Scaling High-Purity Beta-Arbutin Production: A Technical Analysis of Green Glycosylation Strategies
Scaling High-Purity Beta-Arbutin Production: A Technical Analysis of Green Glycosylation Strategies
The global demand for effective and safe skin-whitening agents has placed beta-arbutin at the forefront of the cosmetic and pharmaceutical intermediate markets. As regulatory pressures mount and sustainability becomes a non-negotiable metric for procurement, the industry requires synthesis routes that balance high purity with environmental stewardship. Patent CN103524575A introduces a transformative approach to beta-arbutin production, shifting away from resource-intensive crystallization protocols toward a continuous extraction and solvent-recycling paradigm. This technical insight report analyzes the mechanistic advantages of this method, specifically focusing on how the elimination of intermediate isolation steps enhances overall process efficiency. By leveraging a biphasic system involving high-boiling polar aprotic solvents and low-polarity extractants, manufacturers can achieve a substantial reduction in raw material consumption while maintaining stringent quality standards required for high-purity beta-arbutin.
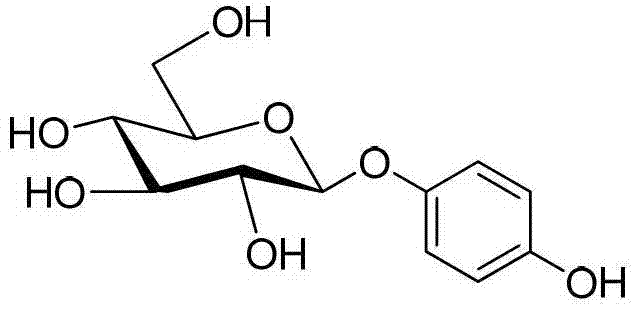
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of beta-arbutin has been bottlenecked by the inefficiencies inherent in the preparation of the glycosyl donor, pentaacetylglucose. Conventional protocols typically mandate the isolation and purification of this intermediate via crystallization, a step that is inherently lossy and generates significant volumes of mother liquor waste. In these traditional workflows, the acetylation of glucose often requires a massive excess of acetic anhydride, ranging from 6 to 10 molar equivalents, to drive the reaction to completion. Following the reaction, the crude mixture must be cooled and crystallized, a process where the yield of pure beta-pentaacetylglucose rarely exceeds 80%. The remaining 20% of the material, which includes unreacted glucose, partially acetylated species, and the alpha-anomer, is frequently discarded as hazardous waste. Furthermore, the reliance on stoichiometric amounts of reagents and the inability to recycle the reaction solvent effectively drives up the cost of goods sold (COGS), making the final API intermediate less competitive in a price-sensitive market.
The Novel Approach
The methodology outlined in the patent data proposes a radical departure from batch-wise isolation by integrating the acetylation and glycosylation steps through a strategic liquid-liquid extraction technique. Instead of isolating solid pentaacetylglucose, the reaction mixture is treated with a low-polarity aprotic solvent, such as toluene, which selectively extracts the fully acetylated product while leaving the polar catalyst, unreacted glucose, and acetic acid in the high-boiling polar phase. This telescoping strategy eliminates the energy-intensive drying and recrystallization steps associated with the intermediate. Crucially, the polar phase, which retains the sodium acetate catalyst, can be regenerated by removing the generated acetic acid via azeotropic distillation and then directly reused for the next batch of acetylation. This closed-loop solvent and catalyst system not only minimizes the discharge of three wastes (wastewater, waste gas, and solid waste) but also theoretically pushes the utilization rate of glucose and acetic anhydride toward 100%, representing a paradigm shift in cost reduction in skin whitening agent manufacturing.
Mechanistic Insights into Sodium Acetate Catalyzed Acetylation and Extraction
The core innovation of this process lies in the differential solubility profiles of the reaction components within a biphasic solvent system. The acetylation of glucose is catalyzed by sodium acetate in a high-boiling polar aprotic solvent like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). Under these conditions, the reaction proceeds efficiently at temperatures between 60°C and 120°C. The mechanistic advantage arises post-reaction, where the addition of a non-polar solvent like toluene creates a distinct phase separation. Pentaacetylglucose, being significantly less polar due to the masking of hydroxyl groups with acetyl moieties, partitions preferentially into the toluene layer. Conversely, the sodium acetate catalyst, being ionic, remains strictly in the polar DMF or DMSO phase. Unreacted glucose and partially acetylated intermediates, which retain free hydroxyl groups, also exhibit higher affinity for the polar phase due to hydrogen bonding capabilities. This selective partitioning acts as a continuous purification step, ensuring that only the desired fully acetylated species proceeds to the glycosylation stage, thereby simplifying the impurity profile of the final product.
Following the extraction, the toluene layer containing the crude pentaacetylglucose is washed with water to remove trace acids and then subjected to glycosylation with hydroquinone derivatives using a Lewis acid catalyst such as boron trifluoride etherate. The stereochemical outcome is critical here, as the beta-anomer is the biologically active form required for tyrosinase inhibition. The reaction conditions, typically maintained between 0°C and 40°C, favor the formation of the beta-glycosidic bond. Subsequent deacetylation using methanol and ammonia or sodium methoxide yields the final beta-arbutin. The robustness of this mechanism allows for the recycling of the polar solvent phase; by removing the acetic acid byproduct through toluene azeotropic distillation, the catalyst concentration is preserved, and the solvent volume is maintained. This reduces the need for fresh solvent charging in every cycle, directly addressing the commercial imperative for sustainable chemical manufacturing and reducing the environmental footprint of the facility.
How to Synthesize Beta-Arbutin Efficiently
Implementing this improved synthesis route requires precise control over solvent ratios and temperature gradients to maximize the efficiency of the extraction and recycling loops. The process begins with the acetylation of glucose in DMF or DMSO, followed by a critical extraction step where the volume ratio of the extracting solvent to the reaction solvent must be optimized to ensure complete transfer of the product. Detailed operational parameters, including specific molar ratios of acetic anhydride to glucose and the precise timing for catalyst replenishment, are essential for maintaining consistent batch-to-batch quality. For R&D teams looking to adopt this technology, understanding the phase behavior of the acetylated sugars is paramount to preventing the co-extraction of impurities. The standardized synthetic steps below outline the critical path from raw materials to the final purified crystal, ensuring that the theoretical yield improvements described in the patent are realized in practical application.
- Acetylate glucose using acetic anhydride and sodium acetate in a high-boiling polar aprotic solvent like DMF or DMSO at 60-120°C.
- Extract the resulting pentaacetylglucose into a low-polarity solvent such as toluene, leaving unreacted glucose and catalyst in the polar phase.
- React the toluene extract directly with hydroquinone derivatives using a Lewis acid catalyst, followed by deacetylation to yield pure beta-arbutin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this extraction-based synthesis protocol offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage is the drastic simplification of the supply chain for raw materials; by enabling the recycling of the high-boiling polar solvent and the sodium acetate catalyst, the facility reduces its dependency on continuous large-volume solvent deliveries. This consolidation of material flows enhances supply chain reliability and mitigates the risk of production stoppages due to solvent shortages. Furthermore, the elimination of the intermediate crystallization step reduces the overall processing time per batch, effectively increasing the throughput capacity of existing reactor trains without the need for capital expenditure on new equipment. The reduction in waste generation also translates to lower costs associated with waste treatment and disposal, a significant factor in regions with stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant reduction in the consumption of acetic anhydride and the elimination of losses associated with intermediate purification. Traditional methods often suffer from yield losses of up to 20% during the crystallization of pentaacetylglucose, whereas this telescoped method retains the material in solution for immediate downstream processing. Additionally, the ability to recycle the catalyst and solvent means that the variable cost per kilogram of product decreases substantially over multiple batches. By avoiding the purchase of fresh solvent and catalyst for every run, manufacturers can achieve a leaner cost structure that provides a competitive edge in the global market for cosmetic ingredients.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reduced volume of waste and the recycling of key process fluids simplify the inventory management of the production site. The process is less sensitive to fluctuations in the purity of starting materials because the extraction step acts as a buffer, separating the product from unreacted glucose which can be re-acetylated in the recycled polar phase. This robustness ensures a more consistent output of high-purity beta-arbutin, reducing the incidence of out-of-specification batches that can disrupt supply commitments to downstream formulators. The simplified workflow also reduces the operational complexity, making it easier to train personnel and maintain consistent production schedules.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the use of common industrial solvents like toluene and DMF, which are well-understood in terms of handling and safety. The reduction in three wastes (wastewater,废气, and solids) aligns with modern green chemistry principles, making the facility more compliant with evolving environmental standards. The ability to operate with a closed-loop solvent system minimizes the release of volatile organic compounds (VOCs) and reduces the load on wastewater treatment plants. This environmental stewardship not only lowers regulatory compliance costs but also enhances the brand reputation of the manufacturer as a sustainable partner for global cosmetic and pharmaceutical brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved beta-arbutin synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational realities. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers highlight the specific advantages of the extraction-recycling loop over traditional batch processing.
Q: How does the new extraction method improve yield compared to traditional crystallization?
A: Traditional methods rely on crystallizing pentaacetylglucose, which traps mother liquor and limits yield to around 60%. The new extraction method transfers the product directly into an organic phase, theoretically allowing near 100% utilization of glucose and acetic anhydride.
Q: Can the polar solvent system be recycled in this process?
A: Yes, the high-boiling polar solvent (DMF or DMSO) remaining after extraction contains the catalyst and unreacted materials. By removing acetic acid via azeotropic distillation, this phase can be reused for subsequent batches, drastically reducing waste.
Q: What represents the primary cost driver in conventional arbutin synthesis?
A: The excessive consumption of acetic anhydride (often 6-10 equivalents) and the loss of material during the purification of the pentaacetylglucose intermediate are the main cost drivers, both of which are addressed by this telescoped protocol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Arbutin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is essential for maintaining competitiveness in the fine chemical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the extraction-telescoping method for beta-arbutin are implemented with precision and safety. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the beta-anomeric purity and residual solvent levels. Our infrastructure is designed to support the complex solvent recycling loops required by this process, guaranteeing a stable and continuous supply of high-quality intermediates for our global partners.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing capability can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical performance data rather than theoretical projections. Let us collaborate to bring superior, sustainably produced beta-arbutin to your formulation pipeline.
