressure vessels. This shift not only drastically simplifies the equipment requirements but also improves the overall atom economy of the reaction. For procurement teams, this translates to a more stable cost structure and reduced dependency on specialized, high-maintenance reactor systems, thereby enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into Dehydrogenative Aromatization
The core of this technological advancement lies in the efficient formation and subsequent aromatization of the enamine intermediate, a process that demands precise catalytic control to ensure high yield and purity. The reaction begins with the condensation of 1,3-cyclohexanedione and 2-aminoethanol, typically facilitated by an acid catalyst such as p-toluenesulfonic acid in a solvent like toluene. This step forms a stable enamine structure that serves as the precursor for the ring-closing aromatization. The critical transformation occurs in the second step, where the enamine undergoes cyclization and dehydrogenation in the presence of a supported metal catalyst. The patent specifies the use of catalysts such as Pd/C, Pt/C, or even Raney Nickel, which facilitate the removal of hydrogen atoms to establish the aromatic indole system. This mechanism avoids the harsh conditions of ammonia-based cyclization, instead relying on the intrinsic reactivity of the enamine double bond and the catalytic surface to drive the formation of the heterocyclic ring. Understanding this mechanism is vital for R&D directors aiming to replicate the process, as it highlights the importance of catalyst selection and temperature control in achieving the desired 56% to quantitative yields observed in the experimental data.
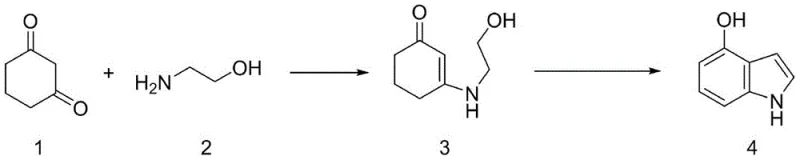
Controlling the impurity profile in this synthesis is paramount, especially given the intended application in the production of active pharmaceutical ingredients like pindolol. The new method inherently reduces the risk of metal contamination and chlorinated byproducts, which are common pain points in the conventional route. Since the reaction byproduct in the second step is hydrogen gas, there is no accumulation of salt waste that could complicate downstream purification or damage equipment. The use of solvents like trimethylbenzene allows for high-temperature reflux without pressure buildup, ensuring that side reactions are minimized. For quality assurance teams, this means that the crude product requires less aggressive purification steps to meet stringent purity specifications. The elimination of corrosive ammonia also means that the final product is less likely to contain trace metal impurities derived from equipment corrosion. This clean reaction profile supports the production of high-purity pharmaceutical intermediates that can seamlessly integrate into downstream API synthesis without requiring extensive additional cleaning or validation steps.
How to Synthesize 4-Hydroxyindole Efficiently
Implementing this synthesis route in a pilot or production setting requires careful attention to the specific reaction parameters outlined in the patent data to ensure reproducibility and safety. The process is divided into two distinct stages: the formation of the enamine intermediate and its subsequent catalytic conversion to the final indole product. Operators must ensure that water is effectively removed during the first step, often using a water separator during reflux, to drive the equilibrium towards the enamine. The second step demands precise temperature control, typically between 80°C and 200°C, depending on the solvent and catalyst system employed. While the patent provides specific examples using Pd/C and Raney Nickel, the flexibility of the catalyst system allows for optimization based on available infrastructure. The detailed standardized synthesis steps, including exact molar ratios, solvent volumes, and workup procedures, are critical for maintaining batch-to-batch consistency. For a comprehensive guide on executing this protocol, please refer to the technical instructions below.
- Condense 1,3-cyclohexanedione with 2-aminoethanol in a solvent like toluene to form the enamine intermediate, removing water via azeotropic distillation.
- Subject the isolated enamine compound to dehydrogenative cyclization using a supported metal catalyst such as Pd/C or Raney Nickel at elevated temperatures.
- Purify the crude 4-hydroxyindole product through vacuum distillation and recrystallization to achieve high-purity specifications suitable for API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers compelling advantages that directly address the core concerns of procurement managers and supply chain heads regarding cost and continuity. The primary driver for cost optimization is the substitution of raw materials; replacing expensive 2-chloroacetaldehyde with 2-aminoethanol results in a significant reduction in direct material costs. Additionally, the elimination of high-pressure reaction vessels reduces capital expenditure on specialized equipment and lowers the associated maintenance and inspection costs. The process also generates hydrogen gas as a byproduct, which is environmentally benign compared to the salt waste and corrosive effluents of the traditional method, leading to substantial cost savings in waste treatment and environmental compliance. These factors combine to create a more economically sustainable manufacturing model that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both raw material efficiency and operational simplification. By avoiding the use of high-cost chlorinated precursors and eliminating the need for pressure-rated reactors, the overall cost of goods sold is drastically reduced. The ability to use standard atmospheric reactors also lowers energy consumption associated with maintaining high-pressure systems. Furthermore, the recovery and reuse of metal catalysts, such as Pd/C, add another layer of value by minimizing catalyst consumption costs. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of widely available and stable raw materials like 1,3-cyclohexanedione and 2-aminoethanol. Unlike specialized chlorinated compounds that may face supply constraints, these starting materials are commodity chemicals with robust global supply networks. The simplified process flow also reduces the risk of production delays caused by equipment failure or complex safety protocols associated with high-pressure ammonia handling. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to maintain consistent production schedules and meet market demand more effectively.
- Scalability and Environmental Compliance: The scalability of this method is enhanced by its compatibility with standard chemical processing equipment, facilitating easier commercial scale-up of complex pharmaceutical intermediates. The absence of corrosive ammonia and chlorinated byproducts simplifies waste management and reduces the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only ensures compliance with increasingly strict environmental regulations but also enhances the corporate sustainability profile of the manufacturer. The clean reaction profile minimizes the need for extensive effluent treatment, further streamlining the path from pilot scale to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of the new route. Understanding these details is crucial for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios. The answers provided reflect the specific advantages related to safety, cost, and scalability that distinguish this approach from conventional methods.
Q: What are the primary safety advantages of this new 4-hydroxyindole synthesis route?
A: The novel method eliminates the need for high-temperature and high-pressure reaction vessels required by conventional ammonia-based processes. By avoiding pressurized ammonia, the risk of equipment corrosion and potential leakage is significantly reduced, enhancing overall operational safety for commercial scale-up of complex pharmaceutical intermediates.
Q: How does the new catalyst system impact production costs compared to traditional methods?
A: The process replaces expensive 2-chloroacetaldehyde with cost-effective 2-aminoethanol. Furthermore, the use of recoverable supported metal catalysts and the generation of hydrogen gas as a clean byproduct, rather than salt waste, contributes to substantial cost savings in waste treatment and raw material procurement.
Q: Is this synthesis method scalable for industrial manufacturing of beta-blocker intermediates?
A: Yes, the reaction conditions operate at atmospheric pressure with standard organic solvents like toluene or trimethylbenzene. This simplicity facilitates easier commercial scale-up of complex pharmaceutical intermediates, ensuring consistent supply continuity for downstream API production like pindolol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxyindole Supplier
The technical potential of this dehydrogenative aromatization route underscores the importance of partnering with a CDMO that possesses the expertise to translate patent innovations into commercial reality. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary infrastructure to handle catalytic hydrogenation and dehydrogenation processes safely and efficiently, ensuring that the benefits of this new method are fully realized. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-hydroxyindole meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence ensures that the transition from lab-scale discovery to industrial supply is seamless and reliable.
We invite global partners to explore how this optimized synthesis route can enhance their supply chain efficiency and reduce overall manufacturing costs. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine the best fit for your production needs. Let us help you secure a stable, high-quality supply of this critical intermediate while optimizing your operational expenditures through advanced chemical engineering.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
