Advanced Rhodium-Catalyzed Synthesis of Carbazole Derivatives for Commercial Scale-Up
Introduction to Next-Generation Carbazole Manufacturing
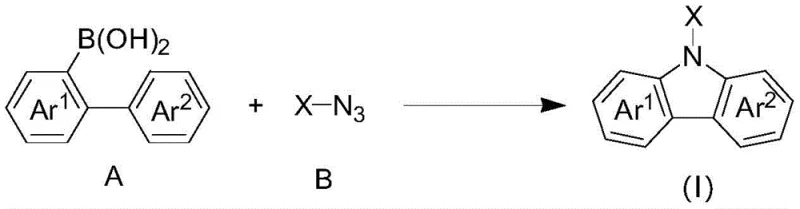
The escalating demand for high-performance nitrogen-containing fused-ring compounds in the pharmaceutical and optoelectronic sectors necessitates innovative synthetic methodologies that balance efficiency with environmental sustainability. Patent CN108658841B introduces a transformative approach to synthesizing carbazole-based compounds, leveraging a dual catalytic system involving transition metal rhodium and silver oxidants. This technology addresses critical bottlenecks in traditional manufacturing by enabling the direct conversion of polysubstituted biarylboronic acids and azides into complex carbazole scaffolds under remarkably mild conditions. Unlike legacy processes that often require extreme temperatures or hazardous oxidants, this novel pathway operates effectively between -10°C and 100°C, significantly reducing energy consumption and safety risks associated with high-pressure reactors. The versatility of this method allows for the incorporation of diverse functional groups, including halogens, alkyls, and electron-withdrawing moieties, making it an invaluable tool for producing high-purity pharmaceutical intermediates and advanced OLED materials.
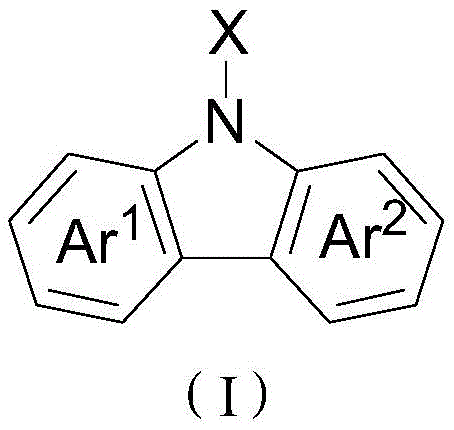
Furthermore, the strategic design of this synthesis route minimizes the number of discrete operational steps, thereby enhancing overall process throughput and reducing waste generation. By utilizing readily available starting materials such as biarylboronic acids, the method circumvents the need for expensive and difficult-to-handle amine sources typically required in classical cyclization reactions. This shift not only streamlines the supply chain for raw materials but also improves the economic viability of producing specialized carbazole derivatives on a multi-ton scale. For R&D directors and process chemists, understanding the nuances of this rhodium-mediated transformation is essential for developing robust manufacturing protocols that meet stringent purity specifications while maintaining cost competitiveness in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of carbazole derivatives has relied heavily on methods that impose severe constraints on process flexibility and operational safety. Traditional routes often involve the dehydrogenation or deamination of 2-aminobiphenyl precursors under high-temperature conditions with strong oxidants, which can lead to significant thermal degradation of sensitive functional groups and the formation of complex impurity profiles. Alternatively, multi-step sequences utilizing Suzuki-Miyaura cross-coupling followed by cyclization require rigorous pre-functionalization of substrates, increasing both the material cost and the environmental footprint due to additional purification stages. These conventional approaches frequently demand strictly anhydrous and oxygen-free environments, necessitating specialized equipment and inert gas handling systems that drive up capital expenditure and operational complexity. Moreover, the limited substrate scope of older catalytic systems often restricts the ability to introduce diverse substituents at specific positions on the carbazole core, hindering the rapid optimization of structure-activity relationships for new drug candidates or electronic materials.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a synergistic rhodium and silver catalytic cycle that facilitates the direct assembly of the carbazole skeleton from biarylboronic acids and azides without extensive pre-modification. This innovative strategy operates under significantly milder thermal conditions, typically between 75°C and 85°C, which preserves the integrity of thermally labile substituents and reduces the risk of runaway exothermic reactions. The use of azides as nitrogen sources allows for a more atom-economical construction of the central pyrrole ring, bypassing the need for separate amination steps that often suffer from poor regioselectivity. Additionally, the reaction demonstrates exceptional tolerance towards a wide array of functional groups, including nitriles, halogens, and trifluoromethyl groups, enabling the synthesis of highly substituted derivatives in a single pot. This streamlined workflow not only accelerates the timeline from laboratory discovery to pilot plant validation but also simplifies downstream processing by minimizing the formation of intractable side products.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The core of this technological breakthrough lies in the sophisticated interplay between the rhodium catalyst and the silver oxidant, which orchestrates a cascade of bond-forming events to construct the rigid carbazole framework. The reaction initiates with the activation of the biarylboronic acid by the rhodium species, likely forming a reactive organometallic intermediate that is poised for subsequent transformation. Concurrently, the azide component undergoes decomposition or activation, potentially generating a nitrene-like species that serves as the nitrogen donor for the cyclization process. The presence of the silver salt is critical, acting as a stoichiometric oxidant to regenerate the active rhodium catalyst and drive the equilibrium towards product formation. This dual-catalyst system ensures high turnover numbers and sustained catalytic activity throughout the reaction duration, which is typically maintained for 2 to 24 hours depending on the steric bulk of the substrates.
From an impurity control perspective, the mechanism offers distinct advantages by avoiding radical pathways that often lead to polymerization or uncontrolled coupling byproducts. The mild basic conditions employed, utilizing reagents like sodium carbonate, help neutralize acidic byproducts without promoting hydrolysis of sensitive esters or amides present on the aromatic rings. Furthermore, the choice of solvent, preferably 1,4-dioxane, provides an optimal polarity balance that solubilizes both organic substrates and inorganic salts, ensuring homogeneous reaction kinetics. Understanding these mechanistic details allows process engineers to fine-tune parameters such as catalyst loading, which can be as low as 0.001 to 5 mol%, to achieve maximum efficiency while minimizing residual metal content in the final API intermediate. This level of control is paramount for meeting regulatory guidelines regarding heavy metal residues in pharmaceutical substances.
How to Synthesize Carbazole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and mixing protocols to ensure consistent batch-to-batch reproducibility. The process begins with the precise weighing of the biarylboronic acid and the specific azide derivative, followed by the addition of the rhodium catalyst, such as pentamethylcyclopentadiene rhodium chloride dimer, and the silver oxidant. A suitable base is then introduced to maintain the pH balance, and the mixture is suspended in the chosen organic solvent. Detailed standardized synthesis steps are provided below to guide technical teams through the optimization and execution of this protocol.
- Charge a reaction vessel with polysubstituted biarylboronic acid, rhodium catalyst (e.g., pentamethylcyclopentadiene rhodium chloride dimer), silver oxidant, base, and polysubstituted azide.
- Add an organic solvent such as 1,4-dioxane and mix uniformly to ensure all reagents are fully suspended or dissolved.
- Heat the mixture to between 75°C and 85°C for 2 to 24 hours, then purify the crude product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rhodium-catalyzed technology presents a compelling value proposition centered on cost stability and operational resilience. By eliminating the need for harsh reaction conditions and complex multi-step sequences, the process significantly reduces utility costs associated with heating and cooling, as well as the capital investment required for specialized high-pressure equipment. The reliance on commercially available reagents like biarylboronic acids and azides mitigates supply chain risks, as these materials are produced by multiple vendors globally, preventing single-source bottlenecks. Furthermore, the high functional group tolerance means that a single platform technology can be adapted to produce a wide library of derivatives, allowing manufacturers to respond rapidly to changing market demands without retooling entire production lines.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis drastically cuts down on solvent usage and waste disposal costs compared to traditional multi-step routes. By avoiding the use of expensive palladium catalysts often required for C-N bond formation and replacing them with efficient rhodium systems that operate at lower loadings, the overall catalyst cost per kilogram of product is substantially optimized. Additionally, the simplified workup procedure, which often involves basic filtration and crystallization rather than extensive chromatography, reduces labor hours and consumable expenses, leading to a more competitive cost structure for high-purity carbazole intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures that production schedules are less susceptible to delays caused by equipment failure or stringent environmental controls. Since the process does not require strictly anhydrous conditions, the handling and storage of raw materials become less cumbersome, reducing the likelihood of batch rejection due to moisture sensitivity. This operational flexibility enhances the reliability of delivery timelines, a critical factor for downstream customers in the pharmaceutical and electronics industries who depend on just-in-time inventory models to maintain their own production efficiency.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the use of standard reactor materials and common solvents that are easily recycled. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the permitting hurdles and compliance costs associated with chemical manufacturing. The ability to run the reaction at atmospheric pressure or low positive pressure further enhances safety profiles, making it easier to obtain regulatory approval for large-scale facilities and ensuring long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for potential integration into their manufacturing portfolios.
Q: What are the key advantages of this rhodium-catalyzed method over traditional carbazole synthesis?
A: This method eliminates the need for harsh high-temperature dehydrogenation or complex pre-functionalization steps required in conventional Suzuki or Buchwald-Hartwig routes, operating under milder conditions with broader functional group tolerance.
Q: Can this process be scaled for industrial production of OLED materials?
A: Yes, the protocol uses commercially available reagents and standard solvents like 1,4-dioxane without requiring strict anhydrous or oxygen-free environments, making it highly suitable for commercial scale-up of complex electronic chemicals.
Q: What is the typical yield range for these carbazole derivatives?
A: Experimental data indicates yields often exceed 80%, with specific examples reaching up to 96%, demonstrating high efficiency and minimal byproduct formation compared to older oxidative cyclization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the rhodium-catalyzed carbazole formation in driving innovation across the pharmaceutical and electronic materials sectors. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest international standards for impurity profiles and residual solvent content.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall time to market.
