Advanced Synthesis of N-Phosphonomethylglycine: A Scalable Route for Global Herbicide Production
The global demand for high-purity agrochemical intermediates continues to drive innovation in process chemistry, particularly for widely used herbicides like glyphosate. Patent CN1239507C introduces a groundbreaking method for producing N-phosphonomethylglycine that addresses critical inefficiencies found in legacy manufacturing protocols. This technology leverages the reaction between specific hexahydrotriazine compounds and triacyl phosphites within an organic solvent matrix, followed by a strategic extraction and saponification sequence. The core innovation lies in the separation of the phosphono compound into an aqueous phase before hydrolysis, a step that fundamentally alters the economic and environmental profile of the synthesis. By preventing the decomposition of the organic solvent during the harsh saponification stage, this process offers a reliable agrochemical intermediate supplier pathway that significantly reduces waste and raw material consumption. For R&D directors and procurement managers alike, understanding this shift from traditional acid halide methods to triacyl phosphite chemistry is essential for optimizing supply chains and reducing the total cost of ownership in herbicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-phosphonomethylglycine has relied heavily on processes that involve significant safety hazards and economic inefficiencies, particularly those utilizing acid halides and phosphorous diesters. As illustrated in prior art such as EP-A-104 775, conventional routes often require the use of phosgene, a highly toxic gas that necessitates specialized containment infrastructure and rigorous safety protocols, thereby inflating capital expenditure. Furthermore, these traditional methods frequently employ expensive phosphorous diesters to achieve acceptable selectivity, which directly impacts the raw material cost structure. A critical technical failure in these older processes is the instability of chlorinated hydrocarbon solvents, such as 1,2-dichloroethane, under the hydrolytic conditions required to liberate the final product. When subjected to the high temperatures and acidic or basic environments needed for saponification, these solvents tend to decompose into glycols and hydrochloric acid, leading to substantial solvent loss and the generation of difficult-to-treat wastewater streams. 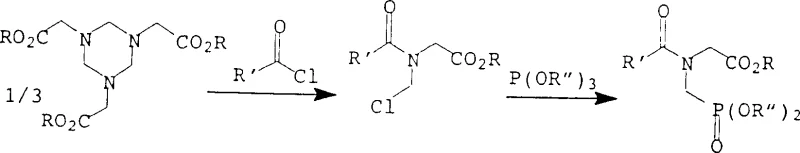
The Novel Approach
In stark contrast to the limitations of phosgene-based chemistry, the method disclosed in CN1239507C utilizes a triacyl phosphite reagent that reacts efficiently with hexahydrotriazine derivatives to form a stable phosphono intermediate. This novel approach eliminates the need for toxic phosgene entirely, replacing it with safer and more cost-effective acylating agents that can be derived from common carboxylic acids. The true brilliance of this工艺 lies in the sequential processing steps where the reaction mixture is extracted with water or an aqueous solution immediately after the phosphorylation step. This extraction transfers the valuable phosphono compound into the aqueous phase while leaving the organic solvent behind, effectively decoupling the product isolation from the solvent exposure to harsh hydrolysis conditions. Consequently, the organic solvent remains intact and can be recycled repeatedly without the degradation issues plaguing conventional methods, offering a sustainable solution for cost reduction in herbicide manufacturing. This shift not only simplifies the purification workflow but also ensures a consistent supply of high-purity intermediates by minimizing side reactions associated with solvent breakdown.
Mechanistic Insights into Triacyl Phosphite-Mediated Phosphorylation
The chemical mechanism underpinning this invention centers on the nucleophilic attack of the hexahydrotriazine nitrogen atoms on the phosphorus center of the triacyl phosphite. The starting material, a hexahydrotriazine derivative of Formula II, serves as a robust scaffold that protects the reactive amine functionalities until the precise moment of phosphorylation. 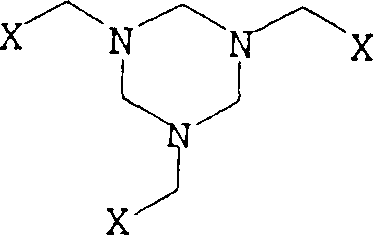 When reacted with a triacyl phosphite of Formula III in an inert organic solvent, the system forms a new phosphorus-nitrogen bond, yielding the intermediate compound of Formula I. This reaction proceeds rapidly, typically completing within 10 to 30 minutes at temperatures ranging from room temperature to 100°C, demonstrating excellent kinetic efficiency. The choice of the acyl group on the phosphite is critical, as it acts as a protecting group that stabilizes the phosphonate moiety during the initial reaction phase but is readily cleavable during the subsequent hydrolysis. The use of aryl groups, such as phenyl or p-tolyl, on the triacyl phosphite is particularly advantageous due to their electronic properties which facilitate both the formation of the P-N bond and the eventual hydrolytic cleavage. This mechanistic pathway ensures high selectivity for the mono-phosphonated product, minimizing the formation of bis-phosphonomethylated by-products that often complicate purification in other synthetic routes.
When reacted with a triacyl phosphite of Formula III in an inert organic solvent, the system forms a new phosphorus-nitrogen bond, yielding the intermediate compound of Formula I. This reaction proceeds rapidly, typically completing within 10 to 30 minutes at temperatures ranging from room temperature to 100°C, demonstrating excellent kinetic efficiency. The choice of the acyl group on the phosphite is critical, as it acts as a protecting group that stabilizes the phosphonate moiety during the initial reaction phase but is readily cleavable during the subsequent hydrolysis. The use of aryl groups, such as phenyl or p-tolyl, on the triacyl phosphite is particularly advantageous due to their electronic properties which facilitate both the formation of the P-N bond and the eventual hydrolytic cleavage. This mechanistic pathway ensures high selectivity for the mono-phosphonated product, minimizing the formation of bis-phosphonomethylated by-products that often complicate purification in other synthetic routes.
Following the formation of the intermediate Formula I, the process employs a sophisticated phase-transfer strategy to isolate the product while preserving solvent integrity.  During the extraction step, the reaction mixture is contacted with water or an aqueous acid/base solution, causing the polar phosphono compound to migrate into the aqueous phase. This step is remarkably fast, with over 95% of the phosphorus transferring to the water phase within just 10 minutes at elevated temperatures. Once separated, the aqueous phase containing the intermediate is subjected to hydrolysis under neutral or basic conditions, which cleaves the acyl protecting groups to reveal the free phosphonic acid functionality. Because the organic solvent has already been removed, the hydrolysis can be conducted at higher temperatures (130-150°C) and pressures without fear of solvent degradation, leading to reaction times as short as 20 minutes. This mechanistic decoupling of reaction and hydrolysis phases is the key to achieving yields higher than 90% while maintaining the purity specifications required for downstream agricultural applications. The resulting N-phosphonomethylglycine can then be precipitated by pH adjustment, yielding a product with minimal inorganic salt contamination compared to acid-hydrolysis methods.
During the extraction step, the reaction mixture is contacted with water or an aqueous acid/base solution, causing the polar phosphono compound to migrate into the aqueous phase. This step is remarkably fast, with over 95% of the phosphorus transferring to the water phase within just 10 minutes at elevated temperatures. Once separated, the aqueous phase containing the intermediate is subjected to hydrolysis under neutral or basic conditions, which cleaves the acyl protecting groups to reveal the free phosphonic acid functionality. Because the organic solvent has already been removed, the hydrolysis can be conducted at higher temperatures (130-150°C) and pressures without fear of solvent degradation, leading to reaction times as short as 20 minutes. This mechanistic decoupling of reaction and hydrolysis phases is the key to achieving yields higher than 90% while maintaining the purity specifications required for downstream agricultural applications. The resulting N-phosphonomethylglycine can then be precipitated by pH adjustment, yielding a product with minimal inorganic salt contamination compared to acid-hydrolysis methods.
How to Synthesize N-Phosphonomethylglycine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the triacyl phosphite and the precise control of the extraction parameters to maximize yield and solvent recovery. The process begins with the preparation of the triacyl phosphite, which can be generated in situ from carboxylic acids and phosphorus trichloride, allowing for the recycling of acyl groups and further reducing raw material costs. Once the phosphorylation of the hexahydrotriazine is complete, the critical extraction step must be optimized for temperature and pH to ensure complete transfer of the intermediate into the aqueous layer. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and agitation speeds, are essential for replicating the high efficiency reported in the patent data. For technical teams looking to scale this process, understanding the interplay between the solubility of the intermediate and the phase separation dynamics is paramount to achieving consistent batch quality.
- React a hexahydrotriazine derivative (Formula II) with a triacyl phosphite (Formula III) in an inert organic solvent such as 1,2-dichloroethane to form the phosphono compound (Formula I).
- Extract the resulting reaction mixture with water or an aqueous solution to transfer the valuable phosphono compound into the aqueous phase, separating it from the organic solvent.
- Hydrolyze the product contained in the aqueous phase under neutral or basic conditions to obtain N-phosphonomethylglycine, avoiding the decomposition of the organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible improvements in operational efficiency and risk mitigation across the manufacturing value chain. The elimination of phosgene from the process removes a major regulatory and safety burden, simplifying facility compliance and reducing insurance costs associated with handling toxic gases. Furthermore, the ability to recycle the organic solvent, such as 1,2-dichloroethane, without significant degradation means that solvent purchase volumes can be drastically reduced, leading to substantial cost savings over the lifecycle of the plant. The use of triacyl phosphites, which can be synthesized from commodity carboxylic acids, offers a more stable and predictable pricing model compared to the volatile market for specialized phosphorous diesters. Additionally, the high selectivity of the reaction minimizes the formation of impurities, reducing the load on downstream purification units and decreasing the volume of waste solvent that requires disposal. These factors combined create a robust supply chain framework that is less susceptible to raw material shortages and regulatory changes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive reagents and the optimization of solvent usage. By utilizing triacyl phosphites instead of costly phosphorous diesters, the direct material cost per kilogram of product is significantly lowered. Moreover, the acyl groups used in the triacyl phosphite act as recyclable protecting groups; upon hydrolysis, the resulting carboxylic acids can be recovered and reused to synthesize fresh triacyl phosphite, creating a closed-loop system that minimizes waste. The avoidance of solvent decomposition means that the expensive chlorinated hydrocarbons do not need to be constantly replenished, further driving down operational expenditures. This comprehensive approach to cost management ensures that the manufacturing process remains competitive even in fluctuating market conditions.
- Enhanced Supply Chain Reliability: From a logistics perspective, this method enhances reliability by relying on widely available raw materials such as formaldehyde, ammonia, and common carboxylic acids, rather than specialized or hazardous reagents like phosgene. The simplified safety profile allows for more flexible transportation and storage options, reducing lead times for high-purity agrochemical intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without compromising yield, ensures consistent production output even if minor variations occur in utility supplies. This stability is crucial for maintaining long-term contracts with global herbicide manufacturers who require uninterrupted supply to meet seasonal agricultural demand. The ability to operate continuously or in large batch modes further supports the scaling of production to meet growing market needs.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than conventional methods, aligning with increasingly stringent global regulations on industrial emissions and waste. The process generates primarily inorganic chlorides as waste, which are easier to treat and dispose of compared to the complex organic mixtures produced by phosgene-based routes. The high atom economy of the reaction, coupled with the recycling of solvents and acyl groups, results in a greener manufacturing profile that appeals to environmentally conscious stakeholders. Scalability is facilitated by the use of standard reactor types such as stirred tanks and extraction columns, which are readily available in existing chemical infrastructure. This ease of scale-up allows manufacturers to rapidly increase capacity from pilot scales to hundreds of metric tons annually without requiring extensive re-engineering of the process flow.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for N-phosphonomethylglycine. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific production requirements.
Q: How does this process prevent organic solvent decomposition?
A: The process uniquely extracts the valuable phosphono compound into an aqueous phase prior to the hydrolysis step. Since hydrolysis typically requires harsh conditions that decompose chlorinated solvents like 1,2-dichloroethane, removing the product first allows the solvent to be recovered and recycled without degradation.
Q: Does this method require the use of phosgene?
A: No, unlike conventional methods described in prior art such as EP-A-104 775, this novel approach utilizes triacyl phosphites and hexahydrotriazine derivatives, completely eliminating the need for toxic and costly phosgene reagents.
Q: What yields can be expected from this synthesis route?
A: The patent data indicates that this method achieves yields higher than 90% for N-phosphonomethylglycine, with very short reaction times ranging from 10 to 30 minutes for the initial phosphorylation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phosphonomethylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN1239507C for the global agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-phosphonomethylglycine adheres to the highest international standards. We are committed to leveraging advanced chemical technologies to drive efficiency and sustainability for our partners, making us the preferred choice for companies seeking a reliable N-Phosphonomethylglycine supplier. Our team of expert chemists and engineers works collaboratively with clients to optimize process parameters and ensure seamless technology transfer from development to full-scale manufacturing.
We invite procurement leaders and technical directors to engage with us to explore how this innovative synthesis method can enhance your supply chain resilience and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your operation. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project needs. Together, we can build a sustainable and efficient supply chain that supports the growing demand for high-performance herbicides worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
