Advanced Synthesis of Tetraaryl-Carbazole Derivatives for High-Efficiency OLED Commercialization
Advanced Synthesis of Tetraaryl-Carbazole Derivatives for High-Efficiency OLED Commercialization
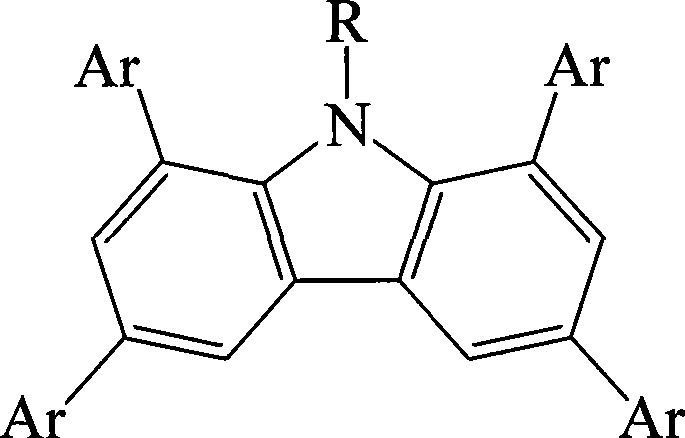
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that combine high quantum efficiency with exceptional thermal and morphological stability. Patent CN101126020B introduces a groundbreaking class of 9-alkyl-1,3,6,8-tetraaryl substituted carbazole derivatives designed specifically to address the limitations of traditional host materials in blue phosphorescent devices. This technology leverages precise molecular engineering to elevate triplet excited state energies while maintaining excellent hole transport properties, a critical balance for next-generation display manufacturing. By utilizing a robust Suzuki coupling strategy, the patent outlines a scalable pathway to produce these complex architectures with high purity, making them ideal candidates for commercial adoption in flat panel displays and white lighting applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on 4,4'-bis(9-carbazolyl-)biphenyl (CBP) as a standard host material for phosphorescent OLEDs due to its balanced charge transport capabilities. However, CBP suffers from a fundamental energetic limitation; its triplet excited state energy is merely 2.56eV, which is insufficient for hosting high-energy blue phosphorescent dopants that require levels above 2.65eV. When used with blue emitters, this energy mismatch leads to reverse energy transfer from the dopant back to the host, drastically reducing the external quantum efficiency of the device to as low as 5.7%. Furthermore, the planar structure of conventional carbazole derivatives often facilitates molecular stacking, resulting in concentration quenching and exciplex formation that degrade color purity and device lifespan under operational stress.
The Novel Approach
The innovative methodology described in the patent overcomes these barriers by strategically introducing four bulky aryl substituents at the 1, 3, 6, and 8 positions of the carbazole core. This tetra-substitution pattern induces a significant twist in the molecular geometry, effectively breaking the planar conjugation and isolating the luminescent center from intermolecular interactions. This structural distortion not only raises the triplet energy level to values compatible with deep-blue emission but also physically prevents the close packing of molecules that leads to quenching. Additionally, the inclusion of a 9-alkyl chain enhances solubility in common organic solvents such as chloroform and toluene, facilitating solution-processable fabrication methods that are more cost-effective than traditional vacuum deposition techniques.
Mechanistic Insights into Suzuki-Catalyzed Arylation
The core synthetic transformation relies on a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, which connects the electron-deficient 1,3,6,8-tetrabromocarbazole intermediate with various electron-rich arylboronic acids. The mechanism initiates with the oxidative addition of the palladium catalyst into the carbon-bromine bonds at the sterically crowded 1 and 8 positions, a step facilitated by the use of tetrabutylammonium bromide (TBAB) as a phase-transfer catalyst. This quaternary ammonium salt plays a crucial role in solubilizing the inorganic base, typically sodium carbonate, within the organic acetone phase, thereby accelerating the transmetallation step where the aryl group is transferred to the palladium center. The reaction is conducted under rigorous nitrogen protection to prevent oxidation of the phosphine ligands or the palladium species, ensuring high catalytic turnover numbers even with the steric bulk of the tetra-substituted substrate.
Impurity control is paramount in electronic grade materials, and this process incorporates specific purification protocols to remove residual palladium and homocoupling byproducts. Following the reflux period, which can extend up to 40 hours for sterically hindered substrates like naphthyl boronic acids, the crude product undergoes liquid-liquid extraction using dichloromethane to separate organic products from inorganic salts. The final purification is achieved through silica gel column chromatography, which effectively separates the desired tetra-aryl product from mono-, di-, or tri-substituted intermediates that may form due to incomplete reaction. This rigorous downstream processing ensures that the final material meets the stringent purity specifications required for OLED applications, where trace metallic impurities can act as quenching sites and significantly reduce device efficiency.
How to Synthesize 9-Alkyl-1,3,6,8-Tetraaryl Carbazole Efficiently
The synthesis protocol is divided into three distinct stages: N-alkylation to protect the nitrogen center, electrophilic bromination to activate the ring positions, and the final cross-coupling to install the functional aryl groups. Each step requires precise control of temperature and stoichiometry to maximize yield and minimize side reactions such as poly-bromination at unwanted positions or dehalogenation. The detailed standardized synthesis steps below outline the specific molar ratios, solvent systems, and workup procedures validated in the patent examples to ensure reproducibility at scale.
- Perform N-alkylation of carbazole using dimethyl sulfate or alkyl bromides in DMSO with KOH and TEBA catalyst under nitrogen atmosphere to secure the 9-position.
- Execute electrophilic aromatic substitution by reacting the 9-alkylcarbazole with excess bromine in glacial acetic acid at 90°C for 10 hours to achieve 1,3,6,8-tetrabromination.
- Conduct Suzuki-Miyaura cross-coupling between the tetrabromo-intermediate and various arylboronic acids using PdCl2 catalyst and TBAB phase transfer agent in acetone-water mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthetic route offers tangible benefits regarding raw material security and processing efficiency. The starting materials, including carbazole, simple alkyl halides, and commercially available boronic acids, are commodity chemicals with stable global supply chains, reducing the risk of production bottlenecks. Furthermore, the use of acetone and water as co-solvents in the final coupling step represents a greener alternative to exclusive reliance on toxic aprotic solvents, simplifying waste treatment protocols and lowering environmental compliance costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of complex vacuum deposition requirements for film formation, enabled by the high solubility of these derivatives, allows manufacturers to transition to solution-based coating techniques like inkjet printing. This shift significantly reduces capital expenditure on vacuum equipment and lowers energy consumption during the device fabrication process. Additionally, the high yields reported in the bromination step (up to 88%) and the robustness of the Suzuki coupling minimize raw material waste, directly contributing to a lower cost of goods sold per kilogram of active material produced.
- Enhanced Supply Chain Reliability: The synthetic pathway avoids the use of exotic or highly regulated reagents, relying instead on established industrial chemistry that can be easily replicated by multiple contract manufacturing organizations. This redundancy ensures supply continuity even if a single vendor faces disruptions. The thermal stability of the final products also implies a longer shelf life and reduced sensitivity to storage conditions, minimizing losses during logistics and warehousing compared to more fragile organic semiconductors.
- Scalability and Environmental Compliance: The reaction conditions, such as the use of glacial acetic acid and aqueous sodium carbonate, are amenable to large-scale batch processing in standard glass-lined reactors without requiring specialized high-pressure equipment. The ability to recrystallize the final products from common solvents like ethanol or toluene simplifies the purification train, reducing the volume of solvent waste generated. This aligns with increasingly strict environmental regulations in the chemical sector, facilitating smoother permitting processes for commercial scale-up facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the performance and handling of these carbazole derivatives, based on the experimental data provided in the patent documentation. Understanding these parameters is essential for integrating these materials into existing device architectures and quality control frameworks.
Q: What is the primary advantage of 1,3,6,8-tetraaryl substitution on carbazole?
A: The tetra-substitution at the 1,3,6,8 positions introduces significant steric hindrance that twists the molecular plane, effectively shortening the conjugation length and raising the triplet excited state energy to prevent concentration quenching.
Q: How does this material improve OLED device stability?
A: These derivatives exhibit exceptional thermal stability and solubility in common organic solvents like chloroform and toluene, allowing for uniform film formation via vacuum deposition or solution processing without degradation.
Q: What is the emission wavelength range of these carbazole derivatives?
A: The synthesized compounds emit in the near-ultraviolet to deep-blue region with a maximum emission wavelength between 390nm and 440nm, offering high color purity with a half-peak width of less than 50nm.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Derivative Supplier
As the demand for high-performance OLED materials continues to surge, partnering with an experienced chemical manufacturer is critical for securing a competitive edge in the display market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and HPLC profiling, to guarantee that every batch meets the exacting standards required for electronic grade applications.
We invite you to engage with our technical procurement team to discuss how this patented technology can be adapted to your specific product roadmap. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into potential process optimizations tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a seamless transition from laboratory innovation to mass market success.
