Industrial Scale Synthesis of 5-Aza-2'-deoxycytidine via Novel Silanization and Halobenzoyl Protection
Industrial Scale Synthesis of 5-Aza-2'-deoxycytidine via Novel Silanization and Halobenzoyl Protection
The pharmaceutical landscape for oncology treatments has been significantly shaped by the development of hypomethylating agents, with 5-Aza-2'-deoxycytidine, commonly known as Decitabine or NSC 127716, standing as a cornerstone therapy for myelodysplastic syndromes (MDS). As detailed in patent CN101570559B, the demand for efficient, scalable, and high-purity manufacturing routes for this critical active pharmaceutical ingredient (API) has never been more pressing. The patent discloses a robust methodology that addresses the longstanding challenges associated with nucleoside analog synthesis, specifically focusing on the stereochemical control and purification bottlenecks that have historically plagued commercial production. By leveraging a unique combination of silanization activation and halobenzoyl protection strategies, this technology offers a pathway to produce Decitabine intermediates with superior reproducibility and reduced toxicity profiles. For global procurement teams and R&D directors, understanding the nuances of this synthetic route is essential for securing a reliable supply chain for this life-saving medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-deoxynucleosides like Decitabine has been fraught with significant technical hurdles that impede cost-effective industrialization. Early methodologies, such as those reported by Pliml et al., relied on cyclization reactions that inherently produced complex mixtures of alpha and beta anomers, necessitating difficult and yield-loss-inducing separation processes. Furthermore, alternative routes described in literature often utilized starting materials like 2-deoxyribosyl derivatives that are notoriously difficult to source and stabilize, leading to supply chain fragility. Perhaps most critically for large-scale manufacturing, many traditional protocols, including those by Michael W. and others, depended heavily on silica gel column chromatography for the purification of intermediates. While acceptable in a laboratory setting, column chromatography is economically and logistically prohibitive at the multi-kilogram or tonne scale due to excessive solvent consumption, low throughput, and the generation of substantial hazardous waste. These legacy methods also frequently suffered from low overall yields and poor stereocontrol, resulting in high levels of the inactive alpha-isomer which complicates downstream processing and increases the cost of goods sold (COGS).
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in CN101570559B introduces a streamlined synthetic strategy that fundamentally alters the economic equation of Decitabine production. The core innovation lies in the use of specific halobenzoyl protecting groups on the sugar moiety combined with a silylated base activation. This approach not only simplifies the reaction conditions but also dramatically improves the diastereoselectivity of the glycosylation step. Unlike prior art that yielded intractable mixtures requiring chromatographic separation, this novel route produces a crude product where the desired beta-isomer constitutes more than 50% of the mixture. This enrichment allows for the isolation of the target compound through simple, scalable recrystallization techniques using common solvents like toluene or methanol. By eliminating the need for column chromatography entirely, the process reduces solvent usage, minimizes waste generation, and significantly shortens the production cycle time. The result is a manufacturing protocol that is not only chemically elegant but also commercially viable, offering a clear path to high-purity API production with excellent reproducibility.
Mechanistic Insights into Silanization-Assisted Glycosylation
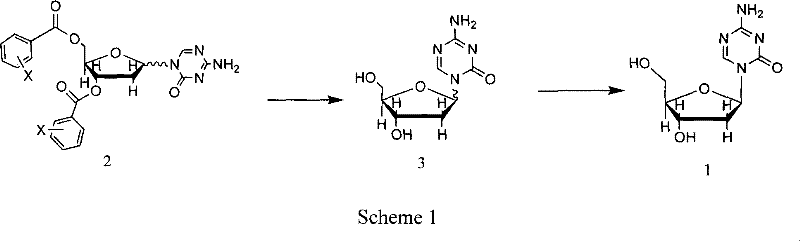
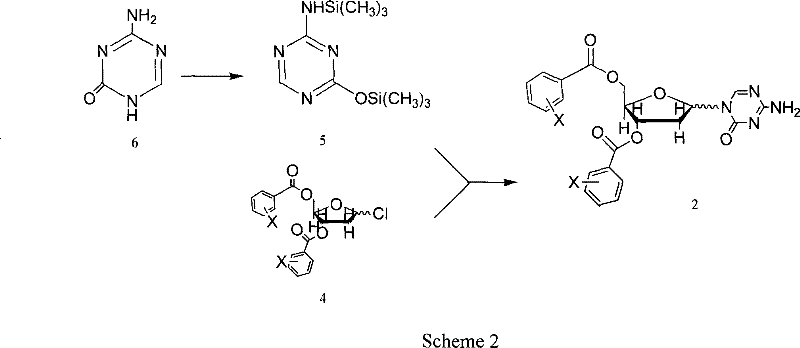
The chemical elegance of this process is rooted in the precise manipulation of nucleophilicity and electrophilicity through silylation and Lewis acid catalysis. The synthesis begins with the activation of 5-azacytosine, a relatively weak nucleophile, via treatment with hexamethyldisilazane (HMDS) and a catalytic amount of trimethylchlorosilane (TMCS). This silanization step converts the exocyclic amine and ring nitrogens into highly reactive silyl derivatives, effectively increasing their nucleophilic character and solubility in organic media. This activated species is then subjected to a glycosylation reaction with 1-chloro-3,5-di-O-halobenzoyl-2-deoxyribose. The presence of the halobenzoyl groups at the 3' and 5' positions serves a dual purpose: they protect the hydroxyl groups from unwanted side reactions and exert a steric influence that aids in directing the stereochemical outcome of the glycosidic bond formation. The reaction is promoted by trimethylsilyl trifluoromethanesulfonate (TMSOTf), a potent Lewis acid that facilitates the departure of the chloride leaving group on the sugar, generating an oxocarbenium ion intermediate that is rapidly attacked by the silylated base.
Following the coupling reaction, the crude intermediate undergoes a critical deprotection and isomer enrichment phase. The removal of the acyl-oxygen protecting groups is achieved under mild alkaline conditions, typically using sodium methoxide in methanol. This transesterification reaction cleaves the benzoyl esters to reveal the free hydroxyl groups of the ribose sugar, yielding the final nucleoside. Crucially, the specific choice of the halobenzoyl protecting group appears to influence the crystallization behavior of the resulting diastereomers. The patent data indicates that the beta-anomer, which is the therapeutically active form, can be selectively crystallized from the reaction mixture, leaving the alpha-anomer in the mother liquor. This mechanism of impurity control via differential solubility is far superior to chromatographic separation, as it leverages thermodynamic stability rather than kinetic adsorption differences. The final product exhibits high chemical purity, with related substances controlled to less than 1.5% by HPLC, and can be further purified to exceed 99.5% purity through additional recrystallization steps, ensuring compliance with stringent pharmacopeial standards.
How to Synthesize 5-Aza-2'-deoxycytidine Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly temperature control and moisture exclusion, to maximize yield and stereocontrol. The process is designed to be telescoped where possible, minimizing the handling of unstable intermediates. For R&D teams looking to replicate or scale this chemistry, the following overview outlines the critical operational phases derived from the patent embodiments. It is important to note that while the chemistry is robust, the quality of the starting materials, particularly the halobenzoyl protected sugar, is paramount for achieving the reported beta-selectivity. The detailed standardized synthesis steps see the guide below.
- Activate 5-azacytosine via silanization using hexamethyldisilazane (HMDS) and trimethylchlorosilane (TMCS) under reflux to enhance nucleophilicity.
- Perform glycosylation by reacting the silylated base with 1-chloro-3,5-di-O-halobenzoyl-2-deoxyribose using TMSOTf as a catalyst in dichloroethane.
- Hydrolyze the protected intermediate using sodium methoxide in methanol, followed by recrystallization to isolate the beta-isomer with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from legacy chromatography-dependent methods to this recrystallization-based process represents a significant opportunity for cost optimization and risk mitigation. The elimination of silica gel columns removes a major bottleneck in production throughput, allowing for larger batch sizes and faster turnaround times without the need for specialized equipment or vast quantities of chromatographic solvents. This structural change in the manufacturing process translates directly into a more resilient supply chain capable of meeting fluctuating market demands for oncology drugs. Furthermore, the use of commodity reagents such as HMDS, TMCS, and halobenzoyl chlorides ensures that raw material sourcing remains stable and cost-effective, insulating the production schedule from the volatility often associated with specialty fine chemicals.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of column chromatography from the purification workflow. In traditional nucleoside synthesis, chromatography can account for a substantial portion of operating expenses due to the high cost of silica gel, the massive volumes of organic solvents required for elution, and the energy costs associated with solvent recovery and disposal. By replacing this unit operation with simple filtration and recrystallization, the process drastically reduces variable costs per kilogram. Additionally, the higher yield of the desired beta-isomer directly from the reaction mixture means that less starting material is wasted on producing inactive byproducts, thereby improving the overall atom economy and reducing the effective cost of the active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex purification steps that are prone to failure or variability. The robust nature of this silanization-glycosylation route, which relies on well-understood chemical principles and stable intermediates, significantly lowers the risk of batch failures. The ability to purify the product via recrystallization using common solvents like methanol and toluene means that production is not dependent on the availability of specialized eluents or resin packs. This flexibility allows manufacturers to maintain consistent production schedules even during periods of raw material constraint, ensuring a steady flow of high-quality intermediates to downstream API producers and ultimately to patients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers distinct advantages that align with modern green chemistry initiatives. The reduction in solvent volume associated with avoiding chromatography leads to a smaller environmental footprint and lower costs for waste treatment and disposal. The process is inherently scalable, as demonstrated by the patent's examples which describe reactions on the hundred-gram to kilogram scale without loss of efficiency. This scalability ensures that the technology can be seamlessly transferred from pilot plants to commercial-scale reactors (100 MT range) without the need for re-engineering the purification strategy. The use of less toxic reagents and the generation of simpler waste streams further facilitate regulatory compliance and permit acquisition for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decitabine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these specifics is crucial for making informed decisions about technology transfer and vendor qualification.
Q: What is the purity profile of the Decitabine produced via this method?
A: The patented process achieves high purity with related substances measuring less than 1.5% by HPLC, and further recrystallization can reduce impurities to below 0.5%.
Q: Does this synthesis require column chromatography for separation?
A: No, a key advantage of this method is that it avoids the need for column chromatography, relying instead on standard recrystallization techniques which are more suitable for large-scale manufacturing.
Q: What is the stereochemical outcome of the glycosylation step?
A: The reaction yields a mixture of diastereomers where the desired beta-configuration content is greater than 50%, facilitating easier isolation of the target isomer compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aza-2'-deoxycytidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology APIs like Decitabine requires more than just a patent license; it demands a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch conforms to the highest international standards. Our facility is equipped to handle the specific requirements of nucleoside chemistry, including moisture-sensitive reactions and cryogenic conditions, guaranteeing the integrity of the final product.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this chromatography-free method. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics against your internal benchmarks. Let us collaborate to secure a sustainable and cost-effective supply of this critical pharmaceutical intermediate.
