Scalable Synthesis of High-Efficiency Indolocarbazole Derivatives for Organic Photovoltaics
Scalable Synthesis of High-Efficiency Indolocarbazole Derivatives for Organic Photovoltaics
The rapid evolution of organic photovoltaics (OPV) has been significantly driven by the development of non-fullerene acceptors, with patent CN111018883B marking a pivotal advancement in this domain. This patent discloses a novel class of A-D-A type conjugated molecules based on an indolocarbazole derivative parent nucleus, designed to overcome the limitations of traditional materials. The core innovation lies in the strategic integration of strong electron-withdrawing end groups onto a multi-fused ring indolocarbazole backbone, resulting in materials that exhibit exceptional spectral absorption and optimized electrochemical energy levels. As a reliable organic photovoltaic material supplier, understanding the nuances of this synthesis is crucial for leveraging its potential in high-efficiency solar cell manufacturing. The disclosed molecules function effectively as either electron donors or acceptors, demonstrating power conversion efficiencies ranging from 10.5% to 18.5% when blended with polymer donors like PBDB-T.
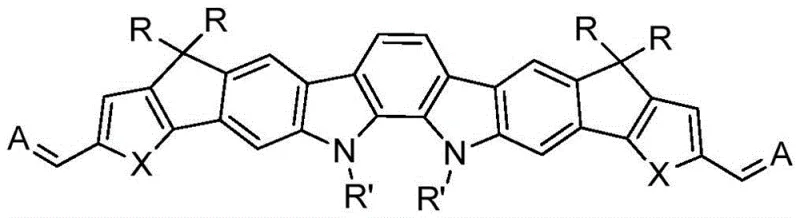
The structural versatility of these molecules allows for fine-tuning of their electronic properties, making them highly adaptable for various optoelectronic applications beyond just solar cells, including potential uses in organic field-effect transistors and photodetectors. The robust nature of the indolocarbazole core ensures thermal stability and morphological stability within the active layer of the device, which are critical factors for the long-term operational lifetime of organic solar modules. By focusing on this specific chemical architecture, manufacturers can access a new generation of materials that promise to lower the levelized cost of energy through improved performance metrics and solution-processable fabrication methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of organic solar cells was dominated by fullerene-based acceptors, which, despite their high electron mobility, suffered from significant drawbacks such as weak absorption in the visible region and limited tunability of energy levels. These conventional materials often required complex synthetic routes involving expensive precursors and harsh reaction conditions that were difficult to control on a large scale, leading to batch-to-batch variability and inconsistent device performance. Furthermore, the spherical geometry of fullerenes often resulted in poor morphological stability in blend films, causing phase separation over time and a consequent degradation of the photovoltaic device efficiency. The reliance on these legacy materials hindered the commercial viability of OPV technology, as the cost reduction in electronic chemical manufacturing was stifled by the high price and supply chain constraints associated with fullerene derivatives.
The Novel Approach
The novel approach presented in this patent utilizes a planar, fused-ring indolocarbazole core that facilitates strong intermolecular pi-pi interactions, thereby enhancing charge transport properties significantly compared to amorphous fullerene counterparts. By employing a straightforward Knoevenagel condensation reaction to attach electron-deficient end groups, the synthesis becomes much more accessible and controllable, allowing for precise modulation of the bandgap and highest occupied molecular orbital (HOMO) levels. This A-D-A architecture not only broadens the absorption spectrum to capture more solar energy but also improves the solubility of the molecules in common organic solvents like chloroform and chlorobenzene, facilitating solution processing. The result is a material system that combines the high efficiency of small molecule acceptors with the processability required for roll-to-roll manufacturing, representing a substantial leap forward in cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Knoevenagel Condensation for A-D-A Architecture
The core chemical transformation driving the formation of these high-performance conjugated molecules is the Knoevenagel condensation, a classic carbon-carbon bond-forming reaction that links the aldehyde-functionalized indolocarbazole core with active methylene compounds serving as electron-withdrawing units. In this specific application, the reaction proceeds under mild basic conditions using pyridine or sodium tert-butoxide in chloroform, where the base abstracts an acidic proton from the methylene group of the end group precursor, generating a nucleophilic carbanion. This nucleophile then attacks the carbonyl carbon of the aldehyde on the indolocarbazole dialdehyde, followed by dehydration to form the conjugated double bond that extends the pi-system across the entire molecule. The efficiency of this mechanism is evidenced by the high isolated yields reported in the examples, ranging from 70% to 87%, which indicates a clean reaction profile with minimal side products.
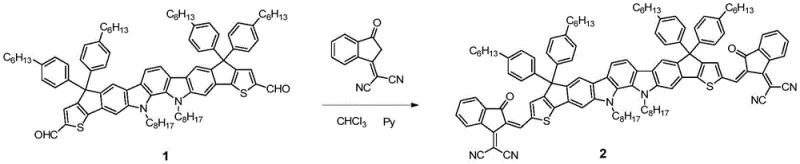
Controlling the purity of the final product is paramount for achieving high device efficiency, and the mechanistic pathway described allows for effective impurity management through standard purification techniques. The use of nitrogen protection during the reflux prevents oxidative degradation of the sensitive conjugated intermediates, ensuring that the final material possesses the intended electronic structure without defect sites that could act as charge traps. The planarity induced by the fusion of the rings in the indolocarbazole core, combined with the linear extension provided by the end groups, promotes self-assembly into ordered domains within the active layer. This structural order is critical for minimizing energy losses during charge separation and transport, directly correlating to the high fill factors and open-circuit voltages observed in the resulting photovoltaic devices.
How to Synthesize Indolocarbazole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced materials, emphasizing the importance of stoichiometric control and inert atmosphere conditions to maximize yield and purity. The process begins with the precise weighing of the indolocarbazole dialdehyde and the electron-withdrawing unit, typically in a molar ratio of 1:2 to ensure complete conversion of the dialdehyde functionality. Following the addition of the solvent and catalyst, the reaction mixture is subjected to rigorous deoxygenation to eliminate any dissolved oxygen that could interfere with the condensation or oxidize the product. Detailed standardized synthesis steps for replicating this high-yield process are provided in the guide below, ensuring that technical teams can implement this chemistry with confidence and reproducibility.
- Prepare the reaction vessel under nitrogen protection and add the indolocarbazole dialdehyde compound along with the electron-withdrawing unit A in a molar ratio of 1: 2 to 1:10.
- Add chloroform as the solvent and introduce pyridine or sodium tert-butoxide as the catalyst base, ensuring the mixture is thoroughly deoxygenated before heating.
- Heat the solution to reflux for 12 hours, then cool, extract, dry, concentrate, and purify via column chromatography to obtain the final conjugated molecule with high yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this indolocarbazole-based synthesis route offers compelling advantages rooted in the simplicity and scalability of the chemical process. The reliance on commercially available solvents like chloroform and common bases such as pyridine eliminates the need for exotic or hazardous reagents that often complicate logistics and increase safety compliance costs. Furthermore, the high reaction yields and the ability to purify the product using standard column chromatography suggest a streamlined downstream processing workflow that reduces waste generation and solvent consumption. These factors collectively contribute to a more resilient supply chain capable of meeting the growing demand for high-performance organic electronic materials without the bottlenecks associated with more complex synthetic pathways.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of ambient pressure reflux conditions significantly lower the operational expenditure associated with the production of these conjugated molecules. By avoiding the need for specialized equipment required for high-pressure or cryogenic reactions, manufacturers can utilize existing infrastructure to scale up production, thereby achieving substantial cost savings in capital investment and maintenance. The high atom economy of the condensation reaction further ensures that raw material costs are optimized, making the final product more competitive in the global market for organic photovoltaic materials.
- Enhanced Supply Chain Reliability: The starting materials, including the indolocarbazole dialdehydes and dicyanoindenone derivatives, are synthesized from readily available petrochemical feedstocks, reducing the risk of supply disruptions caused by scarce natural resources. This accessibility allows for the establishment of multiple sourcing channels for key precursors, enhancing the overall security of supply for downstream device manufacturers. Additionally, the stability of the intermediates and the final product under standard storage conditions simplifies inventory management and reduces the need for specialized cold-chain logistics, further strengthening the reliability of the supply network.
- Scalability and Environmental Compliance: The straightforward nature of the reaction workup, involving simple aqueous washes and drying steps, facilitates easy scale-up from gram-scale laboratory synthesis to kilogram or ton-scale industrial production. The process generates minimal hazardous waste compared to cross-coupling reactions that produce heavy metal salts, aligning well with increasingly stringent environmental regulations and sustainability goals. This environmental compatibility not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing operation, appealing to eco-conscious investors and customers in the renewable energy sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these indolocarbazole derivatives, providing clarity for stakeholders evaluating this technology. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating these materials into existing product lines or developing new applications in the field of organic electronics.
Q: What is the primary advantage of using indolocarbazole derivatives in organic solar cells?
A: Indolocarbazole derivatives offer a rigid, planar fused-ring structure that enhances intermolecular pi-pi stacking, leading to superior charge carrier mobility and broader spectral absorption compared to traditional fullerene acceptors.
Q: What are the typical reaction conditions for synthesizing these A-D-A molecules?
A: The synthesis typically involves a Knoevenagel condensation reaction in chloroform under reflux conditions for 12 hours, utilizing pyridine or sodium tert-butoxide as a base catalyst under a nitrogen atmosphere.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes common solvents like chloroform and standard purification techniques like column chromatography, with reported yields ranging from 70% to 87%, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolocarbazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the indolocarbazole-based conjugated molecules described in patent CN111018883B for the future of organic photovoltaics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global electronic materials market. We are committed to delivering high-purity indolocarbazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our expertise in process optimization allows us to refine the synthesis further, potentially improving yields and reducing costs even beyond the baseline established in the patent literature.
We invite you to collaborate with us to explore how these advanced materials can enhance your product portfolio and drive innovation in your organic solar cell projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing planning, ensuring a seamless transition from laboratory concept to commercial reality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
