Innovative Ionic Liquid Process for High-Yield Benzotriazole UV Absorber Production at Commercial Scale
The present analysis examines Chinese Patent CN102040561B titled 'Method for synthesizing benzotriazole ultraviolet absorber,' which introduces a groundbreaking green chemistry approach for producing critical photostabilizers used across polymer manufacturing sectors. This innovation addresses longstanding industry challenges in synthesizing benzotriazole-based UV absorbers through an integrated one-step reduction process utilizing acidic ionic liquid media. The technology demonstrates significant advancements over conventional multi-stage methodologies by eliminating intermediate isolation requirements while maintaining exceptional product purity standards required by global regulatory frameworks. Our evaluation focuses on how this patented process delivers tangible operational improvements across research development pipelines and commercial manufacturing environments without compromising on quality specifications essential for high-performance applications in polyolefins and coating systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
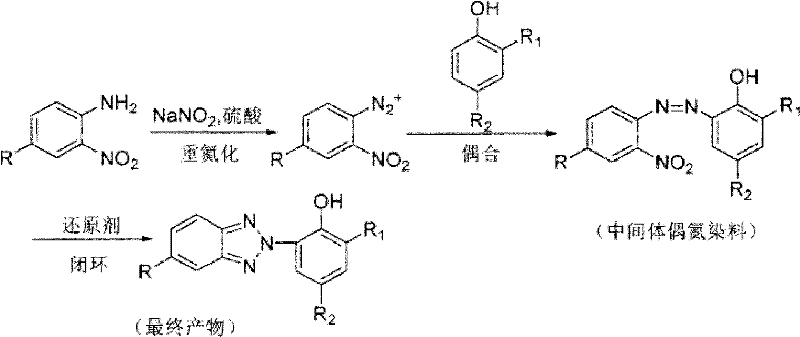
Traditional synthesis routes for benzotriazole UV absorbers suffer from multiple critical deficiencies that impact both product quality and manufacturing economics. The standard two-step reduction process using glucose-zinc powder systems creates significant operational complexity due to inconsistent reaction conditions between nitrogen oxide formation and subsequent reduction stages. This inconsistency frequently leads to azo bond cleavage during reduction steps, generating undesirable amine byproducts that compromise final product purity and necessitate extensive purification procedures. Furthermore, conventional solvents exhibit poor solubility profiles for key intermediates while requiring substantial solvent volumes that generate considerable waste streams requiring costly disposal protocols. The multi-stage nature of these processes also introduces extended production timelines that create supply chain vulnerabilities through increased dependency on intermediate storage and handling procedures that elevate contamination risks during transfer operations.
The Novel Approach
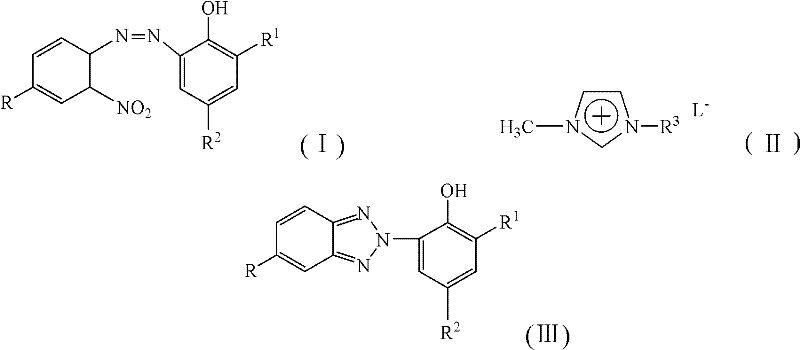
The patented methodology overcomes these limitations through an integrated one-step reduction process conducted within acidic ionic liquid media that maintains optimal reaction conditions throughout the entire transformation sequence. By combining azo intermediates with glucose and zinc powder directly in the catalytic medium at precisely controlled temperatures between 40°C and 50°C, this approach prevents azo bond cleavage while facilitating complete reductive cyclization to the target product. The acidic environment provided by the imidazolium-based catalysts ensures consistent reaction progression without intermediate isolation requirements that plagued previous methods. Crucially, the system demonstrates exceptional solubility characteristics across all reaction components while enabling straightforward product separation through simple toluene extraction protocols that eliminate complex purification sequences previously required to achieve pharmaceutical-grade purity standards.
Mechanistic Insights into Acidic Ionic Liquid-Catalyzed Reduction
The fundamental innovation lies in the precise control of redox chemistry within the acidic ionic liquid medium where the imidazolium cation structure creates an optimal microenvironment for selective reduction. The mechanism proceeds through initial activation of the azo intermediate by protonation from the acidic catalyst system followed by controlled electron transfer from glucose-zinc couple that prevents over-reduction while maintaining molecular integrity throughout cyclization. This carefully balanced redox environment specifically suppresses the competing pathway where azo bonds would otherwise cleave to form amine byproducts by stabilizing the transition state through hydrogen bonding interactions with the catalyst's sulfonic acid groups. The process achieves near-complete conversion through continuous regeneration of active reducing species within the homogeneous phase that maintains consistent reaction kinetics without requiring intermediate workup procedures that typically introduce variability in conventional approaches.
Impurity control is achieved through multiple synergistic mechanisms inherent to the catalytic system where the acidic environment selectively promotes desired cyclization while suppressing side reactions that generate amine impurities. The homogeneous nature of the reaction medium ensures uniform concentration gradients that prevent localized hot spots which typically cause decomposition pathways in conventional solvent systems. Additionally, the catalyst's ability to dissolve both organic intermediates and inorganic reductants creates a stable reaction environment where byproduct formation remains minimal throughout extended reaction periods as confirmed by consistent TLC monitoring results across multiple implementation examples. This inherent selectivity eliminates the need for additional purification steps that would otherwise be required to meet stringent purity specifications demanded by polymer additive applications where trace impurities can significantly impact final product performance characteristics.
How to Synthesize Benzotriazole UV Absorbers Efficiently
This section outlines the standardized procedure derived from patent implementation examples that enables reliable production of high-purity benzotriazole UV absorbers through optimized process parameters. The methodology represents a significant advancement over previous techniques by integrating multiple reaction stages into a single operational sequence that maintains consistent quality metrics across varying production volumes. Detailed operational guidelines below provide step-by-step instructions for implementing this technology within existing manufacturing infrastructure while achieving superior yield characteristics compared to conventional approaches.
- Combine azo intermediate with glucose and zinc powder in acidic ionic liquid at controlled temperature.
- Monitor reaction progress via TLC until complete conversion of starting material.
- Perform post-treatment by toluene extraction, washing, drying, and solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial operational improvements that directly address critical pain points within procurement and supply chain management frameworks across specialty chemical manufacturing sectors. The process redesign eliminates multiple dependency points within traditional supply chains while creating new opportunities for cost optimization through reduced material consumption and simplified logistics requirements. These advantages translate into enhanced business resilience through decreased vulnerability to raw material fluctuations and improved responsiveness to dynamic market demands without requiring significant capital investment in new production infrastructure.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps reduces operational complexity while minimizing solvent consumption through integrated reaction processing that avoids multi-stage purification requirements. The reusable catalytic system significantly lowers raw material costs by enabling multiple reaction cycles without performance degradation while reducing waste disposal expenses associated with conventional solvent-intensive processes.
- Enhanced Supply Chain Reliability: Simplified raw material requirements through consolidated reagent usage create more robust sourcing strategies with reduced dependency on specialized chemical suppliers while maintaining consistent quality outputs across production batches. The streamlined process design minimizes potential failure points within manufacturing workflows that typically cause delivery delays in traditional multi-step synthesis approaches.
- Scalability and Environmental Compliance: The homogeneous reaction system demonstrates exceptional volume scalability from laboratory to industrial production without requiring process re-engineering due to consistent performance characteristics across different batch sizes. The significant reduction in waste generation through solvent minimization and catalyst reusability directly supports environmental compliance initiatives while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations based on patent implementation data and reflect common inquiries from procurement specialists evaluating this technology for commercial adoption within their organizations' supply chain frameworks.
Q: How does the acidic ionic liquid medium improve reduction step efficiency compared to traditional solvents?
A: The acidic ionic liquid provides superior solubility for all reactants while maintaining optimal pH conditions that prevent azo bond cleavage during reduction. This eliminates side reactions that typically generate amine byproducts in conventional methods.
Q: What environmental benefits does this synthesis method offer over existing processes?
A: The process generates significantly less waste through elimination of intermediate isolation steps and enables complete reusability of the catalytic medium. The one-step reaction design reduces solvent consumption by avoiding multi-stage purification requirements.
Q: How does this technology enhance scalability for industrial manufacturing?
A: The simplified operational sequence with consistent reaction conditions across scales allows seamless transition from laboratory to plant production. The reusable catalytic system maintains performance stability during volume increases without requiring specialized equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzotriazole UV Absorber Supplier
We specialize in delivering high-performance photostabilizers through advanced green chemistry processes that combine scientific innovation with industrial practicality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our technical team applies deep expertise in specialty chemical manufacturing to ensure seamless technology transfer from laboratory development to full-scale implementation without compromising on quality or delivery timelines.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today to request detailed COA data and route feasibility assessments for your upcoming projects.
