Advanced N-Methylation Strategy for High-Purity Dextromethorphan Commercial Manufacturing
Advanced N-Methylation Strategy for High-Purity Dextromethorphan Commercial Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency with stringent safety profiles, particularly for central antitussive agents like Dextromethorphan. A pivotal advancement in this domain is detailed in patent CN103073496A, which discloses a novel preparation method for Dextromethorphan (I) that fundamentally alters the traditional synthetic logic. Instead of relying on the historically problematic final O-methylation step, this innovation utilizes a direct N-methylation strategy starting from the intermediate ent-3-methoxymorphinan (II). This approach not only simplifies the operational workflow but also addresses critical quality attributes regarding genotoxic impurities. For R&D directors and procurement specialists alike, understanding this shift is crucial for optimizing the supply chain of reliable pharmaceutical intermediates. The method leverages standard reductive amination techniques, offering a pathway that is both economically viable and chemically elegant, ensuring that the final bulk drug meets or exceeds international pharmacopoeia standards.
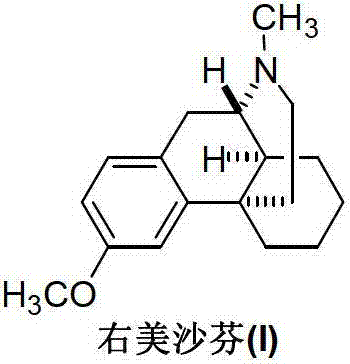
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dextromethorphan Hydrobromide has relied on a multi-step sequence originating from optically pure isoquinoline derivatives. As illustrated in the traditional pathway, the process involves N-formylation, reduction to an N-methyl intermediate, acidic cyclization to form the phenolic morphinan skeleton, and finally, an O-methylation reaction to install the crucial methoxy group. This final O-methylation step presents significant technical hurdles; commonly used methylating agents such as dimethyl sulfate or methyl iodide often induce unwanted N-methylation side reactions, leading to the formation of quaternary ammonium salt byproducts. To mitigate this, manufacturers are forced to employ expensive and difficult-to-source reagents like trimethylphenyl ammonium hydroxide in mixed solvent systems. Furthermore, this conventional route is plagued by the generation of dimethylamine (DMA), a known genotoxic impurity whose residual control is strictly mandated by the USP, EP, JP, and Chinese Pharmacopoeia. The combination of low yields—often hovering around 50% in the final step—and the high cost of selectivity-controlling reagents creates a substantial bottleneck in cost reduction in pharmaceutical intermediates manufacturing.
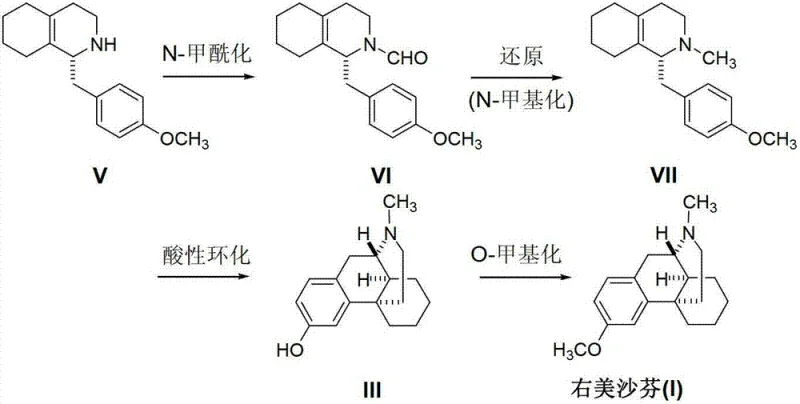
The Novel Approach
In stark contrast to the convoluted traditional pathway, the method disclosed in patent CN103073496A proposes a streamlined synthesis that bypasses the problematic phenolic intermediate entirely. By starting with ent-3-methoxymorphinan (II), which already possesses the requisite methoxy functionality, the synthesis reduces to a single, highly efficient N-methylation transformation. This strategic pivot eliminates the need for harsh O-methylation conditions and the associated risk of quaternary ammonium salt formation. The new route employs a classic Eschweiler-Clarke type reaction or its variants, utilizing accessible formylation reagents like formaldehyde or alkyl formates followed by reduction. This not only drastically simplifies the process flow but also inherently improves the impurity profile by avoiding the generation of genotoxic DMA residues associated with older methylation protocols. For supply chain heads, this represents a move towards commercial scale-up of complex pharmaceutical intermediates that is far more predictable and less prone to regulatory setbacks regarding impurity limits.
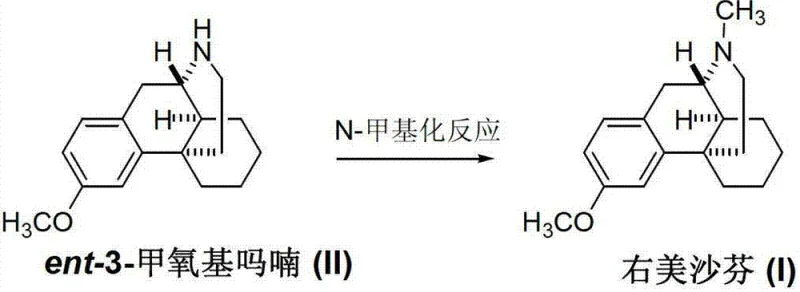
Mechanistic Insights into Reductive N-Methylation
The core of this technological breakthrough lies in the mechanistic elegance of the reductive N-methylation applied to the morphinan nitrogen. The reaction initiates with the condensation of the secondary amine in ent-3-methoxymorphinan with a carbonyl source, such as formaldehyde or methyl formate, to form an iminium ion or a formamide intermediate. In the presence of a reducing agent, this intermediate is subsequently converted into the tertiary amine found in Dextromethorphan. The patent details several viable reduction strategies, including catalytic hydrogenation using Raney nickel or palladium on carbon, as well as hydride reductions using sodium borohydride. This flexibility allows manufacturers to tailor the process based on available infrastructure; for instance, catalytic hydrogenation offers a green chemistry advantage by producing water as the only byproduct, while borohydride reduction provides a straightforward batch process option. The stereochemical integrity of the morphinan skeleton is preserved throughout this mild transformation, ensuring that the optical purity required for the dextrorotatory isomer is maintained without racemization risks often seen in harsher alkylation conditions.
From an impurity control perspective, this mechanism is superior because it avoids the nucleophilic attack on oxygen that characterizes the old O-methylation route. In the conventional method, the phenoxide anion competes with the amine nitrogen, necessitating complex protection-deprotection strategies or highly selective reagents. By reversing the order of operations—installing the methoxy group prior to the final nitrogen methylation—the new process ensures that the nitrogen is the sole reactive site for the final transformation. This specificity significantly reduces the burden on downstream purification processes. Consequently, the resulting crude product requires less intensive workup to meet the rigorous specifications for high-purity pharmaceutical intermediates, directly translating to lower solvent consumption and waste generation. The ability to control the reaction stoichiometry precisely further minimizes the formation of over-alkylated quaternary salts, a persistent issue in the legacy manufacturing processes.
How to Synthesize Dextromethorphan Efficiently
Implementing this patented methodology requires careful attention to reaction parameters to maximize yield and purity. The patent provides three distinct embodiments that serve as a robust foundation for process development. Whether utilizing formaldehyde with catalytic hydrogenation or methyl formate with sodium borohydride, the general protocol involves mixing the ent-3-methoxymorphinan starting material with the formylating agent in a protic solvent like methanol or ethanol. The reaction mixture is then subjected to reduction conditions, either under hydrogen pressure with a metal catalyst or via chemical reduction at controlled temperatures. Following the reaction, standard workup procedures such as filtration to remove catalysts, concentration under reduced pressure, and recrystallization from methanol yield the final off-white solid product. For a detailed breakdown of the specific molar ratios, temperatures, and reaction times optimized in the patent examples, please refer to the standardized guide below.
- React ent-3-methoxymorphinan with a formylation reagent such as formaldehyde, paraformaldehyde, or alkyl formate in a suitable solvent like methanol or ethanol.
- Perform a reduction reaction on the intermediate formamide using sodium borohydride, potassium borohydride, or catalytic hydrogenation with Raney nickel or palladium catalysts.
- Isolate the final Dextromethorphan product through filtration, concentration, and recrystallization from methanol to achieve high purity off-white solids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this N-methylation route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the elimination of expensive and hard-to-source selectivity controlling reagents like trimethylphenyl ammonium hydroxide, which were mandatory in the old O-methylation process. By replacing these niche chemicals with commodity reagents such as formaldehyde, paraformaldehyde, or methyl formate, the raw material cost structure is significantly optimized. Furthermore, the avoidance of genotoxic impurities like DMA reduces the risk of batch rejection and the need for costly analytical testing and remediation, thereby enhancing overall supply chain reliability. This process stability ensures that production schedules are not disrupted by the scarcity of specialized reagents or the failure to meet stringent regulatory limits on toxic residues.
- Cost Reduction in Manufacturing: The new process achieves a dramatic reduction in manufacturing costs by substituting high-priced specialty reagents with inexpensive, bulk-available commodities. The yield improvement from approximately 50% in the conventional final step to over 80% in the new method effectively doubles the output per unit of starting material, drastically lowering the cost of goods sold. Additionally, the simplified workup procedure reduces solvent usage and energy consumption associated with extensive purification steps required to remove quaternary ammonium byproducts. These cumulative efficiencies result in substantial cost savings without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like formaldehyde and standard catalysts like Raney nickel mitigates the risk of supply disruptions often associated with proprietary or niche chemical suppliers. The robustness of the reaction conditions allows for flexible sourcing of raw materials, ensuring continuous production even during market fluctuations. Moreover, the consistent quality of the product, free from difficult-to-control genotoxic impurities, guarantees a steady supply of compliant material for downstream formulation, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as hydrogenation and crystallization that are common in existing pharmaceutical manufacturing facilities. The reduction in hazardous waste generation, particularly the elimination of genotoxic DMA residues, aligns with increasingly strict environmental regulations and corporate sustainability goals. This ease of scale-up facilitates the transition from pilot plant to commercial production, enabling rapid response to market demand for antitussive medications while maintaining a smaller environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in patent CN103073496A, providing a clear understanding of the process capabilities and advantages. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: How does the new N-methylation route improve product safety compared to traditional methods?
A: The novel route avoids the use of methylating agents that generate genotoxic dimethylamine (DMA) byproducts, ensuring compliance with strict pharmacopoeia residual limits.
Q: What yields can be expected from the patented N-methylation process?
A: Experimental embodiments demonstrate yields ranging from 79.6% to 87.8%, significantly outperforming the approximately 50% yield of the conventional O-methylation final step.
Q: Is this process scalable for industrial API production?
A: Yes, the process utilizes common reagents like formaldehyde and standard reduction catalysts like Raney nickel, making it highly suitable for large-scale commercial manufacturing without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dextromethorphan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the N-methylation route described in CN103073496A for the production of high-quality Dextromethorphan. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of reductive amination and catalytic hydrogenation, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product consistency. We are committed to delivering API intermediates that not only meet but exceed global pharmacopoeia standards, providing our partners with a secure and compliant supply base.
We invite pharmaceutical companies and chemical distributors to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of efficiency and quality in the industry.
