Advanced Nickel-Catalyzed Hydrocyanation Technology for Scalable Nylon Intermediate Production
Introduction to Advanced Hydrocyanation Technology
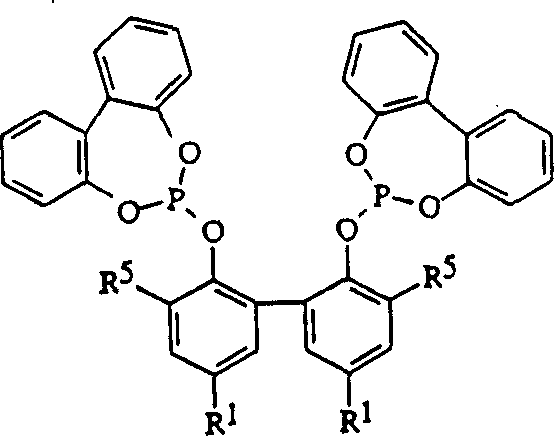
The chemical industry continuously seeks more efficient pathways for producing critical polymer precursors, and Patent CN1076342C represents a significant leap forward in the liquid phase hydrocyanation of diolefins. This intellectual property details an improved process for converting butadiene into non-conjugated acyclic nitriles, which are essential intermediates for manufacturing adiponitrile, the key monomer for Nylon 66. The core innovation lies in the utilization of zero-valent nickel complexes coordinated with novel multidentate phosphite ligands. Unlike traditional methods that often rely on monodentate ligands or require harsh Lewis acid promoters to activate unactivated olefins, this technology achieves high conversion rates and exceptional linearity under milder conditions. For R&D directors and process engineers, understanding the nuances of these ligand structures is paramount, as they dictate the steric and electronic environment around the nickel center, directly influencing the regioselectivity of the cyanide addition. This patent not only covers the hydrocyanation step but also provides a robust method for isomerizing the resulting branched nitriles into the desired linear 3- and 4-pentenenitriles, thereby maximizing atom economy and reducing waste in the overall value chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydrocyanation of butadiene has been performed using catalysts derived from monodentate phosphites, such as those described in early patents like US 3,496,215. While effective to a degree, these conventional systems often suffer from suboptimal selectivity, producing significant quantities of branched isomers like 2-methyl-3-butenenitrile (2M3BN) which are less useful for subsequent reactions leading to adiponitrile. Furthermore, the hydrocyanation of unactivated olefins typically necessitates the addition of Lewis acid promoters to enhance reaction rates, introducing additional complexity to the process workflow. The presence of these promoters can lead to corrosion issues in reactor vessels and requires careful downstream separation to prevent catalyst deactivation or product contamination. From a supply chain perspective, reliance on older catalyst technologies often means dealing with lower turnover numbers and higher catalyst loading requirements, which inflates the cost of goods sold and complicates waste management protocols due to the generation of phosphorus-containing byproducts that are difficult to recycle.
The Novel Approach
The methodology outlined in CN1076342C overcomes these historical bottlenecks by employing specifically designed multidentate phosphite ligands, such as those represented by Formula I through Formula XV. These ligands create a chelating effect that stabilizes the zero-valent nickel species, preventing aggregation and deactivation during the reaction cycle. A distinct advantage of this novel approach is the ability to conduct hydrocyanation and isomerization without the mandatory use of Lewis acid promoters for certain substrates, streamlining the reaction mixture and reducing the corrosive load on equipment. The patent demonstrates that these multidentate systems facilitate the isomerization of branched nitriles back into linear forms with high efficiency, effectively recycling what would otherwise be a low-value byproduct. This dual functionality—acting as both a hydrocyanation and isomerization catalyst—allows for a more integrated process design, potentially enabling single-reactor setups or simplified multi-stage continuous flow systems that significantly enhance operational throughput and reduce capital expenditure requirements for new production facilities.
Mechanistic Insights into Nickel-Catalyzed Isomerization
The catalytic cycle begins with the formation of an active zero-valent nickel species coordinated by the multidentate phosphite ligand. The steric bulk provided by substituents such as tert-butyl groups on the ligand framework plays a critical role in directing the incoming butadiene and HCN molecules to favor linear addition over branched addition. This steric control is essential for achieving the high ratios of linear pentenenitriles (3PN and 4PN) observed in the experimental data, where total yields often exceed 90% under optimized conditions. The electronic properties of the ligand, modulated by groups like methoxy or substituted phenyl rings, fine-tune the electron density at the nickel center, facilitating the oxidative addition of HCN and the subsequent migratory insertion steps. By carefully balancing these steric and electronic factors, the catalyst maintains high activity over extended periods, which is a crucial parameter for commercial viability. The mechanism also involves a dynamic equilibrium between different nickel-hydride and nickel-nitrile intermediates, allowing for the rapid isomerization of double bonds within the nitrile chain to thermodynamically favored linear positions.
Following the initial hydrocyanation, the process addresses the inevitable formation of 2-methyl-3-butenenitrile (2M3BN) through a dedicated isomerization step. As illustrated in the reaction schemes, the same nickel catalyst system effectively migrates the double bond in 2M3BN to produce 3-pentenenitrile (3PN) and 4-pentenenitrile (4PN). This transformation is vital because linear pentenenitriles are the direct precursors for the second hydrocyanation step that yields adiponitrile. The patent data indicates that this isomerization can proceed efficiently at temperatures ranging from 60°C to 150°C, with the catalyst demonstrating remarkable stability even in the presence of free nitriles which typically act as catalyst poisons in other systems. The ability to convert branched isomers back into the linear stream ensures that the overall carbon efficiency of the plant is maximized, minimizing the need for purging or incinerating off-spec material. This mechanistic robustness provides a strong foundation for scaling the process from laboratory benchtop experiments to multi-ton industrial reactors without losing selectivity or activity.
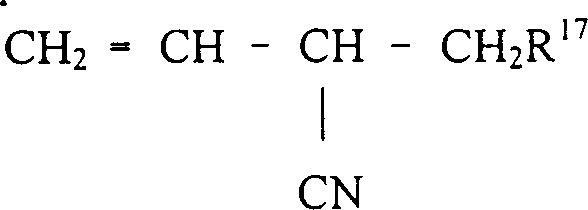
How to Synthesize Linear Pentenenitriles Efficiently
Implementing this technology requires precise control over catalyst preparation and reaction conditions to fully realize the benefits described in the patent. The synthesis begins with the preparation of the chlorophosphite intermediates, which are then reacted with biphenols or naphthols to form the final multidentate ligands. These ligands are subsequently mixed with a zero-valent nickel source, such as Ni(COD)2, in an inert solvent like propionitrile or toluene to generate the active catalyst solution. The hydrocyanation reaction is typically conducted in a sealed vessel under an inert atmosphere to prevent oxidation of the sensitive nickel center, with temperature and pressure carefully regulated to maintain the liquid phase of the reactants. For procurement and operations teams, understanding these specific synthesis parameters is crucial for sourcing the correct raw materials and designing the appropriate containment systems. The detailed standardized synthesis steps see the guide below.
- Prepare the catalyst precursor by mixing zero-valent nickel sources like Ni(COD)2 with multidentate phosphite ligands in an inert solvent.
- React 1,3-butadiene with hydrogen cyanide in the presence of the catalyst at temperatures between 0°C and 150°C.
- Isomerize the resulting non-conjugated nitriles to linear 3- and 4-pentenenitriles using the same nickel catalyst system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers substantial strategic benefits beyond mere chemical yield improvements. The elimination or reduction of Lewis acid promoters translates directly into lower raw material costs and reduced expenditure on corrosion-resistant alloys for reactor construction. Furthermore, the high selectivity for linear products means that downstream purification units, such as distillation columns, can be operated with lower energy consumption since there is less need to separate complex mixtures of branched isomers. This efficiency gain contributes to a significantly reduced carbon footprint for the manufacturing site, aligning with modern sustainability goals and regulatory compliance standards. The robustness of the catalyst system also implies longer campaign lengths between maintenance shutdowns, enhancing overall equipment effectiveness and ensuring a more reliable supply of critical intermediates to downstream polymer producers.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous Lewis acid promoters in many instances, which simplifies the bill of materials and reduces waste disposal costs associated with acidic byproducts. Additionally, the high turnover number of the nickel catalyst means that less precious metal is required per unit of product, driving down the variable cost of production substantially. By maximizing the conversion of branched byproducts into valuable linear intermediates, the process effectively increases the yield from the same amount of butadiene feedstock, providing a direct margin improvement that compounds over large production volumes.
- Enhanced Supply Chain Reliability: The use of stable multidentate ligands reduces the sensitivity of the catalyst to impurities, allowing for the use of commercially available grade solvents and feedstocks without extensive pre-purification. This flexibility mitigates the risk of supply disruptions caused by shortages of ultra-high-purity reagents. Moreover, the ability to operate the isomerization and hydrocyanation steps in a integrated manner reduces the number of unit operations required, simplifying the logistics of intermediate storage and transfer within the plant. This streamlined workflow ensures a consistent and predictable output of adiponitrile precursors, which is critical for maintaining just-in-time delivery schedules to nylon manufacturers.
- Scalability and Environmental Compliance: The liquid phase nature of the reaction facilitates easy scale-up from pilot plants to full commercial production using standard chemical engineering practices. The reduced generation of hazardous waste and the potential for catalyst recycling align with increasingly stringent environmental regulations regarding heavy metal discharge and volatile organic compound emissions. Implementing this technology positions the manufacturing facility as a leader in green chemistry, potentially qualifying for environmental incentives and improving the corporate sustainability profile which is increasingly important for securing contracts with major global polymer brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrocyanation technology. These answers are derived from the specific embodiments and data presented in the patent documentation, providing clarity on operational parameters and expected outcomes. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into existing infrastructure or new greenfield projects. The responses cover catalyst stability, feedstock requirements, and product quality specifications relevant to industrial applications.
Q: What is the primary advantage of multidentate phosphite ligands in hydrocyanation?
A: They provide superior selectivity for linear nitriles over branched isomers without requiring Lewis acid promoters, simplifying the process.
Q: Can this process be scaled for industrial adiponitrile production?
A: Yes, the patent describes liquid phase processes suitable for continuous operation, which are essential for commercial scale-up.
Q: How does the isomerization step improve yield?
A: It converts less desirable branched 2-methyl-3-butenenitrile into valuable linear 3- and 4-pentenenitriles, maximizing overall feedstock efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adiponitrile Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable catalytic processes in the production of high-value chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and risk-mitigated. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Whether you require custom synthesis of specific phosphite ligands or the toll manufacturing of pentenenitrile derivatives, our infrastructure is designed to meet the demanding requirements of the global polymer and specialty chemical markets.
We invite you to engage with our technical procurement team to discuss how this advanced hydrocyanation technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this ligand system. We encourage you to contact us for specific COA data and route feasibility assessments to validate the compatibility of this process with your current supply chain architecture. Let us partner with you to optimize your manufacturing efficiency and secure a competitive advantage in the nylon intermediate sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
