Advanced Ibrutinib Manufacturing: Technical Breakthroughs for Commercial Scale-Up
Advanced Ibrutinib Manufacturing: Technical Breakthroughs for Commercial Scale-Up
The global demand for Btk kinase inhibitors continues to surge, driven by the critical need for effective treatments in B-lymphocyte malignant proliferative diseases such as Mantle Cell Lymphoma (MCL) and Chronic Lymphocytic Leukemia (CLL). At the forefront of this therapeutic revolution is Ibrutinib, a molecule whose market potential is projected to reach billions in annual sales. However, the complexity of its chemical structure has historically posed significant challenges for manufacturers aiming to deliver high-purity material at a viable cost. A pivotal advancement in this domain is detailed in patent CN107383017B, which discloses a highly efficient preparation method that fundamentally reimagines the synthetic pathway. This technical insight report analyzes the profound implications of this patent for R&D directors and supply chain leaders, highlighting how the elimination of hazardous reagents and the optimization of chiral control can redefine the standards for reliable pharmaceutical intermediate supplier partnerships.
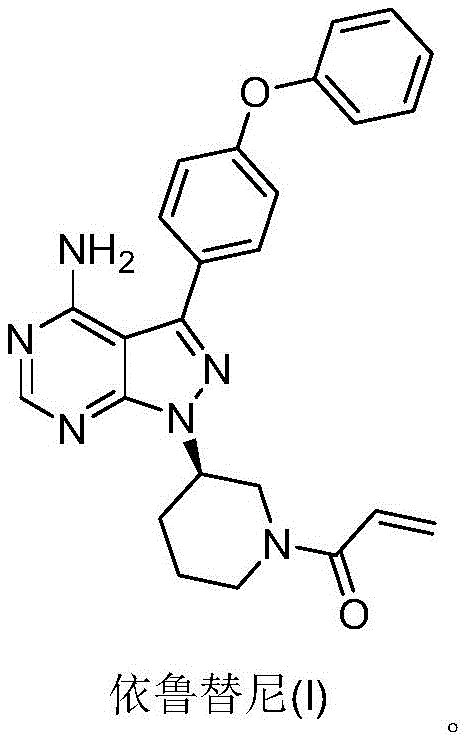
The structural complexity of Ibrutinib requires precise stereochemical control, particularly at the piperidine ring junction. Traditional methods often struggle to maintain optical purity while managing the accumulation of functional groups. The methodology outlined in CN107383017B addresses these pain points by introducing a route that is not only operationally simpler but also inherently safer and more scalable. By shifting away from legacy protocols that rely on unstable intermediates, this new approach offers a robust framework for cost reduction in API manufacturing. For procurement managers, understanding the nuances of this synthetic evolution is crucial, as it directly impacts the long-term stability of supply and the overall cost of goods sold. The following analysis dissects the technical merits of this innovation, providing a clear roadmap for integrating these advancements into your procurement strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ibrutinib has been plagued by inefficient pathways that hinder industrial scalability and compromise product quality. Prior art, including patents such as US20110039190 and US20100254905, typically relies on a linear accumulation of functional groups starting from 4-phenoxybenzoic acid. A critical bottleneck in these conventional routes is the reliance on the Mitsunobu reaction to introduce the chiral center on the piperidine ring. While chemically feasible on a small scale, the Mitsunobu reaction is notorious for causing partial racemization, which inevitably lowers the optical purity of the final product. This necessitates additional, costly purification steps to meet stringent regulatory standards for chiral drugs. Furthermore, these legacy routes often employ hazardous reagents such as trimethylsilyldiazomethane and triphenylphosphine, which pose significant safety risks and environmental disposal challenges, making them unsuitable for modern green chemistry standards.
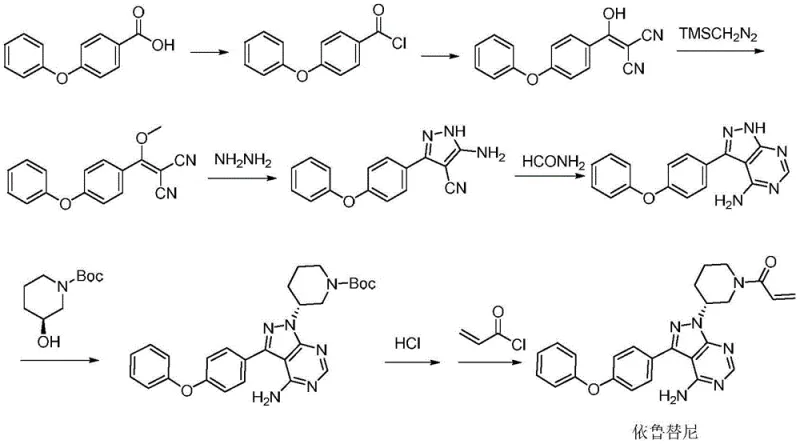
Another significant drawback in earlier methodologies, such as those described in European patent WO2013003629, is the dependence on Suzuki coupling reactions. While Suzuki coupling is a powerful tool, it requires the use of expensive and often unstable boronic acid intermediates. The cost volatility of these boron reagents can introduce unpredictability into the supply chain, affecting both pricing and lead times. Additionally, the removal of transition metal catalysts used in these couplings adds another layer of complexity to the downstream processing, increasing the burden on quality control laboratories to ensure residual metal levels are within acceptable limits. These cumulative inefficiencies result in a manufacturing process that is not only costly but also fragile, with multiple points of failure that can disrupt the continuity of supply for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the method disclosed in CN107383017B presents a streamlined and robust alternative that directly addresses the core inefficiencies of prior art. This novel approach bypasses the problematic Mitsunobu reaction entirely, opting instead for a chiral resolution strategy using D-pyroglutamic acid. This shift is transformative, as it allows for the establishment of the critical chiral center early in the synthesis with high fidelity, thereby preserving optical purity throughout the subsequent steps. The route is characterized by mild reaction conditions and the use of readily available reagents, which significantly lowers the barrier to entry for commercial scale-up. By eliminating the need for hazardous diazo compounds and expensive boronic acids, this method offers a safer and more economically viable pathway for production.
The strategic redesign of the synthetic sequence also enhances the overall yield and purity profile of the final API. The new route utilizes a pyrazole cyclization reaction between a hydrazino-piperidine intermediate and a specific vinylenedicyanomethane derivative. This convergent strategy reduces the total number of steps required to reach the target molecule, minimizing material loss and waste generation. For supply chain heads, this reduction in step count translates to a shorter manufacturing cycle and reduced lead time for high-purity API intermediates. The robustness of this chemistry ensures that the process can be reliably scaled from kilogram to multi-ton quantities without the significant re-optimization often required for more sensitive catalytic processes. This stability is a key value proposition for partners seeking a reliable long-term source of complex oncology therapeutics.
Mechanistic Insights into Chiral Resolution and Pyrazole Cyclization
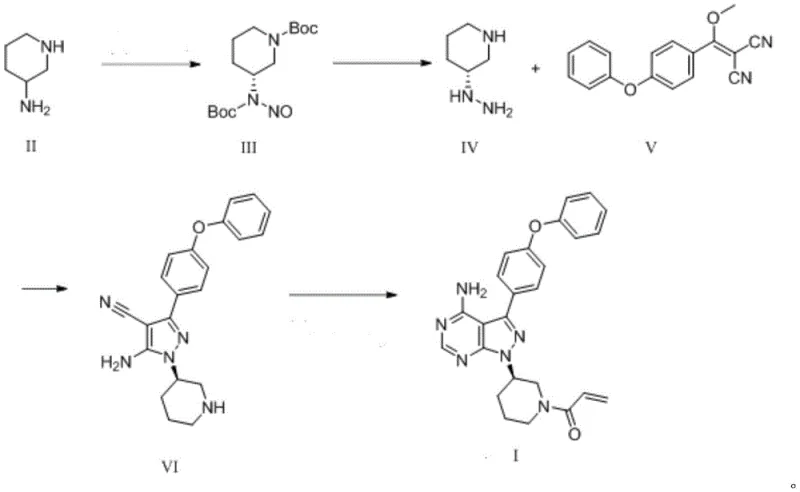
The cornerstone of this improved synthesis lies in the meticulous control of stereochemistry during the initial stages of the pathway. The process begins with the resolution of racemic 3-aminopiperidine using D-pyroglutamic acid in anhydrous ethanol. This step is critical, as it leverages the formation of diastereomeric salts to isolate the desired (R)-enantiomer with high efficiency. The resulting R-3-aminopiperidine pyroglutamate is then subjected to Boc protection and subsequent nitrosation. The use of sodium nitrite and periodic acid in this nitrosation step is a gentle yet effective method to prepare the nitroso intermediate without compromising the integrity of the chiral center. This stands in sharp contrast to inversion methods that risk racemization. The subsequent reduction of the nitroso group to a hydrazine functionality is performed under mild conditions using metal powders like zinc or iron in an acetic acid system, ensuring that the sensitive piperidine ring remains intact.
Following the preparation of the chiral hydrazine intermediate, the synthesis proceeds through a highly efficient pyrazole cyclization. The reaction between the 3R-hydrazino-1-piperidine compound and 4-phenoxyphenyl(methoxy)vinylenedicyanomethane is facilitated by basic conditions, leading to the formation of the pyrazole core with excellent regioselectivity. This cyclization step is pivotal, as it constructs the central scaffold of the Ibrutinib molecule in a single operation. The subsequent pyrimidine ring closure is achieved using cyclizing agents such as formamide or orthoformates under thermal conditions. This sequence avoids the use of transition metal catalysts entirely, thereby eliminating the risk of heavy metal contamination in the final product. The final acrylation step is straightforward, coupling the secondary amine with acryloyl chloride to yield the target molecule with an ee value of up to 99.3%, demonstrating the superior stereocontrol of this novel pathway.
How to Synthesize Ibrutinib Efficiently
The implementation of this synthesis route requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity. The process is designed to be operationally simple, utilizing standard laboratory equipment that can be easily adapted for pilot and commercial plant operations. The key to success lies in the careful control of stoichiometry during the chiral resolution and the maintenance of anhydrous conditions during the protection steps. Detailed standard operating procedures for each transformation, including specific temperature ranges and workup protocols, are essential for reproducibility. For technical teams looking to adopt this methodology, the following guide outlines the critical stages of the process.
- Perform chiral resolution of 3-aminopiperidine using D-pyroglutamic acid, followed by Boc protection and nitrosation to yield nitroso compound III.
- Execute nitro reduction in a reducing agent/AcOH system and remove the Boc protecting group to obtain 3R-hydrazino-1-piperidine compound IV.
- Conduct pyrazole cyclization with 4-phenoxyphenyl(methoxy)vinylenedicyanomethane, followed by pyrimidine cyclization and final acrylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical improvements offered by patent CN107383017B translate directly into tangible commercial benefits. The primary advantage is the substantial cost savings achieved through the simplification of the raw material portfolio. By removing the dependency on expensive boronic acids and hazardous diazo reagents, the direct material cost of the synthesis is drastically reduced. Furthermore, the elimination of transition metal catalysts removes the need for specialized scavenging resins and extensive metal testing, which lowers both the cost and the time associated with quality assurance. This streamlined cost structure allows for more competitive pricing models, providing a significant buffer against market volatility in raw material costs.
- Cost Reduction in Manufacturing: The novel synthetic route eliminates several high-cost reagents and complex purification steps associated with traditional methods. By avoiding the Mitsunobu reaction and Suzuki coupling, the process reduces the consumption of expensive chiral auxiliaries and boron reagents. This simplification leads to a lower cost of goods sold (COGS) without compromising the quality of the final API. Additionally, the higher overall yield of the new route means that less starting material is required to produce the same amount of product, further enhancing economic efficiency. These factors combine to create a manufacturing process that is financially sustainable and resilient to price fluctuations in the chemical market.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents significantly de-risks the supply chain. Unlike unstable boronic acids that require special storage and handling, the reagents used in this new pathway are commodity chemicals with robust global supply networks. This ensures a consistent flow of materials and reduces the risk of production stoppages due to raw material shortages. The simplified process also shortens the manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand. For supply chain heads, this reliability is crucial for maintaining inventory levels and ensuring uninterrupted drug production for patients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process highly scalable and environmentally friendly. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the costs associated with waste disposal and compliance reporting. The process is designed to be easily transferred from laboratory to commercial scale, with minimal need for re-engineering. This scalability ensures that production capacity can be rapidly expanded to meet growing market demand. The green chemistry profile of this route also enhances the corporate sustainability image, which is an increasingly important factor for pharmaceutical partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of this technology for your specific production needs. The insights provided here cover aspects of purity, cost, and scalability that are critical for stakeholders.
Q: How does this new route improve optical purity compared to Mitsunobu reactions?
A: The novel method avoids the Mitsunobu reaction, which is known to cause partial racemization. Instead, it utilizes chiral resolution with D-pyroglutamic acid, achieving ee values up to 99.3%.
Q: What are the cost advantages of avoiding Suzuki coupling in this synthesis?
A: By eliminating the need for expensive and unstable boronic acid intermediates required in Suzuki coupling, the manufacturing cost is significantly reduced while simplifying the supply chain.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, simple operations, and avoids hazardous reagents like trimethylsilyldiazomethane, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Supplier
The technical potential of the synthesis route disclosed in CN107383017B represents a significant opportunity for optimizing the production of this critical oncology therapeutic. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative chemistry to life. Our facilities are equipped with rigorous QC labs and stringent purity specifications that ensure every batch meets the highest international standards. We understand the complexities of chiral synthesis and are prepared to implement this efficient route to deliver high-purity Ibrutinib that supports your clinical and commercial goals.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing method can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project. Our team is ready to provide the technical support and commercial flexibility needed to secure a stable and cost-effective supply of this vital pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
