Industrial Scale Synthesis of Gemcitabine Hydrochloride via Stable Silyl Protection Strategies
Industrial Scale Synthesis of Gemcitabine Hydrochloride via Stable Silyl Protection Strategies
The pharmaceutical industry continuously demands more robust and scalable pathways for critical oncology agents, and the synthesis of Gemcitabine Hydrochloride stands as a prime example where process optimization directly impacts supply security. Patent CN101492482A introduces a transformative industrial production method that addresses the long-standing instability issues associated with traditional protecting group strategies. By strategically employing tert-butyldiphenylsilyl (TBDPS) chloride alongside benzoyl chloride, this novel route significantly enhances the stability of key intermediates, thereby mitigating the risks of premature deprotection that have historically plagued manufacturing yields. This technical breakthrough not only ensures a final product purity exceeding 99.8% but also establishes a more predictable and controllable reaction environment suitable for multi-ton scale-up. For procurement and R&D leaders, understanding the mechanistic advantages of this silyl-protection strategy is essential for securing a reliable gemcitabine intermediate supplier capable of meeting stringent global quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Gemcitabine has relied heavily on double benzoyl protection strategies to mask hydroxyl functionalities during the construction of the furanose ring. However, this conventional approach suffers from a critical chemical vulnerability: the resulting dibenzoyl-protected intermediates exhibit significant instability under alkaline conditions. As illustrated in the reaction scheme below, the reliance on solely benzoyl groups creates a fragile molecular architecture that is prone to hydrolysis during subsequent processing steps, particularly during the glycosylation and workup phases. This instability often leads to the formation of complex impurity profiles, including partially deprotected species that are difficult to separate from the desired product. Consequently, manufacturers face inconsistent yields and compromised product quality, necessitating extensive and costly purification protocols that erode profit margins and extend lead times for high-purity nucleoside analogs.
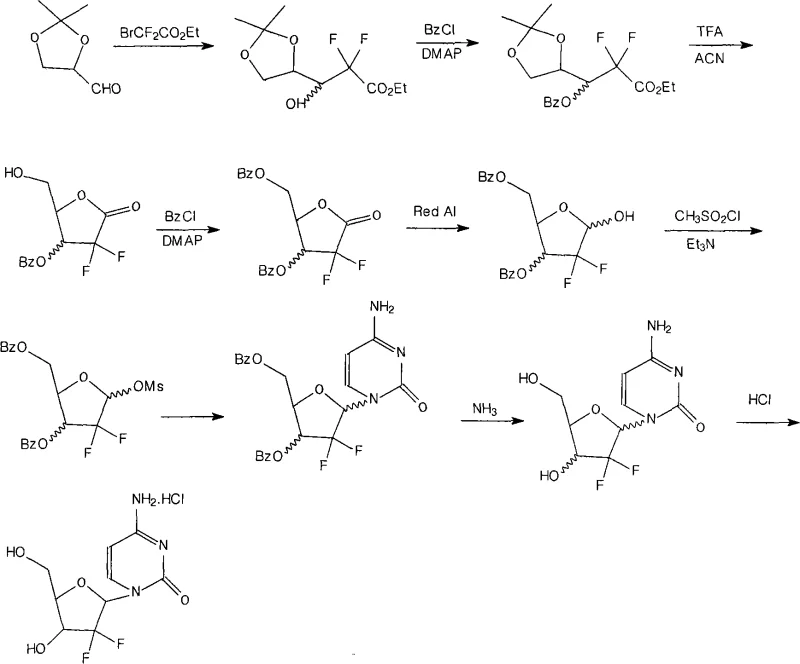
The Novel Approach
In stark contrast to the fragile nature of prior art methods, the novel process detailed in CN101492482A implements a hybrid protection strategy that fundamentally alters the stability profile of the synthetic intermediates. By introducing a bulky TBDPS group at the 5-position prior to benzoylation at the 3-position, the process creates a sterically hindered environment that shields the molecule from nucleophilic attack during basic workups. The reaction pathway begins with a Reformatsky-type addition to a protected glyceraldehyde, followed by cyclization to a lactone, which is then selectively silylated. This strategic modification ensures that the intermediate remains intact throughout the rigorous conditions of reduction, mesylation, and condensation with cytosine. The result is a streamlined workflow that minimizes side reactions and facilitates easier isolation of the target compound, representing a significant leap forward in cost reduction in API manufacturing for this critical anticancer drug.
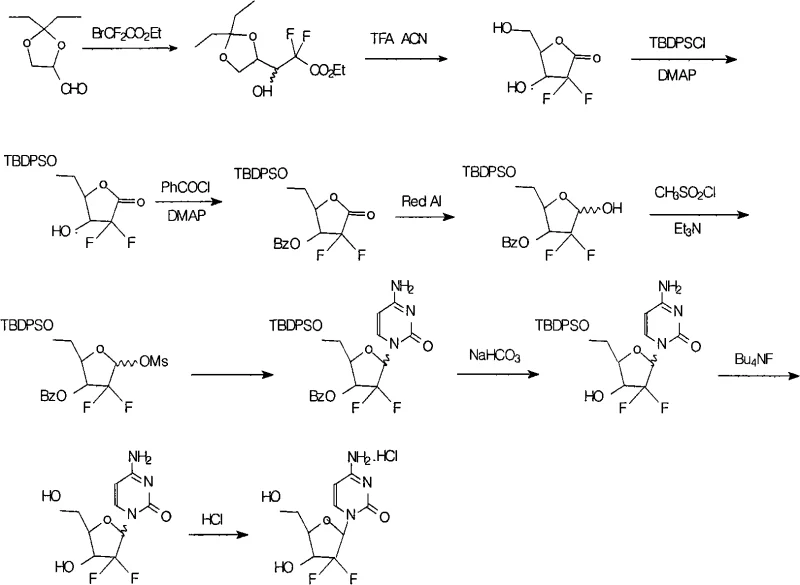
Mechanistic Insights into Selective Silylation and Glycosylation
The core innovation of this synthesis lies in the precise orchestration of protecting group chemistry, specifically the differential reactivity between the silyl ether and the benzoate ester. The mechanism initiates with the formation of a fluorinated lactone, which serves as the electrophilic scaffold for subsequent functionalization. The introduction of the TBDPS group is kinetically favored at the primary hydroxyl position due to lower steric hindrance compared to the secondary hydroxyl, allowing for high regioselectivity without the need for complex separation techniques. Once the 5-position is secured with the robust silyl moiety, the 3-position is capped with a benzoyl group. This dual-protection motif is chemically orthogonal; the benzoyl group can be removed under mild basic conditions (using sodium bicarbonate and trifluoroethanol) without affecting the silyl ether, while the silyl group can be cleaved later using fluoride sources like tetrabutylammonium fluoride (TBAF). This orthogonality provides process chemists with granular control over the deprotection sequence, ensuring that the sensitive glycosidic bond formed with cytosine remains uncompromised.
Furthermore, the glycosylation step itself is optimized through the activation of cytosine using hexamethyldisilazane (HMDS) and ammonium sulfate, generating a highly reactive silylated nucleobase in situ. This activated species attacks the anomeric methanesulfonate (mesylate) intermediate with high stereoselectivity, favoring the formation of the beta-anomer required for biological activity. The presence of the bulky TBDPS group at the 5-position exerts a conformational influence on the furanose ring, potentially enhancing the stereoelectronic effects that drive the preferential formation of the desired stereoisomer. By minimizing the formation of the alpha-anomer and other diastereomeric impurities at the source, the process reduces the burden on downstream purification, directly contributing to the reported total yield of 17.9% and exceptional purity levels. This level of mechanistic control is vital for any entity seeking to become a reliable supplier of complex pharmaceutical intermediates.
How to Synthesize Gemcitabine Hydrochloride Efficiently
The execution of this synthesis requires strict adherence to the defined sequence of protection, functionalization, and deprotection to maximize yield and purity. The process begins with the preparation of the fluorinated lactone scaffold, followed by the critical selective silylation step that defines the novelty of this route. Operators must maintain precise temperature controls during the mesylation and glycosylation steps to prevent degradation of the activated intermediates. The final deprotection sequence involves a two-stage removal of the protecting groups—first the benzoyl esters under mild basic conditions, followed by the cleavage of the silyl ether with fluoride—before final salt formation with hydrochloric acid. For a comprehensive breakdown of the specific reagents, stoichiometry, and reaction conditions required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Perform Reformatsky-type addition on 2,3-O-isopropylidene-D-glyceraldehyde followed by acetonide deprotection to form the fluorinated lactone.
- Execute selective protection using TBDPSCl followed by benzoylation to create a stable dual-protected intermediate resistant to alkaline conditions.
- Reduce the lactone, mesylate the anomeric position, couple with cytosine, and perform sequential deprotection to yield high-purity Gemcitabine HCl.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this TBDPS-based synthesis route offers profound advantages for supply chain resilience and cost management. Traditional methods that suffer from intermediate instability often result in batch-to-batch variability, forcing manufacturers to hold excessive safety stock or face unexpected production delays. By stabilizing the key intermediate, this new process drastically reduces the risk of batch failure, ensuring a more consistent and predictable output volume. This reliability is paramount for procurement managers who need to secure long-term contracts for essential oncology ingredients without the fear of supply interruptions caused by complex purification bottlenecks. The ability to consistently deliver material that meets tight specifications translates directly into reduced operational overhead and a more agile response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of instability-related losses fundamentally alters the cost structure of Gemcitabine production. In conventional processes, the degradation of double-benzoyl intermediates necessitates expensive reprocessing or results in significant yield loss, driving up the cost per kilogram. By preventing this degradation, the novel route maximizes the conversion of raw materials into finished product, effectively lowering the material cost basis. Furthermore, the simplified purification profile reduces the consumption of chromatography media and solvents, which are major cost drivers in nucleoside manufacturing. These efficiencies accumulate to provide substantial cost savings without compromising the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthesizing fluorinated nucleosides, where minor deviations can render a batch unusable. The robustness of the TBDPS protection strategy acts as a buffer against process variability, making the manufacturing line more forgiving and easier to control at scale. This increased process robustness means that production schedules can be adhered to with greater confidence, reducing lead time for high-purity pharmaceutical intermediates. For global buyers, this translates to a more dependable source of supply, mitigating the risks associated with single-source dependencies or volatile production outputs from less optimized facilities.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this optimized route offers a cleaner profile due to higher selectivity and fewer side products. The reduction in impurity load means less solvent is required for recrystallization and washing, aligning with modern green chemistry principles and reducing the environmental footprint of the operation. Additionally, the use of standard, commercially available reagents like TBDPSCl and benzoyl chloride ensures that the supply chain for raw materials is secure and not dependent on exotic or hard-to-source catalysts. This accessibility facilitates seamless commercial scale-up of complex nucleoside analogs from pilot plant to multi-ton industrial reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios or for qualifying new suppliers.
Q: Why is TBDPS protection superior to double benzoyl protection in Gemcitabine synthesis?
A: Conventional double benzoyl intermediates are unstable under alkaline conditions, leading to premature deprotection and reduced yield. The TBDPS (tert-butyldiphenylsilyl) group provides superior steric bulk and chemical stability, preventing unwanted side reactions during the critical glycosylation and workup stages.
Q: What is the expected purity profile for this synthesis route?
A: The patented process achieves a final product purity exceeding 99.8% HPLC area normalization. The enhanced stability of the silyl-protected intermediate minimizes the formation of deprotected impurities, simplifying the final crystallization and purification steps.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the route is specifically designed for industrial production. It utilizes robust reagents like activated zinc and standard protecting group chemistry (TBDPS/Bz) that are scalable from kilogram to multi-ton quantities while maintaining consistent stereochemical control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the TBDPS protection strategy are fully realized in a GMP-compliant environment. We understand that the synthesis of fluorinated nucleosides demands rigorous QC labs and stringent purity specifications to meet the exacting standards of global regulatory bodies. By leveraging our state-of-the-art infrastructure and process chemistry knowledge, we are positioned to deliver Gemcitabine intermediates and APIs that consistently exceed the 99.8% purity benchmark established by this patented technology.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can enhance your supply chain efficiency. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven capability. Partnering with us ensures not just a product, but a strategic alliance focused on long-term value creation and supply security in the competitive oncology market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
