Advanced Crystallization Technology for High-Purity Cefuroxime Sodium Commercial Production
Advanced Crystallization Technology for High-Purity Cefuroxime Sodium Commercial Production
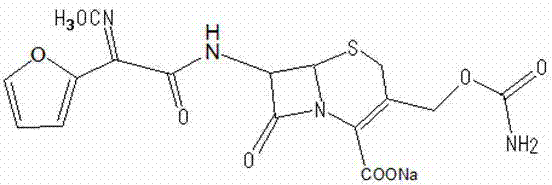
The pharmaceutical industry is constantly evolving towards greener, more efficient manufacturing processes, a trend vividly exemplified by the technological advancements detailed in patent CN103102357A regarding the synthesis of Cefuroxime sodium. This second-generation cephalosporin antibiotic remains a cornerstone in treating lower respiratory tract and urinary system infections, necessitating robust and scalable production methods that ensure both safety and efficacy. The disclosed innovation fundamentally re-engineers the salt formation and crystallization steps by substituting hazardous organic solvents with benign aqueous ethanol systems, thereby addressing critical environmental and operational pain points inherent in legacy manufacturing protocols. By leveraging sodium acetate trihydrate as a superior reacting agent compared to traditional sodium iso-octoate, this methodology not only enhances the physicochemical properties of the final API but also streamlines the downstream processing workflow for industrial applications. For R&D directors and process engineers, understanding the nuances of this transition from acetonitrile-based to ethanol-water-based crystallization is paramount for evaluating potential technology transfers or licensing opportunities. The structural integrity of the target molecule, as depicted below, relies heavily on the precise control of stereochemistry and salt formation during these final processing stages.
Furthermore, the strategic implementation of staged addition protocols and controlled temperature gradients during the crystallization phase offers a reproducible pathway to achieving high-purity standards required by global regulatory bodies. This patent represents a significant leap forward in the domain of reliable pharmaceutical intermediates supplier capabilities, providing a blueprint for cost reduction in antibiotic manufacturing without compromising on the stringent quality metrics demanded by modern pharmacopeias. As we delve deeper into the technical specifics, it becomes evident that this approach mitigates the risks associated with solvent toxicity while simultaneously boosting overall process throughput and yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefuroxime sodium has been plagued by reliance on anhydrous acetonitrile as the primary dissolution medium, a volatile organic compound that poses substantial health risks to production personnel and requires expensive containment infrastructure. Traditional protocols typically involve dissolving Cefuroxime acid in acetonitrile, cooling the mixture to approximately 10°C, and subsequently introducing a sodium iso-octoate acetone solution to induce salt formation and precipitation. This conventional route suffers from multiple critical deficiencies, including the generation of poor crystal forms that complicate suction filtration and drying operations, leading to extended production cycles and increased energy consumption. Moreover, the use of sodium iso-octoate often results in incomplete crystallization, leaving significant amounts of mother liquor residue which directly negatively impacts the weight yield, typically restricting it to a suboptimal range of merely 85% to 90%. Perhaps most concerning for supply chain managers is the poor color stability of the product derived from these older methods, where exposure to moderate temperatures and humidity accelerates discoloration, thereby shortening shelf life and increasing waste. The toxicity of acetonitrile further exacerbates operational costs due to the rigorous safety protocols and waste treatment procedures mandated for its handling and disposal.
The Novel Approach
In stark contrast, the innovative methodology outlined in the patent data introduces a paradigm shift by employing a 95% aqueous ethanol solution as the reaction medium, effectively eliminating the need for toxic acetonitrile and creating a significantly safer working environment for plant operators. This novel approach utilizes sodium acetate trihydrate as the sodium source, which interacts with the Cefuroxime acid dissolved in the ethanol-water matrix to promote the formation of well-defined, stable crystals that are far easier to filter and dry than their predecessors. The process involves a sophisticated temperature control strategy where the solution is maintained between 15°C and 18°C during the dropwise addition of the sodium salt, ensuring optimal nucleation and crystal growth kinetics. By implementing a staged addition technique—where half the volume of the sodium solution is added, followed by a stirring pause, and then the remainder—the method maximizes conversion efficiency and minimizes the inclusion of impurities within the crystal lattice. This results in a dramatic improvement in product quality, with yields consistently exceeding theoretical expectations and purity levels reaching upwards of 99.0% as verified by GC-External Standard methods. Ultimately, this green chemistry approach not only resolves the historical issues of poor filtration and color instability but also aligns perfectly with modern sustainability goals in high-purity API intermediate production.
Mechanistic Insights into Ethanol-Water Crystallization Dynamics
The core mechanistic advantage of this synthesis route lies in the solubility differential and nucleation behavior of Cefuroxime sodium within the ethanol-water binary solvent system compared to the traditional acetonitrile-acetone environment. In the aqueous ethanol medium, the polarity and hydrogen bonding network facilitate a more controlled desolvation process, allowing the Cefuroxime acid molecules to interact uniformly with the sodium ions provided by the sodium acetate trihydrate. The specific choice of sodium acetate trihydrate is critical, as it acts not merely as a base but as a structure-directing agent that influences the hydration state of the forming crystals, leading to a more robust lattice structure that resists degradation under thermal stress. The temperature window of 15°C to 18°C is meticulously selected to balance the supersaturation drive; temperatures too low might induce rapid, uncontrolled nucleation leading to fines and agglomeration, while temperatures too high could prevent adequate precipitation or lead to oiling out. The staged addition protocol serves as a kinetic regulator, preventing local spikes in supersaturation that could trap solvent molecules or impurities, thereby ensuring that the crystal growth proceeds in a thermodynamically favorable manner. This precise control over the crystallization micro-environment is what enables the achievement of such high purity and superior physical characteristics in the final bulk drug substance.
From an impurity control perspective, the washing step using aqueous ethanol plays a pivotal role in defining the final quality profile of the Cefuroxime sodium. Unlike acetone washes which might leave behind organic residues or fail to effectively remove inorganic salts, the ethanol-water wash efficiently solubilizes and removes excess sodium acetate trihydrate and any unreacted starting materials without dissolving the product itself. This selective solvation ensures that the final dried product is free from residual solvents that could pose toxicological risks, adhering strictly to ICH Q3C guidelines for residual solvents. Furthermore, the removal of surface-bound impurities during this washing phase contributes significantly to the observed enhancement in color stability, as many degradation pathways are catalyzed by trace metal ions or acidic residues that are effectively scrubbed away. The resulting crystal habit is less prone to hygroscopic uptake, which is a common failure mode for beta-lactam antibiotics, thus extending the shelf-life and maintaining potency during storage and transportation. This mechanistic understanding underscores why the new method is superior for the commercial scale-up of complex pharmaceutical intermediates where consistency is key.
How to Synthesize Cefuroxime Sodium Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to a strict sequence of operational parameters that govern the dissolution, reaction, and isolation phases of the process. The procedure begins with the preparation of a homogeneous solution of Cefuroxime acid in 95% aqueous ethanol, ensuring complete dissolution before initiating the cooling phase to bring the temperature down to the critical 12-18°C range. Following this, a pre-prepared solution of sodium acetate trihydrate in purified water is introduced dropwise into the reaction vessel, with vigilant monitoring of the internal temperature to maintain it within the narrow 15-18°C window throughout the addition. The detailed standardized synthesis steps, including specific stirring rates, addition times, and filtration parameters, are outlined in the structured guide below to ensure reproducibility and compliance with GMP standards.
- Dissolve Cefuroxime acid in 95% aqueous ethanol and cool the solution to a temperature range of 12-18°C under continuous stirring.
- Slowly add a prepared sodium acetate trihydrate solution dropwise while maintaining the crystallization temperature between 15-18°C, utilizing a staged addition protocol with intermediate stirring pauses.
- Filter the resulting wet product, wash thoroughly with aqueous ethanol to remove residual salts, and vacuum dry at temperatures below 40°C to obtain the final high-purity API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of acetonitrile, a solvent subject to fluctuating market prices and stringent environmental regulations, drastically simplifies the raw material sourcing landscape and reduces the regulatory burden associated with hazardous waste disposal. By switching to ethanol and water, facilities can leverage widely available, cost-effective commodities that are easier to handle, store, and recycle, leading to a substantial reduction in overall operating expenditures. The improved crystal morphology directly impacts downstream processing efficiency, as better filtration characteristics mean shorter cycle times and reduced energy usage for drying, which cumulatively enhances the throughput capacity of existing manufacturing assets. Additionally, the enhanced color stability of the final product reduces the risk of batch rejection due to specification failures, thereby securing supply continuity and minimizing the financial losses associated with off-spec inventory. These factors collectively position this technology as a highly attractive option for cost reduction in pharmaceutical intermediates manufacturing while simultaneously bolstering the resilience of the supply chain against regulatory and operational disruptions.
- Cost Reduction in Manufacturing: The substitution of expensive and toxic solvents like acetonitrile and acetone with economical ethanol and water significantly lowers the direct material costs associated with the production process. Furthermore, the use of sodium acetate trihydrate instead of sodium iso-octoate eliminates the need for specialized reagents that often carry a premium price tag due to complex synthesis or limited supplier bases. The improved filtration and drying characteristics of the new crystal form reduce the energy intensity of the isolation step, leading to lower utility bills and decreased maintenance requirements for processing equipment. By minimizing mother liquor residues and improving conversion rates, the process maximizes the utilization of the valuable Cefuroxime acid starting material, effectively lowering the cost per kilogram of the final API. These cumulative efficiencies create a robust economic model that supports competitive pricing strategies in the global generic antibiotic market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like ethanol and sodium acetate trihydrate mitigates the risk of supply disruptions that are common with specialty solvents or reagents that have single-source dependencies. The simplified process flow reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and more flexible response to sudden spikes in market demand for Cefuroxime sodium. Improved product stability means that inventory can be held for longer periods without degradation, providing a buffer against logistical delays and enabling more strategic stockpiling. The reduction in hazardous waste generation also streamlines the logistics of waste removal, preventing potential bottlenecks that could halt production lines due to full storage tanks or regulatory compliance issues. This reliability is crucial for maintaining long-term contracts with major pharmaceutical buyers who prioritize consistent delivery performance.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this synthesis route make it inherently scalable, as the safety profile of ethanol-water systems allows for larger batch sizes without the exponential increase in safety risks associated with flammable or toxic solvents. Facilities can expand production capacity with fewer modifications to ventilation and containment systems, accelerating the timeline for commercial scale-up of complex antibiotics. The reduced environmental footprint aligns with increasingly strict global emissions standards, future-proofing the manufacturing site against tightening regulations on volatile organic compound (VOC) emissions. Easier waste treatment protocols mean that effluent can be processed more efficiently, reducing the load on wastewater treatment plants and lowering the environmental levies imposed on the facility. This alignment with sustainability goals not only ensures regulatory compliance but also enhances the corporate reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Cefuroxime sodium synthesis method, drawing directly from the experimental data and comparative analysis provided in the patent literature. These insights are designed to clarify the operational benefits and quality improvements that distinguish this process from traditional manufacturing techniques used in the industry today. Understanding these distinctions is vital for stakeholders evaluating the feasibility of adopting this technology for their own production lines or supply networks.
Q: How does the new ethanol-water method improve upon traditional acetonitrile-based synthesis?
A: The novel method replaces toxic acetonitrile with a safer ethanol-water system, significantly reducing occupational health risks and eliminating the need for complex solvent recovery systems associated with volatile organic compounds.
Q: What are the advantages of using sodium acetate trihydrate over sodium iso-octoate?
A: Sodium acetate trihydrate promotes superior crystal morphology, facilitating easier filtration and drying processes while simultaneously enhancing the conversion rate and long-term color stability of the final Cefuroxime sodium product.
Q: Does this synthesis route impact the purity profile of the antibiotic intermediate?
A: Yes, the optimized washing protocol using aqueous ethanol effectively removes residual sodium acetate and impurities, consistently achieving purity levels exceeding 98.5% with improved stability against humidity and heat.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies like the one described in CN103102357A to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity Cefuroxime sodium that meets stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify every batch against international pharmacopeial standards. Our dedication to green chemistry and process optimization allows us to offer products that are not only cost-effective but also environmentally sustainable, reflecting our core values as a responsible chemical manufacturer.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our optimized supply chain solutions. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to quality and reliability as your trusted partner in antibiotic intermediate production.
