Advanced Synthesis of Tofacitinib Intermediates for Commercial Scale Production
Advanced Synthesis of Tofacitinib Intermediates for Commercial Scale Production
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of Janus kinase (JAK) inhibitors, particularly Tofacitinib, which has revolutionized the treatment of rheumatoid arthritis and other autoimmune conditions. Patent CN109503462B discloses a highly efficient synthetic method for the key intermediate (3R, 4R)-N, 4-dimethyl-1-benzyl-3-piperidylamine and its salts. This technology addresses critical bottlenecks in existing manufacturing processes by introducing a novel sequence that leverages orthoformate condensation and selective catalytic hydrogenation. The structural integrity of the target compound (I) and its oxalate salt (K) is paramount for downstream API synthesis, as depicted in the chemical structures below.
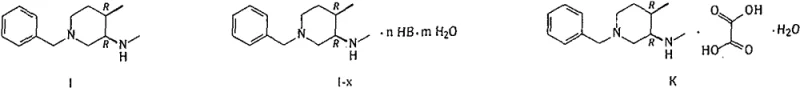
This patent represents a significant leap forward in process chemistry, moving away from hazardous reagents and low-selectivity reductions towards a more atom-economical and stereo-controlled approach. For global supply chains, the ability to produce high-purity intermediates with consistent stereochemistry is not just a technical requirement but a commercial imperative. The method described ensures that the critical chiral centers at the 3 and 4 positions of the piperidine ring are established with high fidelity, minimizing the burden on downstream purification and maximizing overall yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this piperidine scaffold has been plagued by stereochemical inefficiencies and safety hazards. As illustrated in prior art Route 1 (Org. Process Res. Dev. 2014), the process relies on the reduction of a ketimine intermediate (IV-a) derived from a ketone precursor. This reduction step, typically performed with sodium borohydride, suffers from inherent steric hindrance issues caused by the ortho-methyl group. Consequently, the stereoselectivity is suboptimal, yielding a mixture of cis and trans isomers with a ratio of approximately 86.13:13.87 in favor of the desired cis-form. This necessitates complex and yield-loss-inducing separation steps to remove the unwanted (3R, 4S-rel-) isomer pair.
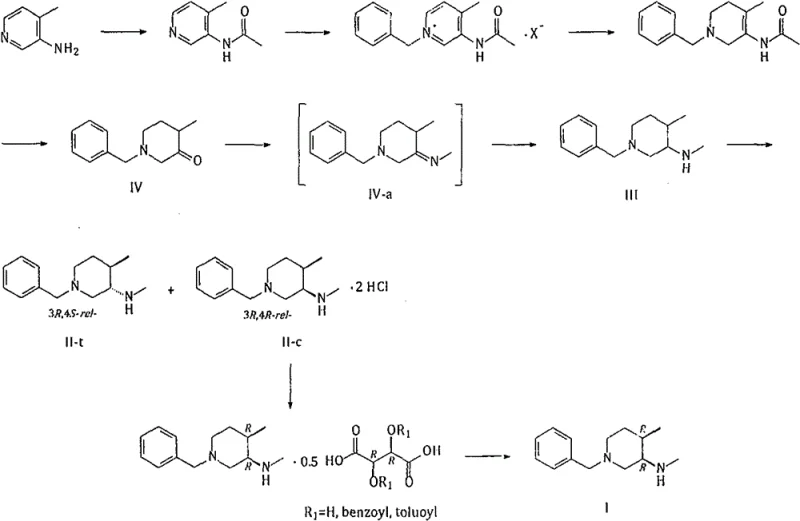
Furthermore, alternative Route 2 (WO 2007012953) attempts to bypass some of these issues but introduces severe safety and cost liabilities. This pathway requires the use of lithium aluminum hydride (LiAlH4) or Red-Al for the reduction of carbamate-protected intermediates. These reagents are notoriously dangerous, requiring strictly anhydrous conditions and specialized handling equipment due to their pyrophoric nature. From a procurement and operational standpoint, the reliance on such hazardous materials increases insurance costs, complicates waste disposal, and poses significant risks during commercial scale-up, making it less attractive for large-volume API manufacturing.
The Novel Approach
The methodology disclosed in CN109503462B fundamentally reimagines the construction of the piperidine ring to overcome these legacy defects. Instead of reducing a ketone or using dangerous hydrides, the process initiates with the condensation of 3-amino-4-methylpyridine with an orthoformate to generate an iminoformate intermediate (A-x). This is subsequently reduced using the much safer and cheaper sodium borohydride. The true innovation lies in the subsequent steps: after Boc protection and quaternization with benzyl halide, the pyridine ring is reduced to an enamine (E) using sodium borohydride, which is then subjected to Rh-catalyzed hydrogenation.
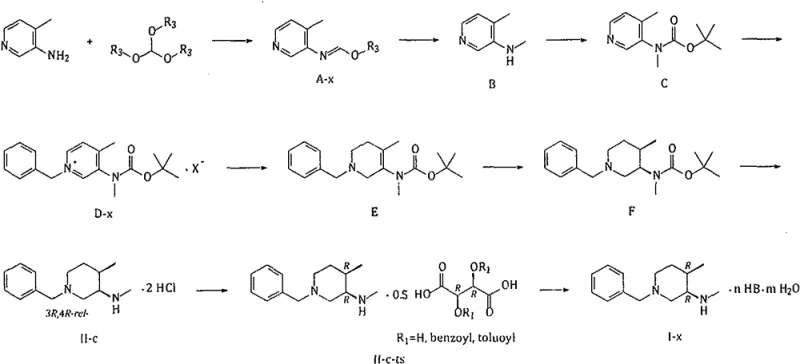
This strategic shift allows for a highly stereoselective cis-hydrogenation step mediated by a Rhodium catalyst. Unlike the non-catalytic borohydride reduction in Route 1, the catalytic hydrogenation on the enamine substrate preferentially delivers hydrogen to the less hindered face, resulting in a crude product mixture with a vastly superior (3R, 4R-rel-)/(3R, 4S-rel-) ratio of 95.33:4.67. Following deprotection and salt formation, this ratio improves further to 99.41:0.59 in the solid state. This dramatic improvement in intrinsic selectivity reduces the load on the final resolution step, leading to higher overall yields and a more robust process suitable for multi-ton production.
Mechanistic Insights into Rh-Catalyzed Cis-Hydrogenation and Chiral Resolution
The core of this technological advancement is the Rh-catalyzed hydrogenation of the tetrahydropyridine enamine intermediate (E). In this mechanism, the rhodium catalyst, typically supported on carbon (Rh/C), facilitates the syn-addition of hydrogen across the double bond of the enamine. The presence of the bulky N-Boc group and the methyl substituent directs the approach of the catalyst and the hydrogen molecules, favoring the formation of the cis-configured piperidine ring. This catalytic cycle is not only highly selective but also efficient; the patent notes that the precious metal catalyst can be recovered and reused, with metal loss kept within manageable limits (approximately 5% per batch), which is crucial for maintaining cost efficiency in continuous manufacturing environments.
Following the establishment of the relative stereochemistry, the process employs a rigorous purification protocol to ensure absolute optical purity. The crude hydrogenation product is subjected to acidic deprotection and salification to form the dihydrochloride salt (II-c). This step acts as a preliminary purification, crystallizing out the major (3R, 4R-rel-) diastereomer while leaving the minor (3R, 4S-rel-) impurity in the mother liquor. The final and most critical step involves chiral resolution using O,O'-di-p-toluoyl-L-tartaric acid (L-DTTA). This resolving agent forms a diastereomeric salt specifically with the desired (3R, 4R) enantiomer. Through careful control of solvent conditions (typically ethanol) and temperature, the desired salt crystallizes with high purity, effectively rejecting the (3S, 4S) enantiomer. This multi-layered approach to impurity control guarantees that the final free base or oxalate salt meets the stringent specifications required for pharmaceutical applications.
How to Synthesize (3R, 4R)-N, 4-dimethyl-1-benzyl-3-piperidylamine Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the novel pathway. The process begins with the thermal condensation of the pyridine starting material with triethyl orthoformate, followed by a controlled reduction. Operators must pay close attention to the temperature profiles during the Rh-catalyzed hydrogenation step, typically maintaining pressures between 0.4-0.5 MPa and temperatures around 70-75°C to ensure complete conversion without over-reduction or catalyst degradation. The subsequent resolution step demands high-purity solvents and precise stoichiometry of the resolving agent to achieve the target optical purity. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- Condense 3-amino-4-methylpyridine with orthoformate to form an iminoformate, followed by metal borohydride reduction to obtain the N-methyl derivative.
- Protect the amino group with Boc, react with benzyl halide to form a quaternary ammonium salt, and reduce with sodium borohydride to generate an enamine intermediate.
- Perform Rh-catalyzed cis-hydrogenation to enrich the (3R, 4R-rel-) configuration, followed by deprotection, salification, and resolution with L-DTTA to isolate the pure enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. By eliminating the need for hazardous reducing agents like lithium aluminum hydride, the process significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) and reduces the regulatory burden associated with handling dangerous goods. The reliance on commodity chemicals such as orthoformates, sodium borohydride, and benzyl chloride ensures a stable and resilient supply chain, insulating production from the volatility often seen with specialized fine chemical reagents. Furthermore, the improved stereoselectivity directly translates to better mass balance and reduced waste generation, aligning with modern green chemistry initiatives and lowering disposal costs.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents like LiAlH4 and Red-Al with sodium borohydride and catalytic hydrogenation results in substantial raw material cost savings. Sodium borohydride is a ubiquitous industrial chemical with a stable price point, whereas lithium-based hydrides command a premium due to their specialized handling requirements. Additionally, the ability to recover and reuse the Rhodium catalyst mitigates the impact of precious metal pricing fluctuations. The higher stereoselectivity of the hydrogenation step means less material is lost during the purification and resolution phases, effectively increasing the yield per kilogram of starting material and driving down the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The starting materials for this route, including 3-amino-4-methylpyridine and various orthoformates, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The simplified reaction conditions, which do not require extreme cryogenic temperatures or strictly anhydrous environments for every step, allow for production in a broader range of facilities. This flexibility enhances supply continuity, ensuring that pharmaceutical partners can secure reliable volumes of this critical intermediate even during periods of market disruption. The robustness of the salt formation and crystallization steps further guarantees consistent product quality across different batches and production sites.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior. Eliminating pyrophoric reagents reduces the risk of fire and explosion, lowering insurance premiums and facility maintenance costs. The aqueous workups and standard solvent systems (toluene, alcohols, acetone) are easier to treat and recycle compared to the complex waste streams generated by aluminum-based reductions. The process is designed for scalability, with the hydrogenation step being readily adaptable to large-scale high-pressure reactors. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to meet growing global demand for Tofacitinib without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines.
Q: How does this novel synthesis method improve stereoselectivity compared to conventional routes?
A: Unlike conventional Route 1 which relies on the reduction of a ketimine intermediate (IV-a) resulting in poor stereoselectivity (approx. 86:14 ratio), this novel method utilizes Rh-catalyzed cis-hydrogenation of an enamine intermediate. This catalytic step inherently favors the formation of the cis-isomer, achieving a significantly improved ratio of (3R, 4R-rel-) to (3R, 4S-rel-) products (95.33:4.67) prior to resolution.
Q: What are the safety advantages of avoiding lithium aluminum hydride in this process?
A: Conventional Route 2 requires the use of lithium aluminum hydride (LiAlH4) or Red-Al for the reduction of carbamate protected intermediates, both of which are hazardous, moisture-sensitive, and expensive reagents. The patented method replaces these dangerous reducing agents with sodium borohydride for the initial reductions and catalytic hydrogenation for the ring saturation, drastically simplifying safety protocols and reducing raw material costs.
Q: How is the optical purity of the final intermediate ensured?
A: Optical purity is ensured through a multi-stage purification strategy. First, the Rh-catalyzed hydrogenation enriches the desired relative configuration. Second, the formation of the dihydrochloride salt precipitates the major (3R, 4R-rel-) isomer while leaving the minor (3R, 4S-rel-) isomer in the mother liquor, improving the ratio to 99.41:0.59. Finally, resolution with O,O'-di-p-toluoyl-L-tartaric acid (L-DTTA) isolates the single 3R, 4R enantiomer with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tofacitinib Intermediate Supplier
The synthesis method detailed in CN109503462B represents a gold standard for the production of high-purity pharmaceutical intermediates, combining safety, efficiency, and scalability. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical purity of intermediates like (3R, 4R)-N, 4-dimethyl-1-benzyl-3-piperidylamine.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and economic advantages of our manufacturing capabilities. Let us collaborate to drive down costs and accelerate the delivery of life-saving medications to patients worldwide.
