Advanced Zirconium-Catalyzed Synthesis and Purification of High-Purity Naphthol AS-PH for Industrial Dye Manufacturing
Advanced Zirconium-Catalyzed Synthesis and Purification of High-Purity Naphthol AS-PH for Industrial Dye Manufacturing
The global demand for high-performance azo dyes necessitates intermediates of exceptional purity and consistent quality, driving continuous innovation in synthetic methodologies. A significant technological breakthrough in this sector is detailed in patent CN111777524B, which introduces a novel post-treatment method for preparing Naphthol AS-PH, a critical coupling component for insoluble azo dyes used extensively in cotton fabric printing. This patent addresses long-standing industry challenges regarding product coloration, purity levels, and the management of alkali-insoluble impurities that have historically plagued traditional manufacturing routes. By shifting away from conventional alkali-soluble acidification techniques toward a sophisticated solvent-based purification strategy, this technology offers a pathway to produce light brown, high-purity Naphthol AS-PH with purity levels exceeding 99.5 percent. For R&D directors and procurement specialists alike, understanding the nuances of this zirconium-catalyzed route and its subsequent ethanol-washing protocol is essential for securing a competitive edge in the fine chemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Naphthol AS-PH has relied heavily on phosphorus trichloride preparation methods followed by purification via alkali dissolution and subsequent acidification or crude recrystallization. While these traditional pathways are well-established, they suffer from inherent technical deficiencies that compromise the final product quality and environmental footprint. A primary issue is the persistent presence of alkali-insoluble substances, which often remain trapped within the crystal lattice or as surface contaminants, leading to purity levels that frequently fall short of the stringent requirements for high-end textile applications. Furthermore, the aggressive chemical environment of alkali-acid treatments often induces side reactions or degradation, resulting in undesirable yellowish coloration of the final powder, which limits its applicability in producing vibrant, true-shade dyes. The recycling of mother liquors in these traditional processes is also problematic, as the accumulation of impurities and the difficulty in recovering catalysts efficiently lead to increased waste generation and higher operational costs over time.
The Novel Approach
In stark contrast to these legacy methods, the approach outlined in CN111777524B leverages a mild yet highly effective zirconium-catalyzed condensation followed by a multi-stage absolute ethanol washing protocol. This novel methodology eliminates the need for harsh alkaline and acidic reagents during the purification phase, thereby preserving the structural integrity of the naphthol derivative and preventing color degradation. The core innovation lies in the precise control of solvent ratios and temperatures during the washing steps, which selectively dissolves and removes impurities while retaining the desired product in high yield. By utilizing mesitylene as a reaction solvent and a specific zirconium complex as a catalyst, the synthesis achieves high conversion rates at temperatures between 130°C and 165°C, with water byproduct effectively removed via nitrogen purging. This streamlined process not only simplifies the operational workflow but also ensures that the final product exhibits a desirable light brown color and an alkali-insoluble content of less than 0.08 percent, meeting the rigorous specifications demanded by modern dye manufacturers.
Mechanistic Insights into Zirconium-Catalyzed Amidation and Solvent Purification
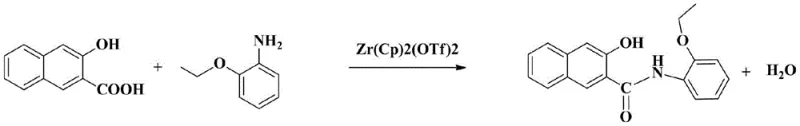
The chemical foundation of this advanced synthesis rests on the efficacy of the bis(cyclopentadienyl)zirconium(IV) bis(trifluoromethanesulfonic acid) THF complex as a Lewis acid catalyst. This specific zirconium species activates the carboxylic acid group of 2-hydroxy-3-naphthoic acid, facilitating a nucleophilic attack by the amine group of o-aminophenetole to form the critical amide bond. The reaction mechanism proceeds efficiently in mesitylene, a high-boiling aromatic solvent that provides a stable thermal environment for the condensation to occur at 140°C to 150°C. The introduction of nitrogen gas during the reaction serves a dual purpose: it maintains an inert atmosphere to prevent oxidation of the sensitive phenolic groups and actively strips away the water of reaction, driving the equilibrium toward product formation according to Le Chatelier's principle. This catalytic system is particularly advantageous because it operates under relatively mild conditions compared to traditional phosphorus halide methods, reducing the energy input required and minimizing the formation of thermal degradation byproducts that contribute to product discoloration.
Following the synthesis, the purification mechanism relies on the differential solubility of the product versus impurities in absolute ethyl alcohol at controlled temperatures. The process involves a sequential washing strategy where the crude mixture is first cooled to 5-10°C to precipitate the product, followed by washing with specific mass ratios of ethanol (0.5 to 0.8 relative to the mixture). This initial wash removes the bulk of the soluble organic impurities and residual solvent. Subsequent washing steps with varying ethanol ratios further refine the crystal surface, leaching out trace alkali-insoluble substances that would otherwise persist. The final leaching step, repeated three times with a low ratio of ethanol, ensures that any remaining mother liquor adhering to the crystals is displaced by fresh solvent, which is then removed during vacuum drying. This physical purification method avoids the hydrolysis risks associated with acid-base treatments, ensuring that the amide bond remains intact and the phenolic functionality is preserved, resulting in a chemically stable and high-purity intermediate suitable for downstream diazo coupling reactions.
How to Synthesize Naphthol AS-PH Efficiently
The synthesis of Naphthol AS-PH via this patented route requires precise adherence to stoichiometric ratios and thermal profiles to maximize yield and purity. The process begins with the charging of dried mesitylene, 2-hydroxy-3-naphthoic acid, and the zirconium catalyst under nitrogen protection, followed by the controlled addition of o-aminophenetole. Maintaining the reaction temperature between 130°C and 165°C for approximately 4 hours is critical to ensure complete conversion while allowing for the continuous removal of water. Once the reaction is complete, the solvent is removed, and the residue undergoes the specialized ethanol washing sequence described in the patent. This standardized approach minimizes batch-to-batch variability and ensures that the final product consistently meets the target purity of over 99.5 percent. For detailed operational parameters, including specific stirring times and exact mass ratios for each washing stage, please refer to the comprehensive synthesis guide provided below.
- React 2-hydroxy-3-naphthoic acid with o-aminophenetole in mesitylene using a bis(cyclopentadienyl)zirconium(IV) catalyst at 130-165°C.
- Remove solvent and cool the mixture to 5-10°C, then perform sequential washing and leaching with absolute ethyl alcohol.
- Dry the refined wet product under vacuum at 105°C to obtain light brown powder with purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel post-treatment method offers substantial strategic benefits beyond mere technical superiority. The elimination of alkali-soluble acidification steps fundamentally alters the cost structure of manufacturing by removing the need for large volumes of corrosive acids and bases, which in turn reduces the burden on wastewater treatment facilities and lowers the cost of hazardous waste disposal. The ability to recycle the mother liquors through activated carbon decolorization and distillation means that both the solvent (absolute ethanol) and the valuable catalyst-substrate complex can be recovered and reused in subsequent batches. This closed-loop approach significantly reduces raw material consumption and mitigates the volatility associated with purchasing fresh reagents, leading to a more predictable and stable cost of goods sold. Furthermore, the simplified operational workflow reduces the complexity of the production line, potentially lowering labor costs and minimizing the risk of operator error during the purification phase.
- Cost Reduction in Manufacturing: The transition to a solvent-based purification system eliminates the expensive and logistically challenging handling of strong acids and alkalis, directly reducing utility and safety compliance costs. By enabling the recycling of the catalyst and solvent through a straightforward distillation and decolorization process, the overall material intensity of the production is drastically lowered. This efficiency gain translates into significant long-term savings, as the dependency on fresh raw materials is diminished, and the throughput of the facility is optimized by shorter cycle times associated with the simpler washing protocol compared to multi-step recrystallization or acidification.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route, characterized by its tolerance to standard industrial conditions and the use of readily available solvents like mesitylene and ethanol, ensures a high degree of supply continuity. The high purity of the final product (>99.5%) reduces the likelihood of downstream rejection by dye manufacturers, thereby minimizing the risk of costly returns or rework orders. Additionally, the ability to effectively recycle catalyst residues means that supply chain disruptions related to the sourcing of specialized zirconium catalysts are mitigated, as the inventory turnover for this critical reagent is significantly extended through internal recovery loops.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with increasingly stringent global regulations regarding industrial effluent. By avoiding the generation of saline wastewater typical of acid-base neutralization, the process simplifies compliance with environmental discharge standards. The scalability of the ethanol washing technique is proven, as it relies on standard filtration and drying equipment found in most fine chemical plants, allowing for seamless expansion from pilot scale to multi-ton commercial production without the need for exotic or custom-built reactor systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in CN111777524B, providing a clear understanding of how this method compares to existing industry standards. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: What is the primary advantage of the new post-treatment method for Naphthol AS-PH?
A: The new method abandons traditional alkali-soluble acidification, utilizing absolute ethanol washing instead. This results in significantly lower alkali-insoluble content (<0.08%) and higher purity (>99.5%) compared to conventional processes.
Q: How does the zirconium catalyst improve the synthesis efficiency?
A: The bis(cyclopentadienyl)zirconium(IV) bis(trifluoromethanesulfonic acid) THF complex facilitates efficient amidation between 2-hydroxy-3-naphthoic acid and o-aminophenetole at moderate temperatures (140-150°C), allowing for effective water removal and high conversion rates.
Q: Can the solvents and catalysts be recycled in this process?
A: Yes, the process allows for the combination of mother liquors from washing steps. After activated carbon decolorization and distillation, absolute ethanol is recovered for reuse, and the substrate containing the catalyst residue is recycled back into the reaction, reducing waste and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthol AS-PH Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN111777524B requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature, solvent ratios, and catalyst loading is maintained even at full industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical alkali-insoluble content metrics that define high-quality Naphthol AS-PH. We are committed to delivering products that not only meet but exceed the performance expectations of the global dye and pigment industry.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this efficient purification technology can drive down your total landed costs. We encourage potential partners to contact our technical procurement team to request specific COA data from our recent batches and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable source of high-purity Naphthol AS-PH that empowers your downstream dye synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
