Advanced Stereoselective Synthesis of Gemcitabine Hydrochloride for Commercial Scale-up
Advanced Stereoselective Synthesis of Gemcitabine Hydrochloride for Commercial Scale-up
The pharmaceutical landscape for oncology treatments continues to evolve, with Gemcitabine Hydrochloride remaining a cornerstone therapy for pancreatic, lung, and breast cancers. The efficiency of its production directly impacts global supply chain stability and patient access. Patent CN101628927A introduces a transformative method for the three-dimensionally selective preparation of 2'-deoxy-2',2'-difluoro-Beta-cytidine hydrochloride. This technical insight report analyzes the proprietary synthesis route which utilizes 1,3,5-tri-O-benzoyl-alpha-D-ribofuranose as a chiral pool starting material. By leveraging this specific precursor, the process bypasses the notorious stereochemical pitfalls of earlier generations of synthesis, offering a streamlined pathway to high-purity intermediates. For R&D directors and procurement leaders, understanding this mechanistic advantage is critical for securing a reliable API intermediate supplier capable of meeting stringent regulatory standards.
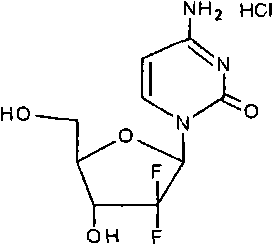
The structural integrity of the final molecule, as depicted above, relies heavily on the precise placement of the fluorine atoms at the 2' position. The patent details a method that not only achieves this but does so with exceptional stereocontrol, ensuring the beta-anomer is the predominant product. This level of control is not merely an academic exercise; it is a commercial imperative that reduces the burden on downstream purification processes. By addressing the root cause of isomeric impurities at the beginning of the synthetic sequence, manufacturers can significantly reduce waste and improve overall process mass intensity. This report will dissect the technical nuances of this approach, providing a clear roadmap for those seeking cost reduction in pharmaceutical manufacturing through superior process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gemcitabine and its precursors has been plagued by stereochemical inefficiencies, particularly those relying on the Reformatsky reaction. In traditional pathways, the construction of the difluoro-ribofuranose core often results in a non-selective mixture of isomers. Documentation indicates that these conventional methods can generate over 30% of the unwanted xylo-isomer alongside the desired ribo-isomer. This lack of selectivity creates a severe bottleneck in production, as the physical properties of these isomers are often too similar for efficient separation via standard crystallization techniques. Consequently, manufacturers are forced to employ extensive chromatographic purification or repeated recrystallization cycles, which drastically lowers overall yield and escalates production costs. Furthermore, the reliance on D-glyceraldehyde derivatives in older routes introduces additional steps and handling complexities that hinder the commercial scale-up of complex nucleosides.
The Novel Approach
In stark contrast, the methodology outlined in CN101628927A leverages a chiral starting material, 1,3,5-tri-O-benzoyl-alpha-D-ribofuranose, to dictate the stereochemical outcome from the outset. This strategic choice eliminates the need for difficult isomer separations later in the process. The route is concise, comprising only four key steps from the protected sugar to the final deprotected nucleoside. By maintaining the stereochemical integrity of the alpha-anomer through the oxidation and fluorination stages, the process sets the stage for a highly selective glycosylation reaction. The result is a synthesis that is not only shorter but also more robust and predictable. This approach represents a significant advancement for any entity acting as a reliable agrochemical intermediate supplier or pharma partner, as it translates directly into higher throughput and reduced operational risk.
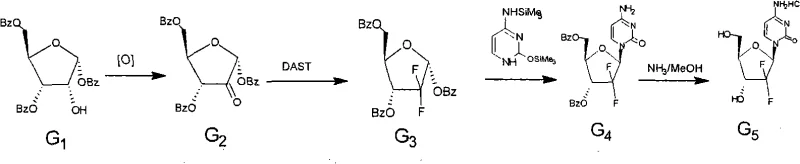
Mechanistic Insights into Stereoselective Fluorination and Glycosylation
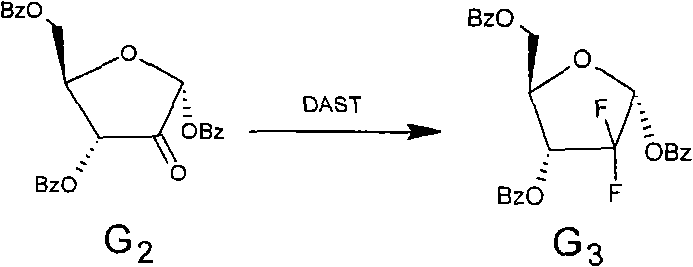
The core innovation of this patent lies in the precise manipulation of the sugar moiety, specifically the oxidation and subsequent fluorination steps. The oxidation of the 2-hydroxyl group to a ketone is achieved using mild yet effective reagents such as Dess-Martin periodinane or Swern oxidation conditions. This step is critical as it prepares the molecule for the introduction of the fluorine atoms. The subsequent fluorination utilizes a synergistic mixture of DAST (diethylaminosulfur trifluoride) and triethylamine-hydrogen fluoride. This specific combination is superior to using DAST alone, as it enhances transformation efficiency and shortens reaction times while maintaining the stereochemical configuration. The mechanism ensures that the alpha-configuration of the starting sugar is preserved in the difluoro-intermediate, which is a prerequisite for the final stereochemical inversion.
Following the formation of the difluoro-sugar, the glycosylation step employs a classic SN2 substitution mechanism. The alpha-anomer of the difluoro-ribofuranose reacts with bis-trimethylsilyl cytosine in the presence of a Lewis acid catalyst such as stannic chloride. Because the starting sugar is predominantly the alpha-anomer, the nucleophilic attack by the cytosine base occurs from the opposite side, resulting in the inversion of configuration to yield the desired beta-anomer product. This mechanistic certainty is the key to the process's high purity profile. By controlling the anomeric ratio of the sugar precursor, the process effectively eliminates the formation of the alpha-nucleoside impurity, which is notoriously difficult to remove. This level of mechanistic control is essential for producing high-purity oncology intermediates that meet the rigorous specifications of global regulatory bodies.
How to Synthesize Gemcitabine Hydrochloride Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions, particularly regarding moisture control and temperature management during the fluorination and glycosylation stages. The process begins with the oxidation of the protected ribofuranose, followed by the critical fluorination step which must be conducted under anhydrous conditions to prevent hydrolysis of the reactive intermediates. The subsequent coupling with the cytosine base requires precise stoichiometry and catalyst loading to ensure complete conversion. Finally, the deprotection of the benzoyl groups is achieved under mild basic conditions, followed by salt formation to yield the stable hydrochloride salt. For detailed operational parameters, safety data, and specific workup procedures, please refer to the standardized synthesis guide below.
- Oxidize 1,3,5-tri-O-benzoyl-2-deoxy-2,2-difluoro-alpha-D-ribofuranose using Dess-Martin reagent or Swern conditions to form the 2-ketone intermediate.
- Perform fluorination on the ketone intermediate using a synergistic mixture of DAST and triethylamine-hydrogen fluoride to yield the alpha-difluoro sugar.
- Execute stereoselective glycosylation with bis-trimethylsilyl cytosine using Lewis acid catalysis, followed by deprotection to obtain the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this stereoselective route offers profound advantages for supply chain resilience and cost management. The primary benefit stems from the elimination of complex purification steps required to separate isomeric impurities. In traditional manufacturing, the removal of the xylo-isomer can consume a significant portion of the production budget and time. By designing a route that inherently avoids the formation of these impurities, the process drastically simplifies the downstream workflow. This simplification translates into reduced solvent consumption, lower waste disposal costs, and a smaller environmental footprint. For procurement managers, this means a more stable cost structure that is less susceptible to fluctuations in raw material prices or waste treatment fees.
- Cost Reduction in Manufacturing: The streamlined nature of this four-step synthesis significantly lowers the operational expenditure associated with Gemcitabine production. By avoiding the use of expensive chiral resolution techniques or extensive chromatography, the process achieves substantial cost savings. The use of readily available chiral starting materials further anchors the cost base, preventing volatility. Additionally, the high yield and purity reduce the need for re-processing batches, ensuring that capital and resources are utilized with maximum efficiency. This economic efficiency is vital for maintaining competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations enhances the reliability of the supply chain. The reagents used, such as DAST and Lewis acids, are commercially available in bulk quantities, reducing the risk of supply disruptions. Furthermore, the shorter synthetic route reduces the overall lead time for production cycles. This agility allows manufacturers to respond more quickly to changes in market demand, ensuring a continuous supply of critical oncology medications. For supply chain heads, this reliability is a key metric in vendor selection and risk mitigation strategies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous heavy metal catalysts in certain steps and the reduction in solvent intensity align with modern green chemistry principles. This facilitates easier regulatory approval and environmental compliance in various jurisdictions. The ability to scale this process from 100 kgs to 100 MT annual commercial production without significant re-engineering makes it an ideal candidate for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Gemcitabine Hydrochloride via this specific patent route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. They are intended to provide clarity on the process capabilities and quality assurances associated with this manufacturing method. Understanding these details is crucial for technical teams evaluating the feasibility of this route for their specific product portfolios.
Q: How does this patent route improve optical purity compared to Reformatsky methods?
A: Traditional Reformatsky reactions generate significant isomeric impurities (>30%) that are difficult to separate. This patent utilizes a chiral starting material (G1) that ensures the formation of the alpha-anomer intermediate, which subsequently drives the stereoselective formation of the desired beta-anomer product during glycosylation, drastically reducing impurity burden.
Q: What are the critical reaction conditions for the fluorination step?
A: The process employs a mixture of DAST and triethylamine-hydrogen fluoride in anhydrous methylene chloride. The temperature is strictly controlled between -10°C and 80°C, with a reaction time of 1 to 12 hours, ensuring high transformation efficiency while minimizing side reactions common with single-agent fluorination.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. It uses readily available chiral starting materials, avoids complex chromatographic separations of isomers, and utilizes robust reagents like Dess-Martin or Swern oxidation which are manageable in large reactors, facilitating commercial scale-up of complex nucleosides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
The technical superiority of the synthesis route described in CN101628927A underscores the importance of partnering with a manufacturer who possesses both the intellectual understanding and the physical infrastructure to execute it flawlessly. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with stringent purity specifications and rigorous QC labs that ensure every batch of Gemcitabine intermediate meets the highest global standards. We understand that in the oncology sector, there is no margin for error, and our commitment to quality is embedded in every step of our operation.
We invite pharmaceutical companies and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can optimize your supply chain. By leveraging our expertise in stereoselective synthesis, we can help you secure a stable, high-quality supply of this critical API intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
