Revolutionizing Naphthol AS-PH Production: A High-Efficiency Zirconium-Catalyzed Route for Industrial Scale-Up
Introduction to Advanced Dye Intermediate Manufacturing
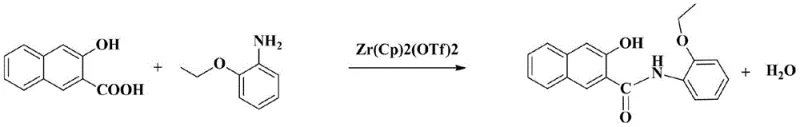
The global demand for high-performance organic pigments and dye intermediates necessitates a paradigm shift from traditional, waste-intensive synthetic routes to more sustainable and efficient catalytic processes. A groundbreaking development in this sector is detailed in patent CN111825565A, which introduces a novel preparation method for Naphthol AS-PH, a critical coupling component for insoluble azo dyes used extensively in cotton fabric printing. This technology replaces the conventional phosphorus trichloride-mediated acylation with a sophisticated one-step coordination catalysis system. By utilizing a bis(cyclopentadienyl)zirconium(IV) bis(trifluoromethanesulfonate) THF complex, the process achieves exceptional conversion rates and product quality while addressing severe environmental concerns associated with phosphorus waste. For industrial stakeholders, this represents not merely a chemical optimization but a strategic opportunity to enhance supply chain resilience and reduce the ecological footprint of pigment manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Naphthol AS-PH has relied heavily on phosphorus trichloride (PCl3) mediated acylation, encompassing both sodium salt and acid acylation variants. While these methods have served the industry for decades, they are plagued by inherent inefficiencies and environmental liabilities that are increasingly untenable in modern regulatory landscapes. The traditional processes typically suffer from moderate yields, often hovering between 80% and 85%, which translates to significant raw material loss and increased cost per kilogram of active ingredient. Furthermore, the reliance on phosphorus reagents generates substantial amounts of phosphorus-containing wastewater and solid waste, known as 'three wastes,' which require expensive and energy-intensive treatment protocols to meet environmental discharge standards. The complexity of these multi-step procedures also introduces more points of failure, leading to variability in product quality and potential impurity profiles that can affect downstream dyeing performance.
The Novel Approach
In stark contrast, the innovative route disclosed in patent CN111825565A leverages advanced organometallic chemistry to streamline production into a single, highly efficient step. By employing a zirconium-based coordination catalyst, the reaction between 2-hydroxy-3-naphthoic acid and o-aminophenyl ethyl ether proceeds with remarkable selectivity and speed. This novel approach eliminates the need for hazardous phosphorylating agents entirely, thereby eradicating the generation of toxic phosphorus byproducts at the source. The process operates under relatively mild thermal conditions compared to harsh traditional methods, yet it delivers a dramatic improvement in yield, reaching up to 98.5%—a gain of approximately 15% over conventional techniques. This leap in efficiency not only maximizes atom economy but also ensures a consistent supply of high-purity material, essential for manufacturers of premium textile dyes seeking reliable dye intermediate suppliers.
Mechanistic Insights into Zirconium-Catalyzed Amide Formation
The core of this technological breakthrough lies in the unique activation mechanism provided by the bis(cyclopentadienyl)zirconium(IV) bis(trifluoromethanesulfonate) THF complex. Unlike traditional coupling reagents that activate the carboxylic acid through the formation of reactive acyl chlorides or anhydrides, this zirconium catalyst functions as a Lewis acid coordinator. It likely facilitates the nucleophilic attack of the amine group from o-aminophenyl ethyl ether onto the carbonyl carbon of the naphthoic acid by coordinating with the oxygen atoms, thereby lowering the activation energy of the transition state. This coordination environment stabilizes the intermediate species and promotes the direct dehydration condensation required to form the amide bond found in Naphthol AS-PH. The presence of the trifluoromethanesulfonate ligands enhances the electrophilicity of the zirconium center, ensuring rapid turnover and high catalytic activity even at the elevated temperatures of 130-165°C utilized in the process.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over the chaotic side-reactions often seen in phosphorus-based chemistries. The specificity of the coordination catalyst minimizes the formation of over-acylated byproducts or degradation products that typically arise from harsh acidic conditions. The patent data indicates that the resulting product consistently achieves purity levels exceeding 99.5%, with some examples reporting up to 99.7% purity after simple recrystallization or washing steps. This high level of chemical fidelity is crucial for R&D directors who require precise impurity profiles to ensure color fastness and hue consistency in the final textile applications. The ability to control the reaction trajectory so precisely reduces the burden on downstream purification units, allowing for a more streamlined and cost-effective manufacturing workflow.
How to Synthesize Naphthol AS-PH Efficiently
Implementing this zirconium-catalyzed synthesis requires careful attention to reaction parameters to maximize the benefits of the new technology. The process begins with the preparation of a reaction vessel under an inert nitrogen atmosphere to prevent moisture interference, which is critical for the stability of the coordination catalyst. The standardized protocol involves charging dried mesitylene solvent, the naphthoic acid substrate, and the precise molar ratio of the zirconium catalyst before initiating the heating phase. Once the optimal temperature range is reached, the amine component is added dropwise to manage the exotherm and ensure complete conversion. Detailed standard operating procedures regarding specific stoichiometric ratios, temperature ramping rates, and work-up protocols are essential for successful technology transfer.
- Charge dried mesitylene, 2-hydroxy-3-naphthoic acid, and the zirconium coordination catalyst into a reactor under nitrogen protection.
- Heat the mixture to 130-165°C and dropwise add o-aminophenyl ethyl ether while maintaining temperature and removing water via nitrogen sweep.
- Cool the reaction, recover solvent, wash crude product with anhydrous methanol, and dry under vacuum to obtain high-purity Naphthol AS-PH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield metrics. The elimination of phosphorus trichloride removes a major hazardous material from the supply chain, reducing storage risks, transportation costs, and regulatory compliance burdens associated with toxic reagents. Furthermore, the drastic reduction in waste generation translates directly into lower disposal costs and reduced liability, contributing to significant overall cost reduction in dye intermediate manufacturing. The robustness of the process ensures a stable supply of high-purity Naphthol AS-PH, mitigating the risk of production delays caused by quality failures or environmental shutdowns common with older, dirtier technologies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substantial increase in yield and the recyclability of key inputs. By achieving yields near 98.5% compared to the traditional 80-85%, the effective consumption of raw materials per unit of product is drastically lowered. Additionally, the patent explicitly highlights the ability to recycle both the expensive zirconium coordination catalyst and the mesitylene solvent. The recovery of the solvent via distillation and the reuse of the catalyst from the mother liquor create a closed-loop system that minimizes fresh material purchases. This circular approach significantly lowers the variable cost of production, providing a competitive pricing advantage without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the capacity of waste treatment facilities. This new method relies on readily available starting materials and a catalyst that, while specialized, is used in small quantities and can be recovered. The simplification of the process from multiple steps to a one-pot reaction reduces the equipment footprint and processing time, allowing for faster batch turnover. This agility enables manufacturers to respond more quickly to market fluctuations and urgent orders, effectively reducing lead time for high-purity dye intermediates and strengthening the reliability of the supply chain for downstream textile clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this zirconium-catalyzed route is inherently designed for green chemistry principles. The absence of corrosive phosphorus halides and the reduction of aqueous waste streams simplify the engineering requirements for large-scale reactors and effluent treatment plants. The process operates cleanly, producing a light yellow powder product that requires minimal purification, which facilitates the commercial scale-up of complex dye intermediates. Compliance with increasingly stringent global environmental regulations is easier to maintain, ensuring long-term operational viability and protecting the brand reputation of the manufacturing entity against ecological scrutiny.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its integration into their production lines. The following questions address common inquiries regarding the practical implementation, safety profile, and quality outcomes of the zirconium-catalyzed process. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of the zirconium-catalyzed method over the traditional phosphorus trichloride process?
A: The zirconium-catalyzed method eliminates the use of hazardous phosphorus trichloride, significantly reducing phosphorus-containing waste ('three wastes'). It achieves a yield improvement of approximately 15% (reaching up to 98.5%) and simplifies the process to a one-step reaction, compared to the complex multi-step traditional methods.
Q: Can the catalyst and solvent be recycled in this new synthesis route?
A: Yes, a key feature of this patented process is the recyclability of both the coordination catalyst and the mesitylene solvent. The post-treatment involves recovering the solvent and combining mother liquors to reuse the catalyst, which drastically lowers raw material consumption and production costs.
Q: What is the expected purity and yield for Naphthol AS-PH using this method?
A: According to the patent data, this method consistently achieves product purity levels of 99.5% to 99.7% and yields ranging from 96.5% to 98.6%, which represents a substantial improvement over the conventional 80-85% yield range.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthol AS-PH Supplier
As the chemical industry evolves towards more sustainable and efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront of adopting these cutting-edge technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN111825565A are fully realized in industrial practice. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest standards for dye intermediate applications. We understand that consistency is key for our clients, and our advanced manufacturing capabilities allow us to deliver Naphthol AS-PH with the reliability and purity required for high-end textile markets.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced zirconium-catalyzed route, we can offer a value proposition that balances superior product quality with optimized production costs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to optimize your supply chain and secure a sustainable source of high-performance dye intermediates for your future growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
