Advanced Synthetic Routes for Temozolomide: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing processes that balance safety, efficiency, and cost-effectiveness, particularly for potent oncology agents like temozolomide. Patent CN1487941A introduces a transformative synthetic methodology that addresses critical bottlenecks in the production of this alkylating agent. By shifting away from the isolation of unstable diazo intermediates and hazardous reagents like methyl isocyanate, this novel process utilizes stable, protected amine precursors to achieve high-purity outcomes. For a reliable pharmaceutical intermediates supplier, adopting such advanced protocols is essential to meet the stringent regulatory requirements of global health authorities. This report analyzes the technical merits of this invention, demonstrating how it facilitates cost reduction in API manufacturing while ensuring a consistent supply of clinical-grade material for downstream drug product formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of temozolomide has relied on pathways that involve significant safety and operational risks, primarily centered around the handling of unstable intermediates. As illustrated in the traditional Scheme I, the process necessitates the conversion of 5-amino-1H-imidazole-4-carboxamide into 5-diazo-1H-imidazole-4-carboxamide, a compound known for its instability and potential hazard profile.
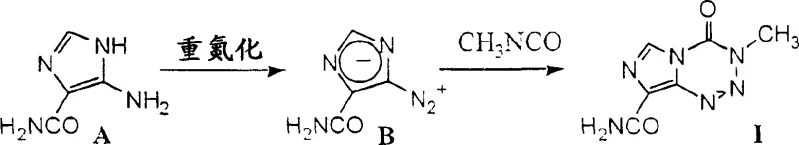
Furthermore, the subsequent cycloaddition step typically employs methyl isocyanate, a reagent that is notoriously difficult to handle and transport on an industrial scale due to its high toxicity and volatility. Literature data suggests that this cycloaddition can require excessively long reaction times, potentially up to 20 days, which severely impacts throughput and equipment utilization. The necessity to isolate the unstable diazo compound creates a bottleneck, forcing manufacturers to operate under strict, often costly, safety containment measures that limit scalability and increase the overall cost of goods sold.
The Novel Approach
The innovative strategy detailed in the patent circumvents these issues by employing a protected amine strategy, specifically utilizing bulky protecting groups such as the 1,1-dimethylethyl (tert-butyl) moiety. This approach allows for the construction of the imidazole core through stable intermediates that can be isolated, characterized, and stored without degradation. The key breakthrough lies in the ability to perform the critical ring-closing steps without generating free, unstable diazo species until the final cyclization event, which occurs spontaneously under controlled conditions.
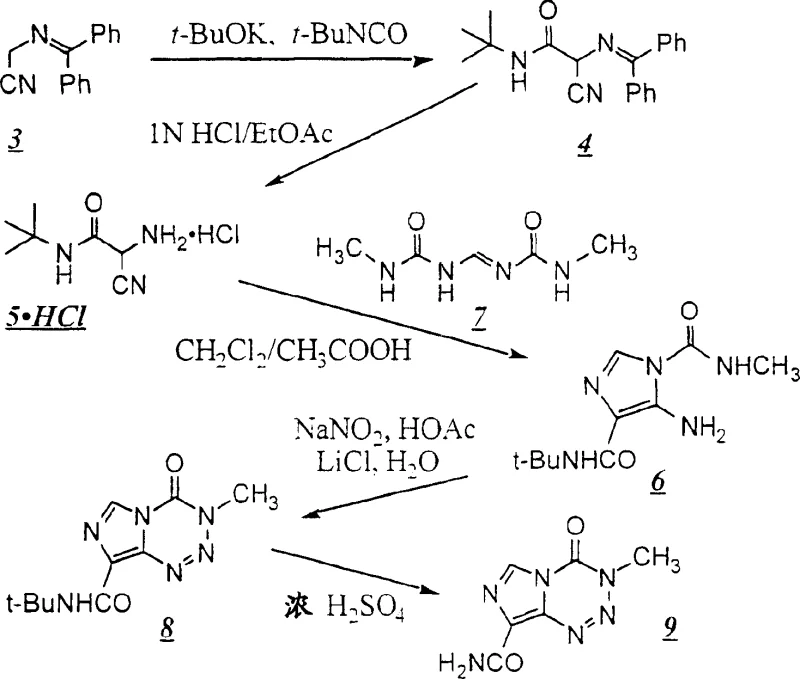
By replacing methyl isocyanate with safer urea derivatives and utilizing stable acetamide precursors, the new route significantly lowers the barrier for commercial scale-up of complex heterocycles. This method not only improves the safety profile of the plant operations but also enhances the overall yield by minimizing side reactions associated with the decomposition of reactive intermediates. The result is a streamlined process that is far more amenable to Good Manufacturing Practice (GMP) standards required for oncology drug production.
Mechanistic Insights into Steric-Directed Cyclization
A critical aspect of this synthesis is the control over regioselectivity during the formation of the fused tetrazine ring. Theoretically, the cyclization of the imidazole precursor could occur at the nitrogen atom of the carbamoyl group, leading to the formation of undesirable azahypoxanthine derivatives. However, the presence of the bulky tert-butyl protecting group exerts a profound steric influence that directs the reaction exclusively towards the desired imidazo[5,1-d]-1,2,3,5-tetrazine framework. This steric hindrance effectively blocks the unfavorable pathway, ensuring that the nucleophilic attack occurs at the correct position to form the bioactive scaffold.
Additionally, the reaction medium plays a pivotal role in optimizing this selectivity. The inclusion of lithium chloride (LiCl) in the aqueous organic solution during the diazotization step has been found to favor the correct orientation of the cyclization. This additive likely influences the solvation of the transition state or the stability of the diazonium intermediate, further suppressing the formation of impurities. Such mechanistic understanding allows process chemists to fine-tune reaction conditions, ensuring that high-purity temozolomide is obtained with minimal downstream purification burden, which is crucial for maintaining economic viability in large-scale production.
How to Synthesize Temozolomide Efficiently
The execution of this synthesis involves a sequence of well-defined chemical transformations that prioritize safety and yield. The process begins with the protection of the amino group, followed by ring construction and final deprotection. Each step is designed to maximize the stability of the intermediates, allowing for rigorous quality control checks before proceeding to the next stage. This modular approach is highly advantageous for contract development and manufacturing organizations (CDMOs) as it allows for flexible production scheduling and inventory management of key building blocks.
- React [(diphenylmethylene)amino]acetonitrile with tert-butyl isocyanate in the presence of a base like potassium tert-butoxide to form the protected acetamide intermediate.
- Hydrolyze the diphenylmethylene protecting group using mild acid to yield the amino-cyano-acetamide hydrochloride salt.
- Condense the amine salt with a urea derivative to form the imidazole ring, followed by diazotization and cyclization to form the protected tetrazine.
- Perform final acidic hydrolysis to remove the tert-butyl group, yielding clinical-grade temozolomide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical elegance. The elimination of hazardous reagents like methyl isocyanate removes a significant logistical burden, as there is no longer a need for specialized transport and storage infrastructure for toxic gases. This simplification directly translates to reduced operational overhead and lower insurance costs, contributing to substantial cost savings in the overall manufacturing budget without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and dangerous reagents, replacing them with stable, commercially available solids. By avoiding the isolation of unstable diazo compounds, the facility reduces the risk of batch loss due to decomposition, thereby improving the effective yield. Furthermore, the shorter reaction times compared to conventional methods mean higher equipment turnover, allowing for increased production capacity within the same timeframe and driving down the unit cost of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Stability is the cornerstone of a resilient supply chain. The intermediates generated in this route, such as the protected acetamides and imidazoles, are stable solids that can be stockpiled. This capability allows manufacturers to buffer against raw material fluctuations and demand spikes. Reducing lead time for high-purity intermediates becomes feasible because the process does not rely on just-in-time generation of hazardous species, ensuring a continuous and predictable flow of material to the final drug product manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, but this route mitigates those risks through inherent safety design. The use of aqueous workups and the avoidance of volatile toxic gases simplify waste treatment and effluent management. This aligns with increasingly stringent environmental regulations, reducing the liability associated with hazardous waste disposal. The robustness of the chemistry ensures that the process performs consistently from kilogram to multi-ton scales, guaranteeing supply continuity for long-term commercial contracts.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating potential partnerships. The following questions address common concerns regarding the implementation and benefits of this technology. These answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers assessing process feasibility.
Q: Why is the conventional diazotization method considered hazardous for industrial scale-up?
A: Conventional methods require the isolation of unstable 5-diazo-1H-imidazole-4-carboxamide, which poses significant safety risks due to potential explosivity and decomposition during storage and transport.
Q: How does the tert-butyl protecting group improve the cyclization selectivity?
A: The bulky steric nature of the 1,1-dimethylethyl (tert-butyl) group directs the cyclization specifically towards the formation of the desired imidazo[5,1-d]-1,2,3,5-tetrazine ring, minimizing the formation of unwanted azahypoxanthine byproducts.
Q: What are the supply chain advantages of using stable intermediates in this synthesis?
A: Using stable, isolable intermediates allows for quality control at multiple stages, reduces the need for immediate downstream processing, and eliminates the logistics challenges associated with transporting hazardous reagents like methyl isocyanate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Temozolomide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1487941A can be successfully implemented at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of temozolomide meets the exacting standards required for oncology therapeutics, providing our partners with absolute confidence in material quality.
We invite global pharmaceutical companies to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this safer, more efficient route. We encourage you to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-value chemical solutions that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
