Revolutionizing Benzotriazole UV Absorber Production with Green Ionic Liquid Catalysis for Commercial Scale-up
Revolutionizing Benzotriazole UV Absorber Production with Green Ionic Liquid Catalysis for Commercial Scale-up
The global demand for high-performance ultraviolet stabilizers in polymer and coating industries has necessitated a shift towards more sustainable and efficient manufacturing processes. Patent CN102040561A introduces a groundbreaking methodology for synthesizing benzotriazole UV absorbers, utilizing acidic ionic liquids as a dual-function reaction medium and catalyst. This innovation addresses critical challenges in traditional synthetic routes by enabling a one-pot reductive cyclization of azo intermediates using glucose and zinc powder under mild thermal conditions. By leveraging the unique physicochemical properties of ionic liquids, such as negligible vapor pressure and tunable acidity, this technology significantly enhances reaction selectivity and product purity. For R&D directors and procurement specialists, this patent represents a pivotal advancement in cost reduction in polymer additive manufacturing, offering a pathway to greener chemistry without compromising on yield or operational feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
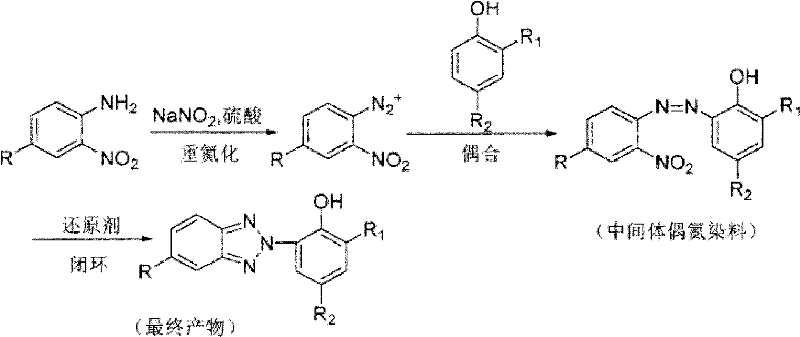
Traditional synthesis of benzotriazole UV absorbers typically involves a multi-step sequence starting with diazotization of arylamines, followed by coupling with alkylphenols to form azo intermediates, and finally, a complex reductive cyclization. The conventional reductive cyclization step is notoriously difficult to control, often requiring harsh conditions that can lead to the cleavage of the azo bond and the formation of unwanted amine by-products. Furthermore, standard protocols frequently employ volatile organic solvents that pose significant environmental and safety hazards, complicating waste management and increasing the overall carbon footprint of the production facility. The separation of the final product from the reaction mixture is often labor-intensive, requiring extensive purification steps to remove residual metals and solvents, which drives up operational costs and extends lead times. These inefficiencies create a bottleneck for commercial scale-up of complex polymer additives, limiting the ability of manufacturers to respond rapidly to market demands while maintaining stringent quality standards.
The Novel Approach
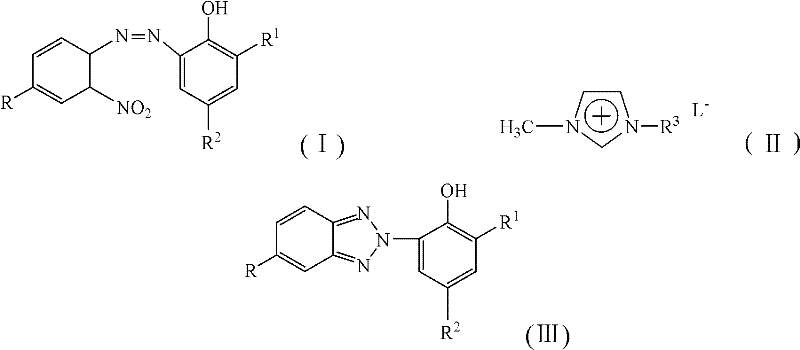
In stark contrast, the novel approach detailed in the patent utilizes acidic ionic liquids to facilitate a streamlined, one-step reduction and cyclization process. By dissolving the azo intermediate, glucose, and zinc powder directly into the ionic liquid matrix, the reaction proceeds smoothly at temperatures ranging from 10°C to 60°C, drastically reducing energy consumption compared to traditional high-temperature methods. The ionic liquid not only acts as a solvent with superior dissolving power for organic reactants but also provides the necessary acidic environment to catalyze the cyclization, effectively replacing hazardous mineral acids. This integration of functions simplifies the workflow, eliminates the need for multiple solvent exchanges, and allows for the recovery and reuse of the ionic liquid medium for over five cycles without significant loss of activity. Consequently, this method offers a robust solution for reducing lead time for high-purity benzotriazole UV absorbers, ensuring a more reliable supply chain for downstream applications in plastics and coatings.
Mechanistic Insights into Ionic Liquid-Mediated Reductive Cyclization
The core of this technological breakthrough lies in the intricate interplay between the glucose-zinc powder reducing system and the acidic ionic liquid environment. Mechanistically, the reaction initiates with the reduction of the nitro group on the azo intermediate, followed by the intramolecular cyclization to form the benzotriazole ring structure. The presence of glucose serves as a mild reducing agent that helps regulate the reduction potential, preventing the over-reduction or indiscriminate cleavage of the azo linkage which is a common failure mode in single-agent reduction systems. Zinc powder acts as the primary electron donor, driving the conversion of the nitro group to the requisite amine species that immediately undergoes condensation. The acidic ionic liquid stabilizes the transition states and intermediates through hydrogen bonding and electrostatic interactions, ensuring that the cyclization occurs with high regioselectivity. This precise control over the reaction pathway minimizes the formation of hydrazine by-products and ensures that the final benzotriazole structure is formed with exceptional fidelity, meeting the rigorous purity specifications required for high-end optical applications.
Furthermore, the solvation properties of the ionic liquid play a critical role in impurity control and product isolation. Unlike traditional organic solvents where solubility differences might precipitate intermediates prematurely, the ionic liquid maintains all reactants in a homogeneous phase throughout the reaction duration. This homogeneity ensures uniform heat distribution and mass transfer, preventing localized hot spots that could trigger decomposition or side reactions. Post-reaction, the distinct polarity difference between the ionic liquid and the organic product allows for facile separation using simple extraction techniques with non-polar solvents like toluene. The ionic liquid remains in the aqueous phase or can be separated due to immiscibility, allowing it to be dried and recycled, while the organic layer contains the crude product with minimal contamination. This mechanism effectively decouples the product from the catalyst system, streamlining the purification process and significantly reducing the generation of hazardous chemical waste associated with catalyst disposal.
How to Synthesize Benzotriazole UV Absorbers Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this green chemistry approach in a laboratory or pilot plant setting. The process begins with the precise weighing of the azo intermediate, glucose, and zinc powder, which are then introduced into the reactor containing the pre-heated acidic ionic liquid. Maintaining the temperature within the optimal range of 40°C to 50°C is crucial for balancing reaction kinetics and selectivity, with reaction progress monitored via thin-layer chromatography (TLC) until the starting material is fully consumed. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding the azo intermediate, glucose, and zinc powder into the acidic ionic liquid solvent at a controlled molar ratio.
- Maintain the reaction temperature between 10°C and 60°C, preferably 40-50°C, and stir for 1 to 3 hours while monitoring progress via TLC.
- Upon completion, extract the product using toluene, wash the organic layer with distilled water, dry over anhydrous magnesium sulfate, and concentrate under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based synthesis offers transformative benefits that extend beyond mere technical feasibility. The ability to recycle the ionic liquid solvent multiple times translates directly into substantial raw material cost savings over the lifecycle of the production campaign, reducing the dependency on expensive and fluctuating organic solvent markets. Moreover, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature equipment, lowering capital expenditure requirements for new production lines and reducing maintenance costs for existing infrastructure. The simplified post-treatment process, which avoids complex distillation columns or chromatographic separations, further accelerates the throughput of the manufacturing facility, enabling faster turnaround times for customer orders. These operational efficiencies collectively contribute to a more resilient and cost-effective supply chain, positioning manufacturers to offer competitive pricing while adhering to increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents and the reusability of the ionic liquid catalyst significantly lower the variable costs associated with solvent purchase, recovery, and disposal. By avoiding the use of stoichiometric amounts of strong mineral acids and reducing the consumption of zinc powder through better efficiency, the overall material cost per kilogram of product is optimized. Additionally, the high yield reported in the patent examples, reaching up to 96%, ensures that raw material utilization is maximized, minimizing waste and improving the overall economic viability of the process. These factors combine to create a leaner manufacturing model that is less susceptible to raw material price volatility.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, operating safely at low temperatures, reduces the risk of unplanned shutdowns due to thermal runaways or equipment failures. The use of readily available and stable reagents like glucose and zinc powder ensures a secure supply of inputs, unlike specialized catalysts that may have long lead times or single-source dependencies. Furthermore, the simplified workflow reduces the complexity of the production schedule, allowing for more flexible batch planning and quicker response to urgent demand spikes. This reliability is critical for maintaining continuous supply to downstream customers in the automotive and packaging industries who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The green nature of this technology aligns perfectly with global sustainability goals, facilitating easier regulatory approval and permitting for new facilities. The reduction in hazardous waste generation simplifies compliance with environmental protection laws, avoiding costly fines and remediation efforts. The process is inherently scalable, as the heat management and mixing requirements are less demanding than traditional exothermic reductions, allowing for seamless transition from kilogram-scale development to multi-ton commercial production. This scalability ensures that the technology can grow with market demand without requiring fundamental changes to the process chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production portfolios.
Q: What are the advantages of using acidic ionic liquids in benzotriazole synthesis?
A: Acidic ionic liquids serve as both solvent and catalyst, offering excellent solubility for reactants, reusability for over five cycles, and eliminating the need for volatile organic solvents, thereby reducing environmental waste.
Q: How does the glucose-zinc powder reduction method improve yield?
A: This method allows for a controlled one-step reduction and cyclization under mild conditions (40-50°C), minimizing side reactions like azo bond fracture and achieving yields as high as 94-96%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates at low temperatures with simple post-treatment steps like extraction and distillation, making it highly suitable for commercial scale-up with reduced safety risks compared to traditional high-temperature methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzotriazole UV Absorber Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this ionic liquid-mediated synthesis to redefine the standards of quality and sustainability in the UV stabilizer market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzotriazole UV absorber meets the exacting requirements of the polymer and coating industries. We are committed to delivering high-purity benzotriazole UV absorbers that enhance the longevity and performance of your end products.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis route can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and efficient supply of critical UV protection additives for your global operations.
