Revolutionizing Indole Derivative Production: A Scalable and Cost-Effective Synthetic Route for Global Pharma Supply Chains
Revolutionizing Indole Derivative Production: A Scalable and Cost-Effective Synthetic Route for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with economic viability, particularly for critical heterocyclic scaffolds like indoles. Patent CN115536571A introduces a groundbreaking preparation method for indole derivatives that addresses long-standing inefficiencies in traditional manufacturing protocols. This innovative technical disclosure outlines a sequential four-step process involving hydroxyl protection, condensation, cyclization, and deprotection, which collectively enable the production of high-purity indole derivatives such as 4-hydroxyindole and 6-hydroxyindole. By leveraging cheap and easily obtained raw materials alongside optimized catalytic systems, this methodology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to streamline their production capabilities. The technical breakthrough lies not only in the chemical transformations themselves but in the strategic selection of reagents that mitigate toxicity and cost while maximizing overall throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxyindole derivatives has been plagued by significant operational and economic hurdles that hinder efficient commercial scale-up of complex heterocyclic compounds. As illustrated in the prior art referenced within the patent background, traditional routes often rely heavily on noble metal catalysts, such as palladium, which introduce substantial cost burdens and supply chain vulnerabilities due to the volatility of precious metal markets. Furthermore, conventional protection strategies frequently utilize benzyl bromide, a reagent known for its extreme irritation and toxicity, posing severe safety risks to personnel and requiring elaborate waste management protocols that inflate operational expenditures. The cumulative effect of these factors is often a disappointingly low total yield, reported in some prior art instances to be approximately 10%, which renders such processes economically unfeasible for large-volume manufacturing. Additionally, the removal of benzyl protection in the final steps typically necessitates further hydrogenation with expensive metal catalysts, compounding the cost and complexity of the purification workflow.
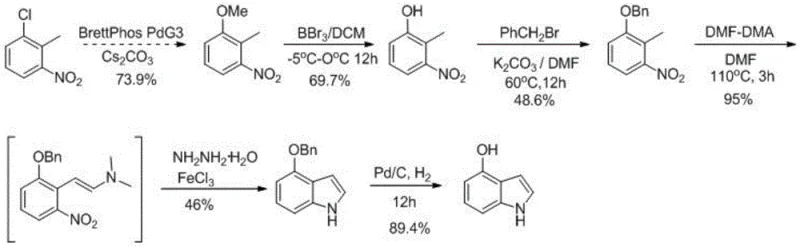
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN115536571A presents a streamlined and economically superior pathway that effectively circumvents the reliance on precious metals and hazardous alkylating agents. This new strategy employs a tetrahydropyranyl (THP) protection group, introduced via reaction with 3,4-dihydro-2H-pyran under mild acidic catalysis, which offers superior stability during subsequent reaction steps and can be removed under gentle acidic conditions without the need for hydrogenation. The core innovation is the utilization of Raney-Nickel as a cyclization catalyst in conjunction with hydrazine hydrate, a combination that facilitates the reductive cyclization of the nitro-enamine intermediate with remarkable efficiency and selectivity. This shift from palladium to Raney-Nickel represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, as it eliminates the need for expensive ligand systems and complex metal scavenging procedures. The result is a robust process capable of achieving overall yields exceeding 65%, with individual steps demonstrating exceptional conversion rates that validate its suitability for industrial application.
Mechanistic Insights into Raney-Ni Catalyzed Cyclization
The mechanistic elegance of this synthesis is most evident in the cyclization step, where the nitro group is reduced and simultaneously participates in ring closure to form the indole core. The use of hydrazine hydrate as the reducing agent in the presence of Raney-Nickel creates a highly reactive environment that selectively reduces the nitro functionality to an intermediate amine or hydroxylamine species, which then undergoes intramolecular nucleophilic attack on the adjacent enamine double bond. This cascade reaction is meticulously controlled by maintaining the reaction temperature between 35°C and 50°C, ensuring that the reduction proceeds rapidly without compromising the integrity of the sensitive enamine moiety or the THP protecting group. The choice of a mixed solvent system comprising tetrahydrofuran and methanol is critical, as it ensures optimal solubility of the organic substrate while facilitating the interaction between the solid catalyst surface and the dissolved reactants. This precise control over reaction parameters minimizes the formation of side products, such as over-reduced species or polymerized byproducts, thereby enhancing the purity profile of the crude product before any purification is attempted.
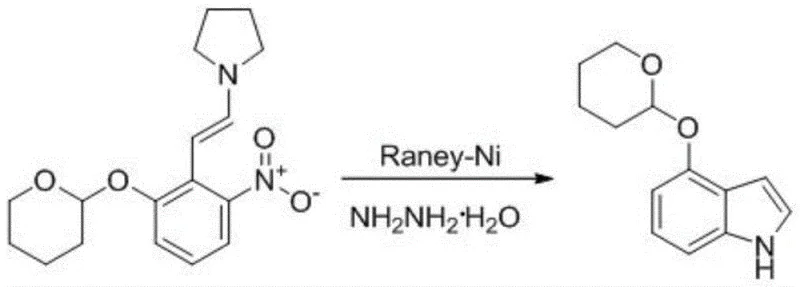
Furthermore, the impurity control mechanism inherent in this route is bolstered by the orthogonality of the protection and deprotection steps. The THP group remains stable during the high-temperature condensation reaction (120-135°C) and the reductive cyclization, preventing unwanted side reactions at the phenolic hydroxyl position that could lead to regio-isomeric impurities. Upon completion of the ring closure, the deprotection is executed using trifluoroacetic acid (TFA) at low temperatures (0-10°C), which cleaves the ether linkage cleanly to regenerate the free phenol. This mild acidic deprotection avoids the harsh conditions often associated with Lewis acid-mediated deprotections, thereby preserving the delicate indole nucleus from acid-catalyzed degradation or polymerization. The combination of selective reduction and orthogonal protection ensures that the final product meets stringent purity specifications required for downstream pharmaceutical applications, reducing the burden on downstream purification units.
How to Synthesize 4-Hydroxyindole Efficiently
For research and development teams looking to implement this technology, the synthesis of 4-hydroxyindole serves as a prime example of the protocol's efficacy and operational simplicity. The process begins with the protection of 3-nitro-4-methylphenol using 3,4-dihydro-2H-pyran and a catalytic amount of pyridine p-toluenesulfonate (PPTS) in dichloromethane, followed by a condensation reaction with DMF-DMA and pyrrolidine to generate the key enamine intermediate. The subsequent cyclization with hydrazine hydrate and Raney-Ni, followed by acidic deprotection, delivers the target molecule in high yield. The detailed standardized synthesis steps below outline the specific molar ratios, temperature profiles, and workup procedures necessary to replicate these results consistently in a laboratory or pilot plant setting.
- Perform hydroxyl protection of the nitrotoluene precursor using 3,4-dihydro-2H-pyran and PPTS catalyst in dichloromethane.
- Execute condensation reaction with pyrrolidine and DMF-DMA in DMF solvent at elevated temperatures to form the enamine intermediate.
- Conduct cyclization using hydrazine hydrate and Raney-Ni catalyst in a THF/methanol mixture to close the indole ring.
- Finalize the synthesis by deprotecting the tetrahydropyranyl group using trifluoroacetic acid to obtain the target hydroxyindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis route offers transformative advantages that directly address the pain points of cost volatility and material availability in the fine chemical sector. By eliminating the dependency on palladium catalysts and benzyl bromide, manufacturers can achieve substantial cost savings through the use of commodity chemicals that are readily available in the global market. The replacement of noble metals with Raney-Nickel not only reduces the direct material cost but also simplifies the supply chain by removing the need for specialized metal recovery vendors and complex regulatory compliance associated with heavy metal residues in API intermediates. This shift enhances supply chain reliability by diversifying the source of critical reagents, ensuring that production schedules are not disrupted by shortages of exotic catalysts or protected starting materials.
- Cost Reduction in Manufacturing: The economic impact of switching to this route is profound, driven primarily by the substitution of high-cost inputs with low-cost alternatives. The elimination of palladium catalysts removes a significant line item from the bill of materials, while the use of hydrazine hydrate and Raney-Nickel provides a highly efficient reduction system at a fraction of the cost of hydrogenation with precious metals. Furthermore, the simplified workup procedures, which avoid complex chromatographic purifications often needed to remove metal traces, reduce solvent consumption and labor hours, leading to a leaner and more cost-effective manufacturing process. These cumulative efficiencies translate into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as 3,4-dihydro-2H-pyran and pyrrolidine ensures a consistent and predictable supply of raw materials. Unlike specialized ligands or sensitive organometallic complexes that may have long lead times or limited suppliers, the reagents in this process are produced at scale by multiple chemical manufacturers worldwide. This redundancy in the supply base mitigates the risk of production stoppages due to raw material shortages, providing procurement managers with greater confidence in meeting delivery commitments. Additionally, the robustness of the THP protecting group allows for the storage of intermediates without significant degradation, offering flexibility in production planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as extraction, crystallization, and filtration that are easily transferred from the laboratory to multi-ton reactors. The avoidance of toxic benzyl bromide and the use of aqueous workups with saturated brine and sodium bicarbonate align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards simplifies the permitting process for new production lines and reduces the costs associated with hazardous waste disposal. The ability to scale this process from 100 kgs to 100 MT annual commercial production without significant re-engineering makes it an ideal candidate for meeting the growing demand for high-purity indole derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indole derivative preparation method. These answers are derived directly from the technical specifications and experimental data provided in patent CN115536571A, ensuring accuracy and relevance for stakeholders evaluating this technology for adoption. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this new method improve upon traditional palladium-catalyzed synthesis?
A: The new method replaces expensive and toxic palladium catalysts and benzyl bromide protecting groups with cost-effective Raney-Ni and tetrahydropyranyl protection, significantly lowering material costs and simplifying purification.
Q: What is the overall yield efficiency of this indole derivative preparation?
A: According to patent CN115536571A, the overall yield of the indole derivative prepared by this method can reach more than 65%, with individual step yields often exceeding 90%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and easily obtained raw materials, avoids extreme conditions, and employs robust catalysts like Raney-Ni, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the technical potential of the route described in CN115536571A and is fully prepared to support your transition to this more efficient manufacturing paradigm. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel process are realized in practical, large-scale operations. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications protocols, guaranteeing that every batch of indole derivatives we produce meets the highest standards of quality and consistency required by top-tier pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume and current sourcing strategy. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-quality indole intermediates into your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
