Advanced Reductive Alkylation Technology for High-Purity Glyphosate and Derivatives Manufacturing
Advanced Reductive Alkylation Technology for High-Purity Glyphosate and Derivatives Manufacturing
The global demand for effective broad-spectrum herbicides continues to drive innovation in agrochemical intermediate manufacturing. Patent CN85109397A introduces a transformative methodology for synthesizing N-phosphonomethylglycine, commonly known as glyphosate, and its various derivatives through a highly selective reductive alkylation process. Unlike traditional routes that rely on harsh oxidation steps or struggle with poor selectivity, this invention leverages the condensation of glycine precursors with formylphosphonic acid in an aqueous medium. This technical breakthrough addresses long-standing challenges in impurity profiles and reaction efficiency, offering a robust pathway for producing high-purity agrochemical intermediates. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is crucial for evaluating next-generation supply chain strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glyphosate has been plagued by significant chemical inefficiencies and safety concerns inherent to earlier patented processes. For instance, methods described in U.S. Patent 3,969,398 involve the oxidation of N-phosphonomethyliminodiacetic acid, a step that introduces complexity in waste management and requires stringent control over oxidizing agents. Furthermore, prior art regarding the reductive alkylation of amines, such as the work by Kutani and Bowman, demonstrated that reacting primary amines with straight-chain aldehydes typically resulted in substantial dialkylation rather than the desired monoalkylation. This lack of selectivity leads to complex mixture profiles containing di-substituted byproducts, necessitating expensive and energy-intensive purification steps to isolate the target secondary amine. Additionally, many conventional condensation reactions require organic solvents like benzene or toluene, posing severe environmental and occupational health risks that modern regulatory frameworks increasingly penalize.
The Novel Approach
The methodology disclosed in CN85109397A fundamentally shifts the paradigm by utilizing formylphosphonic acid or its acetals as the carbonyl source in an aqueous environment. This approach capitalizes on the unique reactivity of the phosphono-aldehyde group to achieve high conversion rates to the monoalkylated product with minimal dialkylation. By conducting the reaction in water, the process inherently aligns with green chemistry principles, eliminating the need for hazardous organic solvents and simplifying the downstream workup. The patent highlights that maintaining the reaction pH between 5.5 and 12.0, preferably 7.0 to 12.0, is critical for shifting the equilibrium towards the desired imine intermediate without degrading the sensitive phosphonic acid moiety. This precise control allows manufacturers to achieve yields exceeding 90% under optimized conditions, drastically reducing the burden on purification infrastructure and enhancing the overall economic viability of large-scale production.
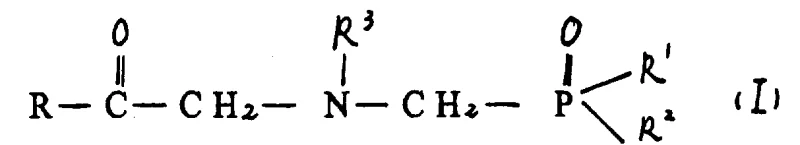
Mechanistic Insights into Aqueous Reductive Alkylation
The core of this technology lies in the controlled condensation of an amine precursor, such as glycine, with a carbonyl compound like formylphosphonic acid. As illustrated in the structural definitions, the amine reactant (Formula II) undergoes nucleophilic attack on the carbonyl carbon of the phosphono-aldehyde (Formula III or IV). This initial step forms an aldimine intermediate, which is unstable and exists in equilibrium with the starting materials. The novelty of this process is the immediate in situ reduction of this imine bond before it can react further with another equivalent of the carbonyl compound, which would lead to the undesired tertiary amine. The aqueous medium plays a dual role here: it solubilizes the zwitterionic nature of glycine and stabilizes the transition states through hydrogen bonding, while the specific pH adjustment ensures the amine is sufficiently nucleophilic yet not so basic as to trigger Cannizzaro-type side reactions of the aldehyde.
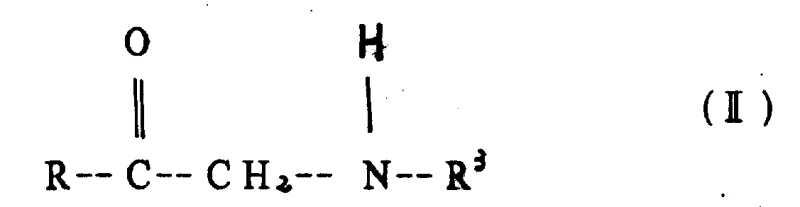
Following the condensation, the reduction step is executed either via catalytic hydrogenation or electrochemical means. In catalytic hydrogenation, transition metals like palladium on carbon or Raney nickel facilitate the heterolytic cleavage of molecular hydrogen, transferring hydride equivalents to the carbon-nitrogen double bond of the imine. Alternatively, the patent describes an electrochemical pathway where the imine is reduced at a cathode with high hydrogen overpotential, such as mercury or lead. This electrochemical option is particularly significant for industrial scale-up as it removes the requirement for high-pressure hydrogen gas infrastructure, replacing it with electrical current control. The ability to tune the reduction potential allows for exquisite selectivity, ensuring that other functional groups on the molecule, such as the carboxylic acid or phosphonic acid moieties, remain intact during the transformation.
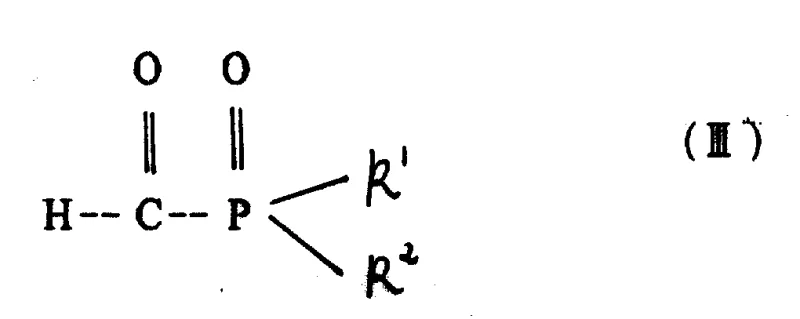
Impurity control is intrinsically built into the reaction kinetics defined by this patent. By avoiding the isolation of the imine intermediate, the process minimizes exposure to conditions that might promote hydrolysis or polymerization. The data indicates that operating at a pH above 12.0 or below 5.5 significantly degrades selectivity, likely due to the protonation state of the amine nitrogen affecting its nucleophilicity or the stability of the aldehyde. Therefore, rigorous pH monitoring and control systems are essential process analytical technology (PAT) tools for any facility adopting this route. The result is a crude reaction mixture with a high assay of the target N-phosphonomethylglycine, simplifying the final crystallization or salt formation steps required to meet commercial specifications for agrochemical active ingredients.
How to Synthesize N-Phosphonomethylglycine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and physical parameters outlined in the patent examples. The process begins with dissolving the amine and carbonyl precursors in water, followed by precise pH adjustment using bases like sodium hydroxide. Detailed standardized synthetic steps see the guide below.
- Prepare an aqueous reaction medium containing glycine (or its salt/ester) and formylphosphonic acid (or its acetal/hydrate) with a molar ratio of carbonyl to amine between 1.0 and 1.2.
- Adjust the pH of the reaction mixture to a range between 7.0 and 12.0 using alkali metal hydroxides or amines to optimize condensation equilibrium and prevent side reactions.
- Perform reduction via catalytic hydrogenation using Pd/C or Raney Nickel, or alternatively utilize electrochemical reduction in a divided cell to yield the final N-phosphonomethylglycine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this reductive alkylation technology offers profound strategic benefits beyond mere chemical elegance. The shift to an aqueous-based system fundamentally alters the cost structure of manufacturing by removing the dependency on volatile organic solvents. This elimination not only reduces raw material expenditure but also drastically cuts down on the capital and operational costs associated with solvent recovery distillation columns and explosion-proof infrastructure. Furthermore, the high selectivity of the reaction means that less starting material is wasted on forming difficult-to-remove dialkylated byproducts, directly improving the atom economy and reducing the cost of goods sold (COGS) for the final herbicide active ingredient.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive organic solvents and reduces the load on waste treatment facilities. By achieving high conversion rates in water, manufacturers can avoid the complex extraction and purification sequences typical of older methods. This streamlined workflow translates to significant operational savings, as energy consumption for heating and cooling is optimized, and the throughput of existing reactor vessels can be increased due to shorter cycle times and simplified workup procedures.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like glycine and formylphosphonic acid derivatives ensures a stable supply of raw materials. Unlike specialized reagents that may have single-source bottlenecks, the inputs for this process are widely available in the global chemical market. Additionally, the robustness of the aqueous system allows for flexible manufacturing across different geographic locations without requiring highly specialized containment systems, thereby diversifying supply risk and ensuring continuity of supply for critical agrochemical intermediates.
- Scalability and Environmental Compliance: The ability to use electrochemical reduction provides a scalable alternative to high-pressure hydrogenation, which often faces regulatory hurdles in densely populated industrial zones. This flexibility allows companies to adapt their production technology to local safety regulations more easily. Moreover, the reduced generation of hazardous waste aligns with increasingly stringent environmental, social, and governance (ESG) mandates, protecting the company from future regulatory liabilities and enhancing its reputation as a sustainable manufacturer in the eyes of global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance.
Q: How does this reductive alkylation method improve selectivity compared to conventional alkylation?
A: Conventional methods often suffer from substantial dialkylation when reacting primary amines with aldehydes. This patented process utilizes specific pH control (5.5-12.0) and aqueous conditions to favor the formation of the monoalkylated imine intermediate, which is then selectively reduced to the secondary amine, minimizing di-substituted byproducts.
Q: What are the advantages of using an aqueous medium for glyphosate synthesis?
A: Using water as the primary solvent eliminates the need for volatile organic compounds (VOCs) like benzene or toluene used in older condensation protocols. This significantly reduces environmental hazards, simplifies waste treatment, and lowers raw material costs associated with solvent recovery and disposal.
Q: Can electrochemical reduction replace catalytic hydrogenation in this process?
A: Yes, the patent explicitly describes electrochemical reduction as a viable alternative. Using a cathode with high hydrogen overpotential (like mercury or lead) in a divided cell allows for the reduction of the imine intermediate without requiring high-pressure hydrogen gas, offering a different safety and scalability profile for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glyphosate Supplier
As the agrochemical industry evolves towards more sustainable and efficient manufacturing practices, partnering with a technically proficient CDMO is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this reductive alkylation method can be successfully translated to industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of N-phosphonomethylglycine meets the exacting standards required for formulation into effective herbicides.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your operation. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organic synthesis can drive value and reliability for your agrochemical portfolio.
